Facts About Boron


Boron is a multipurpose element. It's a crucial nutrient for plants, an important component in the nuclear industry and the main ingredient of a bizarre fluid called oobleck.
Perched next to carbon on the Periodic Table of Elements, boron is a metalloid, a substance with both metallic and nonmetallic properties. It's a complicated element, as Stony Brook University professor Artem Oganov told the New York Times in 2009.
"Boron is a truly schizophrenic element," Oganov said. "It's an element of complete frustration. It doesn't know what it wants to do. The outcome is something horribly complicated."
Compounds of boron, particularly borax, have been used by humans for thousands of years, according to Chemicool. Borax (sodium tetraborate) forms naturally during the evaporation of some salt lakes, according to AZoM, an online reference site for the engineering and materials science community. In the eighth century A.D., borax was exported from Tibetan lakebeds along the Silk Road for use by Arabic goldsmiths and silversmiths; it was also used to make ceramic glazes in China.
These early uses echo in borax's (and ultimately, boron's) name: The word is from the Arabic "buraq," or white. However, the element itself was not extracted until 1808, according to the Royal Society of Chemistry. Even then, chemists weren't able to get a pure form of boron. That goal was not reached until a century later, in 1909, when U.S. chemist Ezekiel Weintraub isolated 99 percent pure boron.
Just the facts
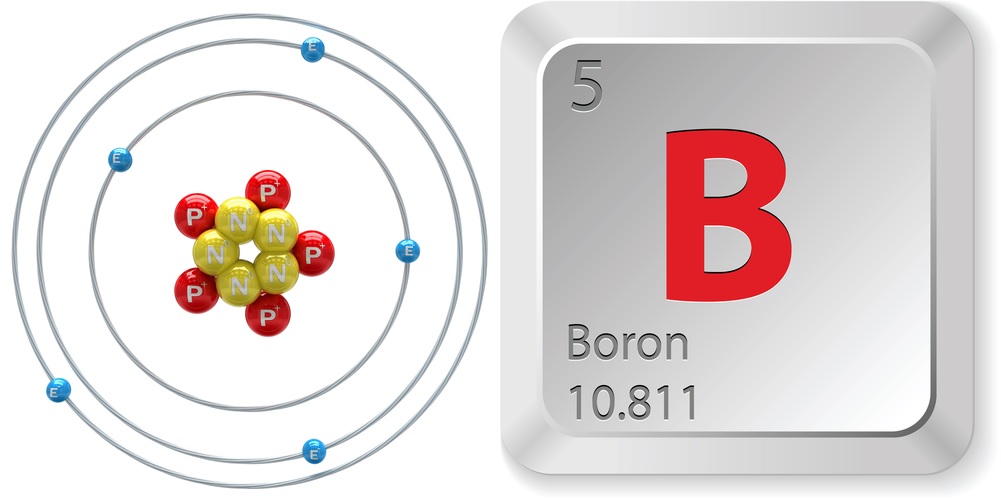
According to Jefferson Lab, the properties of boron are:
Get the world’s most fascinating discoveries delivered straight to your inbox.
- Atomic number (number of protons in the nucleus): 5
- Atomic symbol (on the Periodic Table of Elements): B
- Atomic weight (average mass of the atom): 10.81
- Density: 2.37 grams per cubic centimeter
- Phase at room temperature: Soil
- Melting point: 3,767 degrees Fahrenheit (2,075 degrees Celsius)
- Boiling point: 7,232 degrees F (4,000 degrees C)
- Number of isotopes (atoms of the same element with a different number of neutrons): 6
- Most common isotopes: B-10 (natural abundance 19.9 percent) and B-11 (natural abundance 80.1 percent)
Ooky oobleck
As part of borax, boron is a very common household element that is found in many detergents. (Pro tip: A borax-sugar solution will also kill ants!) It's also in the recipe for the science-fair goop oobleck, a liquid with some very strange properties. A mixture of borax solution and liquid glue creates a substance that is liquid when it is poured but solid when it is under pressure. Oobleck is a non-Newtonian fluid, which means that its viscosity depends on the shear force applied to it. The glue and oobleck join up to create long, thin polymer molecules. A "tap" or strong pressure forces the molecules in the fluid together, creating a solid. A slow movement like pouring or a gentle poke allows the molecules to flow against one another, making the oobleck behave like a fluid. The same concept is what makes Silly Putty both able to flow and bounce. (Oobleck can also be made with a mixture of cornstarch and water.)
But boron isn't all fun and games. The isotope boron-10 happens to be excellent at absorbing neutrons. This is extremely handy for nuclear fission, which is driven by careening neutrons knocking uranium atoms apart. The key to this process is balancing it so that every fission event triggers just one more fission event; otherwise, the reactions speed up like a runaway train and the reactor is said to go supercritical. Bad news all around.
To keep nuclear reactions in balance, reactors are equipped with devices called control rods, often made of boron or other elements, according to the U.S. Nuclear Regulatory Commission. Boron absorbs excess neutrons, preventing them from zinging into too many uranium atoms.
Who knew?
- Boron may have been the key to the evolution of life on Earth. The element stabilizes ribose, part of RNA, the self-assembling molecule that may have preceded DNA. (Viruses are essentially roving RNA strands.) A June 2014 study found that boron is present in the oldest rocks on Earth, which date back 3.8 billion years. This research proves that the early Earth had the ingredients needed to build RNA.
- Or maybe that first RNA got its boron from space. A 2013 study found that a Martian meteorite that landed in Antarctica contained 10 times the boron of any extraterrestrial object previously measured.
- Boron, in its crystalline form, is the second-hardest element behind carbon (in its diamond form), according to Chemicool.
- Unlike many elements, which form in fusion reactions within stars, boron formed after the Big Bang by a process called cosmic ray spallation. During this process, colliding cosmic rays split the nuclei of atoms, causing fission.
Current research
Boron doesn't have a lot of pop-culture cachet, but science has a lot to say about this surprisingly intriguing element. For example, plant biologists have long known that without boron, plants don't grow. The element is an essential nutrient.
But why? No one knew until August 2014, when researchers at the University of Missouri ferreted out the answer. Boron, they found, is crucial to the stem cells of plants. Portions of the plant called meristems are made of stem cells, which themselves are capable of giving rise to all the different cells that make up a plant. Without boron, these meristems wither, the researchers reported in the journal Plant Cell. In the eastern United States, farmers have to supplement their soil with boron in order to boost crop yields.
Boron may be a boon to tech as well. In July 2014, researchers discovered the first boron "buckyball," a cagelike structure similar to the soccer-ball-shaped carbon buckyballs often used in nanotechnology. Carbon nanostructures (known as fullerenes) were first discovered in the 1980s, and they prompted a wave of research in the hunt for other intriguing atom clusters.
"If you look at the main group elements, there is no better place to start than boron," said Lai-Sheng Wang, a chemist at Brown University who discovered the first boron buckyball. Boron atoms bond strongly with each other, Wang told Live Science, and the element has a very high melting point. It's tough stuff.
Wang and his colleagues began by putting clusters of boron atoms together to see the shapes they'd form as they bonded, a process that required them to use lasers to dislodge electrons from the cluster. The electron's ejection speed can then be used to determine how it was originally bonded in the atom cluster, allowing the researchers to map out the structure, which they call a borospherene.
When 39 or fewer boron atoms bond together, they form a flat structure. But at 40, the structure becomes a spherelike "cage," the researchers reported in the journal Nature Chemistry.
Wang and his team have also discovered that some of boron's flat structures could be very useful. In research published in January 2014 in the journal Nature Communications, the scientists found that 36 atoms of boron will form a disk with a perfect hexagonal hole in the middle — an arrangement that theoretically makes it possible to create a stable, one-atom-thick sheet of boron. If such a sheet can be created, it would be the boron equivalent of graphene, which is an atom-thick sheet of carbon molecules. Graphene holds a great deal of promise for tech because it is a cheap, strong, flexible conductor.
The boron version — or "borophene," as Wang and his colleagues are calling it — might have similar applications as an electrical and thermal conductor, but researchers aren't yet sure. They'll need to make real borophene sheets first, and then test their properties.
"Sometimes when you go the lab and make these things, nature has its own way," Wang said.
Follow Live Science @livescience, Facebook & Google+.
Additional resources
- Read more about experiments at Brown University to make a graphenelike material using boron.
- Find more facts about boron at the Royal Society of Chemistry.
- Learn about the history, sources and properties of boron from Los Alamos National Laboratory.

Stephanie Pappas is a contributing writer for Live Science, covering topics ranging from geoscience to archaeology to the human brain and behavior. She was previously a senior writer for Live Science but is now a freelancer based in Denver, Colorado, and regularly contributes to Scientific American and The Monitor, the monthly magazine of the American Psychological Association. Stephanie received a bachelor's degree in psychology from the University of South Carolina and a graduate certificate in science communication from the University of California, Santa Cruz.

 Live Science Plus
Live Science Plus





