Preeclampsia could be treated with 'blood filtering' therapy, early study hints
A blood-filtering therapy for preeclampsia is safe for pregnant patients and their babies, according to a new pilot study.
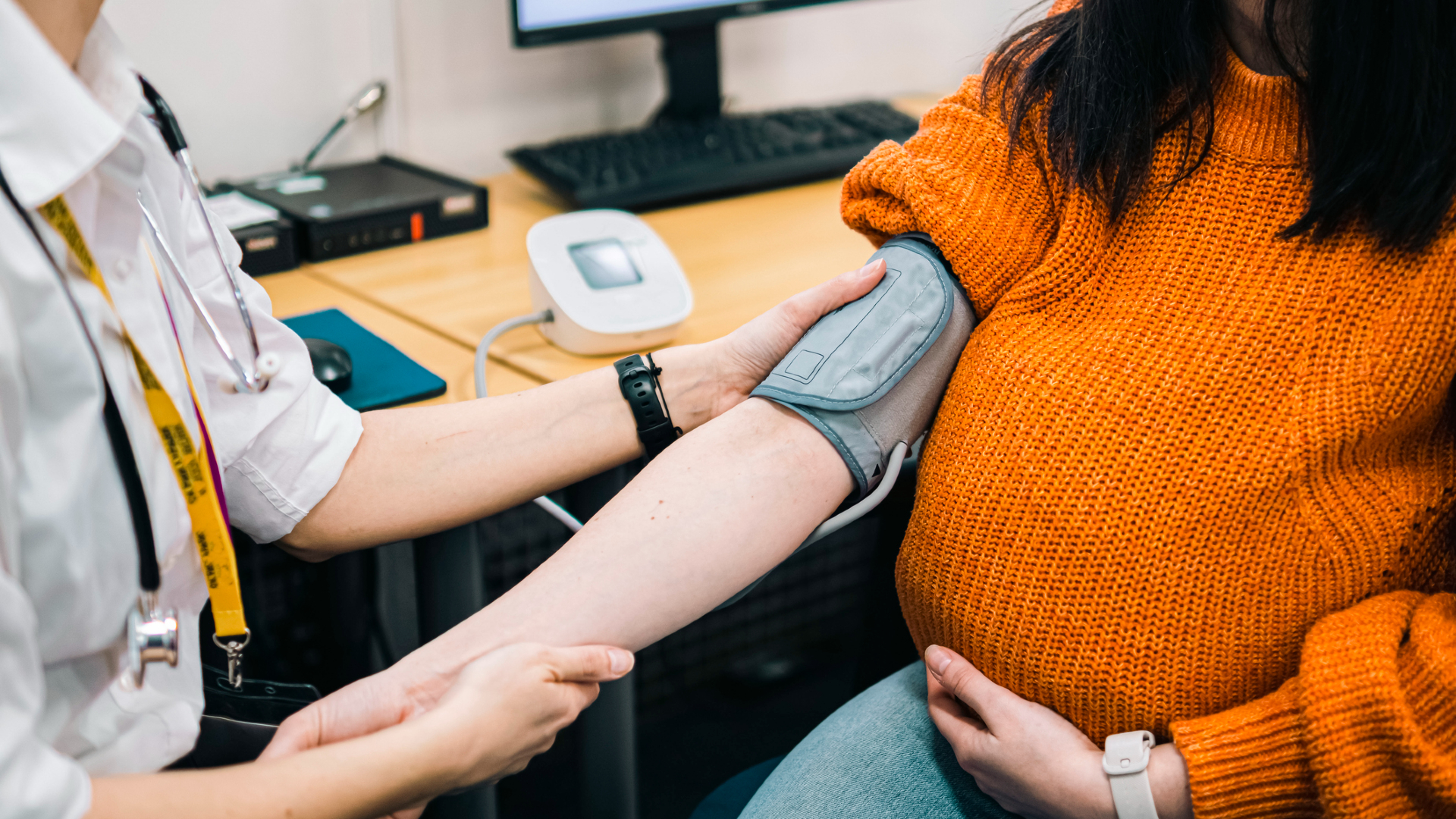
A blood-filtering technique could be a promising treatment for preeclampsia, a potentially deadly disorder of pregnancy that involves high blood pressure.
The new therapy is safe for both the pregnant person and the fetus, according to a new pilot study published Monday (April 27) in the journal Nature Medicine. Future trials will examine how well the treatment works, but early data hints that it can reduce circulating levels of a placental protein linked to the disease.
Although low-dose aspirin has been shown to reduce the risk of preeclampsia in high-risk patients, no intervention has been successful at treating the condition once it has been diagnosed.
Now, "we're finally on the verge of developing a targeted treatment for this condition," study co-author Dr. Ravi Thadhani, a nephrologist and chief medical officer at Cedars-Sinai Medical Center in Los Angeles, told Live Science.
A new avenue for treatment?
Currently, the only way to address preeclampsia is for the pregnant patient to give birth, although the condition can sometimes persist postpartum or even develop after birth.
Ideally, preeclampsia is managed and monitored until the baby reaches full term, at 37 weeks. But that's not always possible, meaning babies are often delivered prematurely. Preterm delivery can lead to health issues such as breathing problems and developmental disabilities, especially if the baby is born before 32 weeks gestation.
For the pregnant person, preeclampsia can cause dangerous damage to the liver, kidneys and heart, among other issues. And if not treated, it can lead to eclampsia, which involves seizures and can result in coma or death.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Thadhani hopes the blood-filtering treatment can "prevent this disease from getting worse and forcing the hand of the obstetrician to deliver" prematurely.
The new treatment relies on a technique called apheresis, in which blood is drawn and then returned after select components are removed. In this case, a protein called soluble fms-like tyrosine kinase-1 (sFlt-1) gets filtered from the bloodstream. sFlt-1 increases in the bloodstream during healthy pregnancies to regulate blood vessel formation around the placenta, but its levels often rise earlier and by excessive amounts in preeclampsia, causing vascular damage that may contribute to the condition.
Thadhani and colleagues previously demonstrated that the ratio of sFlt-1 to placental growth factor, another circulating protein, could predict the risk of preeclampsia, with a ratio of 40 or above tied to an especially severe form of the disease. Based on this finding, and additional studies from their lab and other groups, they reasoned that sFlt-1 might be good to target with a treatment.
They first administered the therapy to three pregnant baboons, observing that each treatment session decreased circulating sFlt-1 by about 50%. They then confirmed the treatment's safety in healthy human volunteers who were not pregnant before moving to test it with pregnant patients.
They recruited 16 patients with preterm preeclampsia, which is diagnosed before 34 weeks of pregnancy; the participants were a median of 30 weeks pregnant upon hospital admission. Seven participants received a single session of the therapy, which confirmed that the strategy could safely reduce sFlt-1 in people. The remaining nine patients then underwent between one and three treatment sessions. In this latter group, each treatment session brought sFlt-1 levels down by nearly 17%, although these levels rebounded in some patients.
The main purpose of this small study was to confirm the treatment's safety, and the participants weren't compared directly with untreated patients. Still, the participants who were treated carried their pregnancies for a median of 10 days after their hospital admission, while a group of untreated patients who weren't enrolled in the study carried for only four days post-admission.
These results suggest that the treatment can prolong pregnancy in preterm preeclampsia, though a proper clinical trial is needed to know for sure.
More to learn
While Thadhani's team is targeting sFlt-1 levels, some scientists disagree about how the protein contributes to preeclampsia.
"The camps are still split on whether it has a role in preeclampsia development or it's a consequence of something else happening in the body," said Dr. Lana McClements, leader of the Cardio-Obstetrics Research Group at the University of Technology Sydney, who was not involved with the study.
Not all cases of preeclampsia are alike, McClements said, and multiple factors other than sFlt-1 may drive the disease. That said, Thadhani and his collaborators focused on testing the treatment in participants with early-onset preeclampsia, which is associated with high sFlt-1 levels. In that way, they may have identified a suitable patient population for their approach, she said.
McClements called the pilot study's results "promising," but she noted that sFlt-1 levels dropped by less than 17% in the human patients following each treatment, compared with around 50% in the baboon experiment. And in people, the levels sometimes rebounded and then plateaued.
Based on this pilot study, Thadhani plans to test the treatment with a gold-standard clinical trial and at earlier time points in pregnancy.
"The women that we treated in this paper were quite sick," he said. "They were literally on the verge of delivery. The next step here is to start earlier so that we can deliver more treatments and keep the disease more quiet."
This article is for informational purposes only and is not meant to offer medical advice.
Thadhani, R., Hiemstra, T. F., Vatish, M., Stepan, H., Cerdeira, A. S., Brockelsby, J., James, T., Lia, M., Cornelis, A., Krause, E., Spath, M. R., Grüttner, B., Todorova, P., Hagmann, H., Yeung, K. R., Xu, B., Heffernan, S., Pears, S., Waugh, R., . . . Karumanchi, S. A. (2026). Targeted removal of soluble Fms-like tyrosine kinase 1 in very preterm preeclampsia: a pilot trial. Nature Medicine. https://doi.org/10.1038/s41591-026-04333-6

Lauren Schneider is a health and science journalist currently pursuing a master's degree from the Science, Health and Environmental Reporting Program at New York University. She earned a bachelor's degree in neuroscience at The University of Texas at Austin prior to becoming a writer. In her spare time, you can find Lauren watching movies, swimming, editing Wikipedia, or spending time with Lucy, her impossibly cute black cat.
You must confirm your public display name before commenting
Please logout and then login again, you will then be prompted to enter your display name.

 Live Science Plus
Live Science Plus









