Brain Power: Mind Control of External Devices
A person moving mouse cursor on a computer screen is an insignificant accomplishment. Heck, even a monkey can do it.
But if the person is a quadriplegic controlling the cursor with nothing but brainwaves, now that would be interesting. And it's been done.
Brain-computer interface systems, although still in developmental and experimental stages, are becoming increasingly more powerful and applicable. Advancements in neural signaling research make it one of the hottest fields in biomedical engineering.
Better sensing systems
Earlier this year, researchers indeed trained four people suffering epilepsy to move a computer cursor with the power of thought. The patients, who were waiting to have brain surgery, were already fitted with small sheets of signal-detecting electrodes on the surfaces of their brains.
The patients were asked to perform certain tasks - such as opening and closing their hands and sticking out their tongue - while scientists determined what brain signals were associated with these movements.
Next, the signals from these movements were matched up with movements of the cursor on the screen. For example, the thought of opening of the right hand might move the cursor to the right. The subjects were then asked to move the cursor from one spot to another on the screen by thinking about making the movements.
Sign up for the Live Science daily newsletter now
Get the world’s most fascinating discoveries delivered straight to your inbox.
The patients had some difficulty at first, but each was able to control the cursor with their thoughts and with over 70 percent accuracy after a few minutes. One patient was operating at 100 percent accuracy by the end of the trial.
"All our subjects were able to control the computer cursor using imagined representations of motor movements," said Daniel Moran of Washington University.
This study was the first to prove that sensors placed on the surface of the brain are preferable to the standard forms of sensors - either embedded deep in the brain tissue or worn as a cap. They are less intrusive than an embedded variety and potentially more stable and powerful than the cap, which receives weak brain signals that have passed through the skull.
The BrainGate
Only a handful of clinical studies include quadriplegics as participants. One at Brown University and Cyberkinetics Neurotechnology Systems, Inc., is working to develop a system called BrainGate.
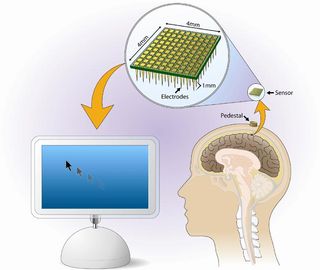
In this-one patient pilot study, a sensor is implanted on the surface of the primary motor cortex, the area of the brain responsible for movement. The sensor, smaller than a penny, has hair-thin electrode probes that penetrate about a millimeter into the brain and are designed to pick up electrical impulses from the motor neurons.
About twice a week the participant performs cursor-moving tasks with his thoughts that are meant to demonstrate proof of principle of the technology and to evaluate the quality, type, and usefulness of neural output control that patients can achieve.
BrainGate offers several advantages over other systems, its creators say.
"First, BrainGate provides an interface with a computer that works immediately, without weeks or months of training," John Donoghue, director of Brown's Brain Science Program and a co-founder of Cyberkinetics Neurotechnology Systems Inc., told LiveScience. "Secondly, a user can operate the device without requiring great concentration.
Cursor control is "about as natural as using one's own arm," Donoghue said. The patient can, for example, carry on a conversation while moving the cursor.
"And, thirdly, because BrainGate connects directly to the part of the brain that ordinarily controls hand movement and gestures, it provides significantly more utility than devices that rely on 'substitutes' for the brain's own arm movement signal, such as eye movements. Using eye movements, for example, to control a computer prevents one from looking elsewhere during use -- something that is very unnatural and cumbersome."
The goal of Donoghue's study and its follow-ups is to develop a safe, effective, and unobtrusive universal system for physically disabled people to control a wide array of devices, such as computers and wheelchairs, with their thoughts.
"Moving a wheelchair with BrainGate is beyond the scope of this study," said Donoghue. "Although, our first trial participant has used his thoughts to control a TV and move a robotic hand and arm."
Monkey see, monkey do
Since it can difficult to get government permission to run human clinical trial of this kind, the bulk of the research for brain/machine interfaces has been done with monkeys.
In 2003, researchers at Duke University taught rhesus monkeys to consciously control the movement of a real time robotic arm using only feedback from a video screen and their thoughts. The monkeys appeared to operate the robotic arm as if it were their own limb.
A team led by neurobiologist Miguel Nicolelis implanted a tiny array of brain-signal-detecting microelectrodes in the monkey's brain. They trained the monkey with a joystick that moved a cursor on a screen, and, once the monkey had mastered that task, the robotic limb was added to the feedback loop. After a few days, the monkey realized the connection between moving the cursor and moving the arm.
Once that connection was established, the researchers removed the joystick. The monkey slowly figured out that it could still move the cursor and robotic arm by moving its own arm. After a few days without the joystick, the monkey realized that it could move the robotic arm without moving its own.
It had mastered a neuroprosthetic limb.
"[The monkey's] arm muscles went completely quiet, she kept the arm at her side and she controlled the robot arm using only her brain and visual feedback," said Nicolelis. "Our analyses of the brain signals showed that the animal learned to assimilate the robot arm into her brain as if it was her own arm."
The brain circuitry had actively reorganized itself to incorporate an external device.
"Actually, we see this every day, when we use any tool, from a pencil to a car," said Nicolelis. "As we learn to use that tool, we incorporate the properties of that tool into our brain, which makes us proficient in using it."
Andrew Schwartz, a neurobiologist at the University of Pittsburgh School of Medicine, has taken the progress made at Duke to another level. Schwartz has trained monkeys to feed themselves pieces of fruit using just their brainwaves and a robotic arm.
Unlike the experiment at Duke, where the robotic arm wasn't in close proximity to the monkey, Schwartz has restrained the monkey's arm and placed the robotic limb as close to the monkey as possible.
The robotic arm moves like a normal arm - it has fully mobile shoulder and elbow joints. The "hand" is a simple gripper that allows the monkey to grab its food.
"The robotic limb takes the desired hand position as input and has on-board hardware that controls the torque motors to move the limb to the desired decoded position, Moran explained. "This is really no different than sending a desired cursor position to a computer."
Schwartz has recently made improvements to the computer algorithms that will make it easier for the monkeys to learn to operate the robotic arm. The improvements will also help Schwartz and his team to develop brain devices with smoother, more responsive, and precise movements.
Looking to the future
Commercial application of brain-computer interface systems is still years away, and developers have set the bar high.
Moran would like to return movement to the body, saying his "overall research goal is to transmit cortical signals over a break in a spinal cord." He would also like to see the development of better neuroprosthetic limbs.
Donoghue has similar goals for the near-term usage of the BrainGate. In addition to developing a smaller, wireless device for the patient to wear, he is demonstrating that human brain waves could be used to control neuroprosthetic limbs.
"The ultimate goal of the neuromotor prosthesis is to use physical systems - smart sensors and implantable electronics - to restore a considerable degree of function to paralyzed limbs," Donoghue said. A neural sensing system with adequate processing of signals could potentially drive muscles through implanted stimulators."
"[For the] long term, we want to develop a system that allows humans with paralysis to move their limbs in a way that they can carry out useful movements and lead independent lives. And we want to develop a new class of neurotechnologies that can diagnose and treat disease and restore lost functions in humans," said Donoghue. "These are ambitious goals, but we believe they are realizable."
Ancient Behaviors Hard-Wired in Human Brain
Your Brain Works Like the Internet
