
'A cure on the horizon': Are we finally close to ending type 1 diabetes?
It's a running joke that a cure for type 1 diabetes is only five years away and has been for 50 years, but new stem cell trials and immune drugs hint that we're closer than ever to a functional cure for the disease.
My first clue that something was wrong was the smell of cotton candy.
It was March 15, 2016, and I was changing my toddler's sheets because he had soaked through his diaper. The bed was wet, but it didn't smell like urine. Other details I'd brushed off came to mind, and I began to worry.
He'd lost his chubby baby cheeks. He'd been finishing his morning sippy cup of water, rather than flipping it over and cackling in glee. And he seemed a bit more tired.
I called the pediatrician, who tested my son's urine. It showed dangerously high sugar levels. After a harrowing trip to the hospital, we had a diagnosis. My son had type 1 diabetes, like 2 million children and adults in the U.S., which meant his rogue immune system had destroyed almost all of the insulin-making beta cells in his body. He was 1 year old.
A decade later, he is a thriving sixth grader.
But in his own words, "diabetes really sucks."
"There are times when I can't eat because my blood sugar is high. Sometimes, when I'm really low, I'm scared that I'm going to die," he told me.
Get the world’s most fascinating discoveries delivered straight to your inbox.
If he lives to old age, he will have changed the sensor that measures his blood sugar and the "site" — the tube that delivers insulin — tens of thousands of times, will have spent more than half a million hours with high or low blood sugar, and will have incurred millions of dollars of medical care. Getting off this blood sugar roller coaster for good would dramatically reshape his future.

Science Spotlight takes a deeper look at emerging science and gives you, our readers, the perspective you need on these advances. Our stories highlight trends in different fields, how new research is changing old ideas, and how the picture of the world we live in is being transformed thanks to science.
"I could get more sleep. And I wouldn't have to do any site or sensor changes, or get shots," my son said. "It would be a better life."
There's a running joke in the type 1 diabetes community that a cure is "only five years away" — and has been for at least 50 years.
But there may be a kernel of truth in it now.
The first treatments to replace destroyed beta cells are in late-stage trials that will likely conclude in the next few years. And scientists are developing innovative ways to keep those transplanted cells alive without harsh side effects.
Many hurdles remain before such treatments can be widely used in kids with the disease. But a picture of a post-type-1-diabetes (T1D) world is emerging. Not too far off, those who have dangerously erratic blood sugars or who already need to suppress their immune systems for other reasons may be freed of the disease with stem cell transplants. And after that, otherwise healthy adults and kids may receive stem cell transplants.
"Cell replacement therapies are our flagship program because we believe it's imminent to provide a functional cure for people with type 1 diabetes," meaning the symptoms of the disease are eliminated, said Dr. Sanjoy Dutta, chief scientific officer at Breakthrough T1D, a nonprofit that supports research and advocacy to prevent, treat and cure the disease.

My son was not yet 2 years old when he was diagnosed with diabetes. Soon after diagnosis, he was prescribed a continuous glucose monitor (CGM), which he wears all the time. In this photo, you can see the outline of the CGM under his sleeve.
From death sentence to chronic disease
My son's most noticeable symptom — the sweet smell of his urine — was a textbook sign of the disease. "Diabetes mellitus" (the full name of the disease) means "sweet siphon" in Greek, and its symptoms were first described 3,500 years ago in ancient Egypt.
We now know type 1 diabetes occurs when the immune system mistakenly destroys insulin-producing beta cells in the pancreas, which are clustered in cells called islets.
As a result, the body can't move glucose into tissue. Instead, glucose piles up in the blood, and the kidneys go into overdrive as they try to flush it out. Meanwhile, muscle and fat tissue starves. Without glucose for fuel, the body breaks down fat for energy and turns it into compounds called ketones. This water-intensive process dehydrates the body even further, and these ketones accumulate in the blood, turning it acidic.
A person with untreated type 1 diabetes will be ravenous and thirsty. As they eat and drink, and pee and pee, their body will slowly devour itself as their fat and muscle melt away. Without treatment, a person with type 1 diabetes will invariably go into a coma and die.

Thanks to continuous glucose monitors, people with diabetes can get blood sugar readings up to every minute.
The solution to this problem is insulin. In 1921, Frederick Banting and Charles Best isolated insulin from pancreatic islet cells in dogs and eventually devised a way to make insulin from the pancreas of a cow.
Science had transformed type 1 diabetes from a death sentence to a chronic disease — an accomplishment that earned Banting the 1923 Nobel Prize in medicine.
We've come a long way since then. People now routinely live decades after their diagnosis. Scientists have made faster-acting insulin, designed insulin pumps that automate the delivery of the drug, and created continuous glucose monitors that reveal blood sugar levels up to every minute. These innovations improve average blood sugar levels and reduce complications.
Even five years ago, I wouldn't have predicted we were as far along as we are.
Lori Sussel, director of basic and translational research at the Barbara Davis Center for Diabetes at the University of Colorado Anschutz Medical Campus
But none of these advancements has fundamentally changed the relentless nature of the disease. Injected insulin doesn't work as fast as the version the body produces. And because dozens of factors — including diet, exercise, stress, illness and puberty — can affect how much insulin the body needs, patients are always at risk of under- or overdosing insulin.
"This is a disease that requires 24/7 superhuman management," Dutta told Live Science.
People who inject insulin typically face hours of high and low blood sugar every day. Decades of high blood sugar can destroy the kidneys, damage nerves, and cause blindness and heart attacks. Critically low blood sugar can kill within hours, as the brain is starved of energy.
Despite the medical advances, type 1 diabetes still shortens life expectancy by more than a decade, on average.
Replacing lost cells
The reductions in both the length and quality of life have pushed scientists to continue seeking a cure for diabetes. Merely managing the disease isn't enough.
Since the 1990s, more than 1,000 people worldwide have received transplants of islet cells from cadavers; in the U.S., this remains an experimental procedure, so it is done only in clinical trials. Some of the islet-cell recipients have been cured of the disease — at least for a time.
But the donor pancreases the islet cells are taken from are in very short supply. And cells can be damaged, either before or during harvesting, so their quality is often poor. As a result, cadaveric islet cell transplants aren't very effective; five years after transplantation, only 1 in 10 people who receive them don't need to inject insulin.
Another challenge is that recipients must take immune-suppressing drugs for life. As a result, cadaveric transplants are mostly offered to people who already need another type of transplant, such as a kidney transplant.
For islet transplants to represent a viable cure, they need to be easy to mass-produce in consistently high quality, and scientists must prevent the immune system from destroying the islet cells.
We are getting very close to that first goal: producing high-quality beta cells from scratch.
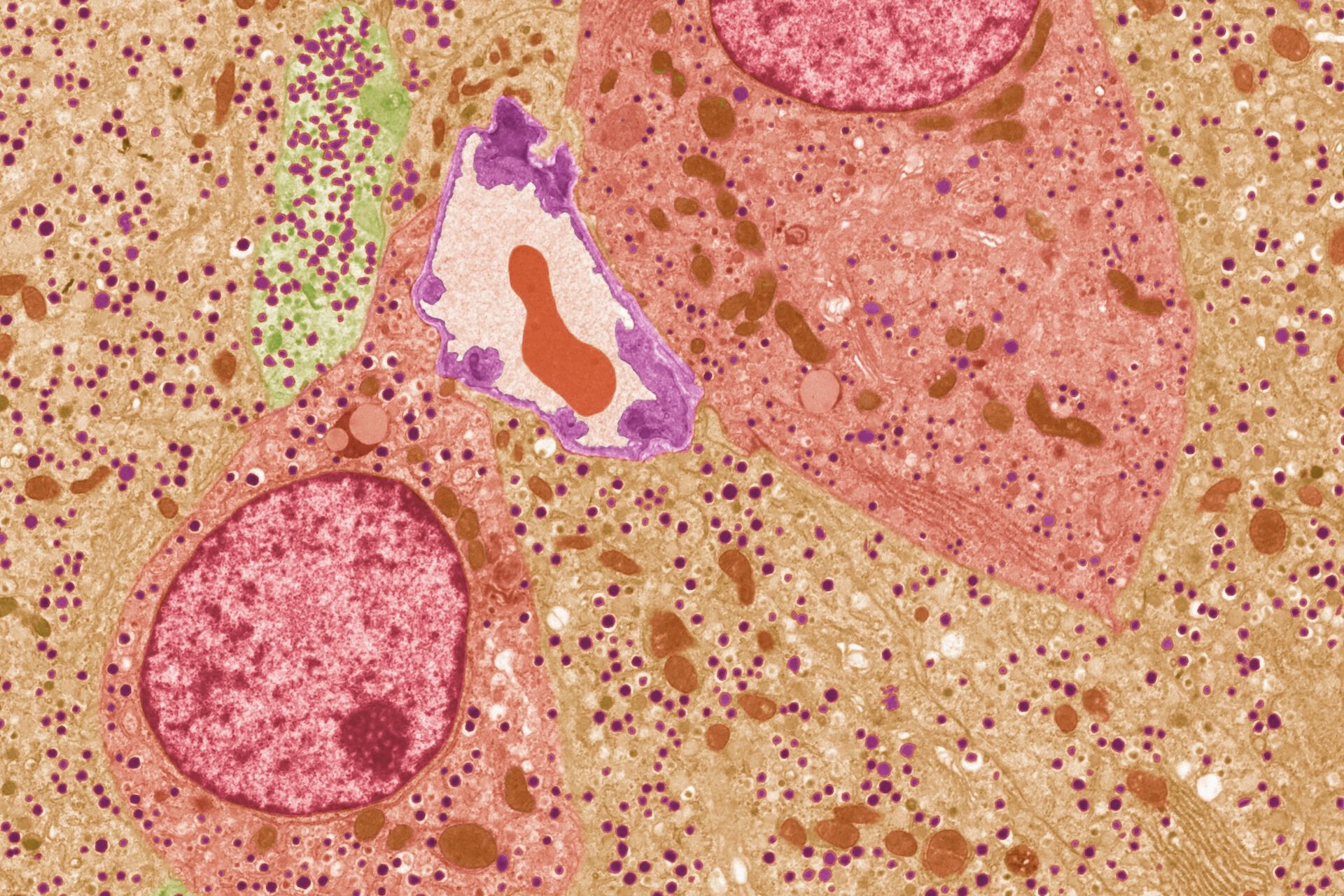
Pancreatic islet cells under a microscope. Islets are bundles of cells that produce a variety of hormones, including the blood-sugar-lowering hormone insulin.
From embryonic cell to beta cell
In the past several years, scientists have made incredible progress in deciphering the chemical symphony that directs pluripotent embryonic stem cells — cells that can become any type of cell in the body — to morph into islets.
"Even five years ago, I wouldn't have predicted we were as far along as we are," said Lori Sussel, director of basic and translational research at the Barbara Davis Center for Diabetes at the University of Colorado Anschutz Medical Campus, who does basic islet research using mouse and human stem cell models.
Some treatments built on this research are already in late-stage development. For instance, Vertex Pharmaceuticals, a Boston-based biotech company, reported in 2025 in the The New England Journal of Medicine (NEJM) that 10 out of the 12 patients who received the company's embryonic stem-cell-derived product, called VX-880, do not have to take insulin a year out from their transplant. And all of them are making some insulin, Felicia Pagliuca, senior vice president for cell and genetic therapy research at Vertex, told Live Science.
(The two patients who are still insulin-dependent took steroids during treatment against study instructions. Steroids are known to raise blood sugar, which can damage beta cells, the authors of the NEJM study wrote.)
The trial started out with 14 participants, but two people died during it. However, the study authors, and presumably the safety monitoring boards for the trial, concluded that those deaths were unrelated to the treatment. Nonetheless, as with any potential treatment, more data is needed to see if further issues crop up among patients undergoing these transplants.
Immunosuppression is worse than having to take insulin every day.
Lori Sussel, director of basic and translational research at the Barbara Davis Center for Diabetes at the University of Colorado Anschutz Medical Campus
The company is doing just that, by testing VX-880 in a larger trial that will enroll 50 patients with hypoglycemia unawareness, meaning they can't physically feel when their blood sugar gets dangerously low. These patients face a higher baseline mortality risk, Sussel said. The trial will need a year's worth of data before it can be submitted to the Food and Drug Administration, Pagliuca said. But if those results are positive, that would mean a scalable source of reliable stem cells could be available for some people with type 1 diabetes.
Vertex isn't the only team working on reprogramming cells to become islets. Researchers in China have reprogrammed a woman's own fat cells to become beta cells. One advantage of this approach is that those cells aren't immediately recognized as foreign, because they come from the patient's own body. A downside, however, is that creating a treatment that is unique to each individual is likely to be cost-prohibitive as a broad-scale cure, Sussel said.
Immune invisibility cloak
Creating the stem cells is just the first step. Once they're in the body, they have to survive. That has historically required strong immunosuppressive drugs, just as other transplants do.
Immunosuppressive drugs — such as tacrolimus and sirolimus — leave people susceptible to infections and can damage the kidneys, and may increase the risk of cancer. Tacrolimus is also toxic to the transplanted islet cells themselves, said Dr. Piotr Witkowski, director of the Pancreas and Pancreatic Islets Transplantation Program at UChicago Medicine.
But the average kid with type 1 diabetes has decades of relatively healthy life ahead of them, so putting their heart and kidneys at risk with immunosuppressive drugs presents a higher risk than benefit.
"Immunosuppression is worse than having to take insulin every day," Sussel told Live Science. Thinking about it for my own son, I can't help but agree; diabetes is a huge hassle, but running the risk of fatal infections or higher cancer rates down the line to eliminate that hassle doesn't seem worth it.
Scientists are exploring several ways to solve the immune attack problem.

While many children now have their blood sugar monitored via a CGM attached to the body, many people still use older technology to spot check their blood sugar. That involves doing a finger stick to test their blood using a blood glucose monitor.
One is to make transplanted beta cells "invisible" to the immune system. To do that, one company is taking a page from pregnancy. During pregnancy, 50% of the genes — and, therefore, proteins — in the fetus come from the father, said Dr. Sonja Schrepfer, scientific founder of Seattle-based Sana Biotechnology (Hypoimmune Platform) and researcher-scientist at Cedars Sinai Medical Center in Los Angeles.
So Schrepfer's team studied which molecules help shield the fetus from immune attack to sustain the pregnancy.
They identified a few. Two molecules act as unique fingerprints present on the outside of every cell in the body. When the immune system detects a foreign fingerprint, it destroys the cells carrying it. During pregnancy, the fetus dials down how many of these fingerprints are made, especially in the placenta, Schrepfer said. So, to keep transplanted islet cells safe from the immune system, "we have to get rid of that fingerprint," Schrepfer told Live Science.
But that's just the first step in preventing rejection. Certain immune cells, called natural killer cells and macrophages, constantly patrol the body and will automatically kill cells without a fingerprint, she said.
Scaling it up is going to be very expensive, and so I think right now, that's probably one of the rate-limiting steps, is the expense.
Lori Sussel, director of basic and translational research at the Barbara Davis Center for Diabetes at the University of Colorado Anschutz Medical Campus
However, Schrepfer's research showed that cells naturally produce a special molecule, at low levels, which essentially screams "Don't kill me." When cells overexpress this molecule, the macrophages and natural killer cells come close to transplanted cells but then drift away, leaving the cells alone, Schrepfer said.
So Sana Biotechnology has used the gene-editing cut-and-paste tool CRISPR, as well as a harmless virus, to genetically modify beta cells collected from a cadaver to lack the cell fingerprints and to overexpress the "don't kill me" molecule. These "hypoimmune" cells are, in theory, tolerated by the immune system.
In August 2025, Sana reported that it had treated the first patient with these cells earlier that year, and in March 2026 said the cells were still evading the immune system and making insulin . While larger safety and efficacy trials are still a ways off, the new results raise hopes that this approach will lead to stem-cell-derived beta cells that don't require immune suppression.
Vertex Pharmaceuticals is also investigating gene-edited stem cell transplants that can evade immune detection, but the company has not tested them in humans yet, Pagliuca said.
Gentler drugs
Other ways to prevent transplant rejection are under development. Unlike the gene-edited islet cells, these methods rely on a more established drug approval pathway.
One is a drug called tegoprubart, which was initially developed to prevent kidney transplant rejection. (It is also in trials as a treatment for delaying ALS progression.)
When immune cells first recognize a foreign protein, they need a second signal to mobilize the rest of the immune system to attack, Witkowski told Live Science. Tegoprubart silences that second "attack signal" by inactivating a molecule called CD40 ligand, he said.
In a study involving 12 patients, Witkowski and his colleagues transplanted cadaveric islet cells and gave the participants tegoprubart instead of the standard-of-care drug, tacrolimus. Tegoprubart led to greater beta cell survival than is typical with tacrolimus.
In March, Witkowski presented results at a diabetes conference showing that all 10 patients who received transplants more than four weeks earlier are so far are insulin-free and as of yet, none of the participants have experienced severe side effects or rejection.
Later this year, the team is also planning to start a 10-person trial with Breakthrough T1D to test tegoprubart in those with kidney dysfunction and T1D — a group that has historically been prevented from undergoing these transplants, Witkowski said.
Moreover, tegoprubart is just one of several gentler immune-suppressing protocols that are currently being studied, Dutta said. Others include antithymocyte globulin and teplizumab, a drug that has been demonstrated to prevent the progression of early-stage type 1 diabetes, he added.
I would hate to say we're five years away. What I would say is that there is a cure on the horizon.
Lori Sussel, director of basic and translational research at the Barbara Davis Center for Diabetes at the University of Colorado Anschutz Medical Campus

For my son, knowing researchers are working on a cure for type 1 diabetes gives him hope.
Hope on the horizon
Type 1 diabetes research is littered with dozens of once-promising cures that ultimately fizzled, so it pays to be cautious about these new approaches.
Indeed, there are still some kinks to work out. For instance, when embryonic stem cells are converted to beta cells, most of the resulting cells do produce insulin, but some also make other hormones, such as glucagon, which raises blood sugar, and somatostatin, which inhibits growth hormone, Sussel said. Plus, the process for making these cells is laborious and time-consuming.
"Scaling it up is going to be very expensive, and so I think right now, that's probably one of the rate-limiting steps, is the expense," Sussel said.
And, of course, longer-term studies will need to pin down how long stem cell transplants will last and whether they need to be readministered periodically. There could also be side effects that take years or decades to emerge. As a parent, I'd have to be confident the deaths seen in the Vertex trial were completely unrelated to treatment before I'd be on board.
Everyone I spoke with was wary of pinning a time frame to a post-diabetes future.
"I would hate to say we're five years away," Sussel said. "What I would say is that there is a cure on the horizon."
But there was a palpable excitement among the researchers I spoke with, all of whom said breakthroughs are coming at a rapid pace.
A world where no one has type 1 diabetes is still a long way off. But little by little, a functional cure is coming for more and more people.
"Ideally all people receive it one day, but we're going to start small and then go big," Dutta said.
RELATED STORIES
My son realizes he probably won't see a cure for a while. But knowing something is coming gives him hope. And that would make it all the more heartbreaking if these treatments don't pan out.
"When I imagine myself as an adult, I don't see myself with diabetes," he said.
"I'll be very disappointed if these treatments don't work."

Tia is the editor-in-chief (premium) and was formerly managing editor and senior writer for Live Science. Her work has appeared in Scientific American, Wired.com, Science News and other outlets. She holds a master's degree in bioengineering from the University of Washington, a graduate certificate in science writing from UC Santa Cruz and a bachelor's degree in mechanical engineering from the University of Texas at Austin. Tia was part of a team at the Milwaukee Journal Sentinel that published the Empty Cradles series on preterm births, which won multiple awards, including the 2012 Casey Medal for Meritorious Journalism.
You must confirm your public display name before commenting
Please logout and then login again, you will then be prompted to enter your display name.

 Live Science Plus
Live Science Plus





