Gallery: The Art in Biomedical Research

2012 Bio-Art Competition Winners
The first-ever Bio-Art competition honored 10 images that are visually arresting and that illustrate a cutting-edge concept in biomedical research.
Scaffold for Cartilage Regeneration

Due to a lack of blood vessels and other characteristics, cartilage heals very slowly. One way to accelerate natural cartilage repair and growth is to use tissue engineering, or the artificially-stimulated production of functional replacement tissue. The image shows a three-dimensionally woven biomaterial scaffold. The scaffold consists of multiple layers of resorbable fiber bundles that have been woven into a porous structure. The scaffold is then seeded with cells that grow to become new tissue as the fibers are resorbed. The fibers provide stiffness and strength in a manner that mimics native collagenous tissues such as cartilage. This work to use tissue engineering to generate replacement cartilage is supported by NIH funding from the National Institutes of Arthritis and Musculoskeletal and Skin Diseases.
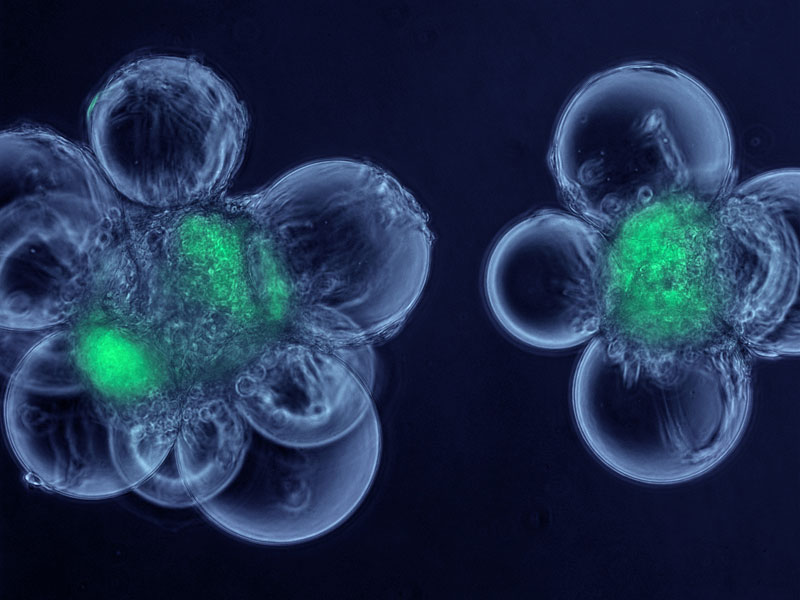
Muscle Stem Cell Factories

This micrograph shows cells called myoblasts attached to spherical microcarriers, which allow the growth of adult stem cells that have been isolated from skeletal muscle. The stem cells are shown in green. By combining these cells in a bioreactor, the muscle stem cells can be greatly increased in number and then separated from the myoblast “feeder” cells. The image was produced in the course of studies aimed at creating artificial “stem cell factories” and was supported by NIH funding from the National Heart, Lung and Blood Institute.
Electricity from Fish
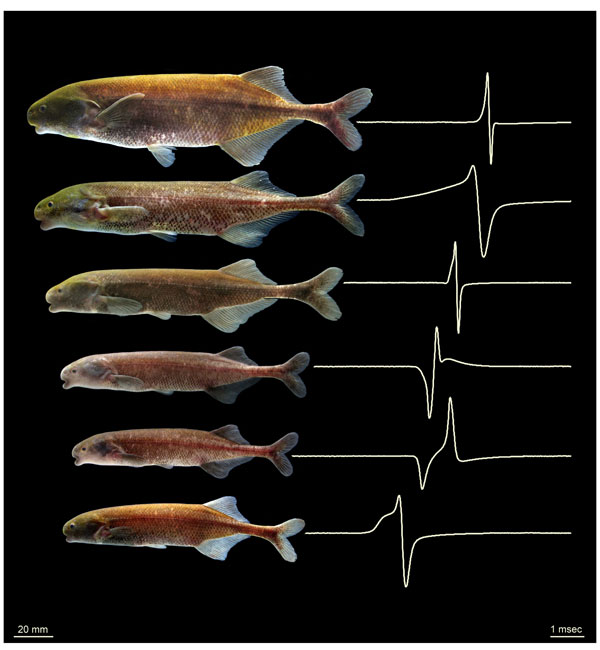
Closely related electric fish species from the Okano River of Gabon, collected in the vicinity of the abandoned Fang village, “Na.” Each species is shown along with a recording of its electric organ discharge, which these fish use to communicate with one another and electro-locate prey, much like bats use echolocation. Electric fish recognize other members of their own species using the species-specific waveforms of these heartbeat-like discharges. NIH funding from the National Institute of General Medical Sciences allows Arnegard and colleagues to investigate how this electrical variability is caused by some of the same genetic mutations that, in humans, lead to congenital heart defects and childhood epilepsy.
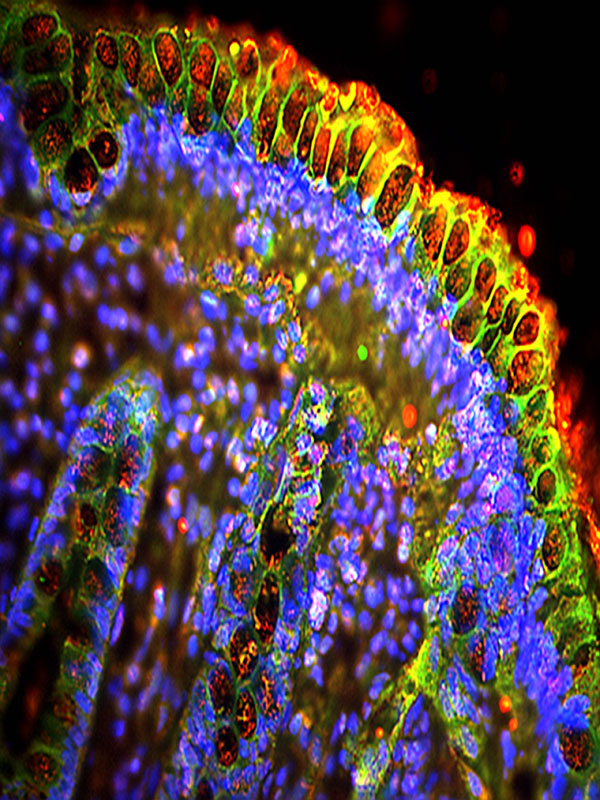
Seeing Capillary Cells
Visualizing the smallest biological structures has traditionally required the use of highly specialized electron microscopy equipment. Using a combination of genetic-based labeling and a variety of fluorescent proteins, researchers have developed a way to push the limits of the light microscope to view the smallest of biological structures. The image shows foot processes of genetically labeled cells (red) covering walls of capillaries (green) in a kidney of a mouse. This work is supported by the NIH National Institute of Diabetes and Digestive and Kidney Diseases.
Coping with Good Gut Bacteria
The control of inflammatory responses in the intestinal lining is critical to the prevention of unwanted immune reactions to the “good bacteria” in the gut. Expressed on the surface of the cells of the intestine, the single IgG IL-1-related receptor (Sigirr) is a key regulator of this process. Tissue from a colon biopsy stained for Sigirr and other cellular components is shown. Therapeutic targeting of Sigirr could be useful in the treatment of inflammatory bowel disease or other inflammatory disorders of the gastrointestinal tract.
Understanding Glaucoma
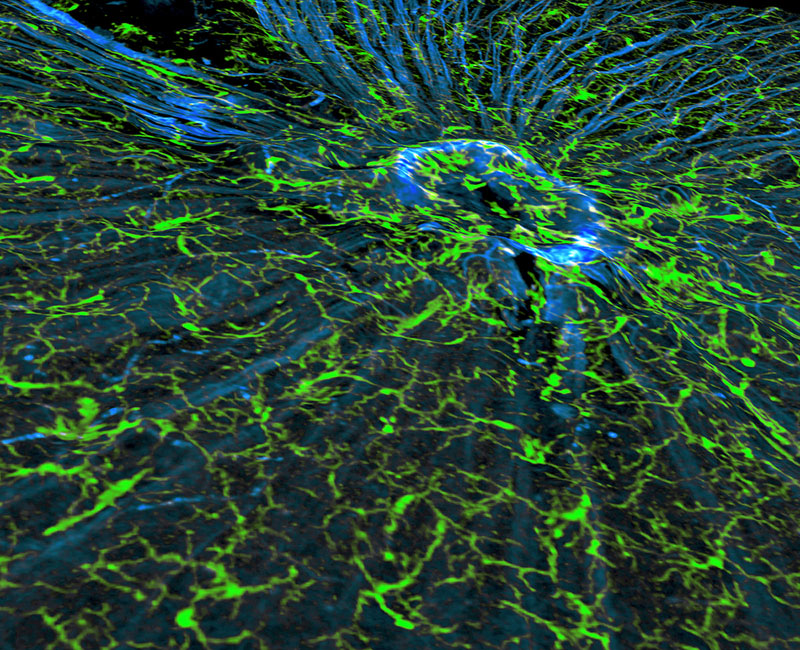
The image features nerve fibers (blue) and their attendant microglia (green), the cells responsible for immune defense in the central nervous system, as the fibers converge to form the optic nerve in a mouse retina. In glaucoma, the retina and optic nerve experience neurodegeneration that causes blindness. Researchers have discovered that mouse retinal microglia undergo changes preceding irreversible neuronal decline and death. Similar changes may take place in humans. By detecting and tracking the activity of microglia, researchers aim to identify and target early pathways underlying the onset of glaucoma. This research to further the development of more effective treatments to diagnose and slow glaucoma is supported by NIH funding from the National Eye Institute.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Study of Limb Development
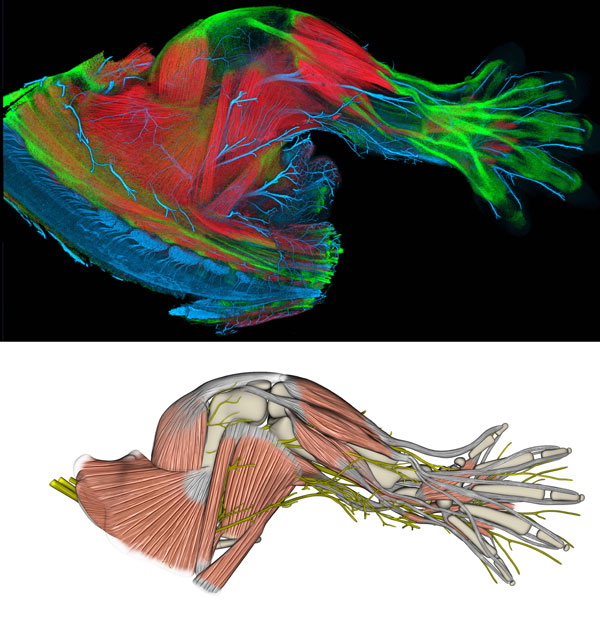
(Top) This is a limb from a transgenic, embryonic mouse, showing well established musculoskeletal and nervous systems. The limb is stained with a variety of techniques to differentiate muscle, tendon, bone, and nerve, and rendered into a three-dimensional image using FluoRender, a publicly available rendering program developed at the University of Utah. (Bottom) This is a model, derived from the upper panel image, showing muscles, tendons, bones, and nerves. This model is part of a collaborative effort between geneticists and computer scientists to develop a 3-dimensional interactive atlas of limb development. NIH funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development supports this effort.
Brain, Heart, and Lung Communication
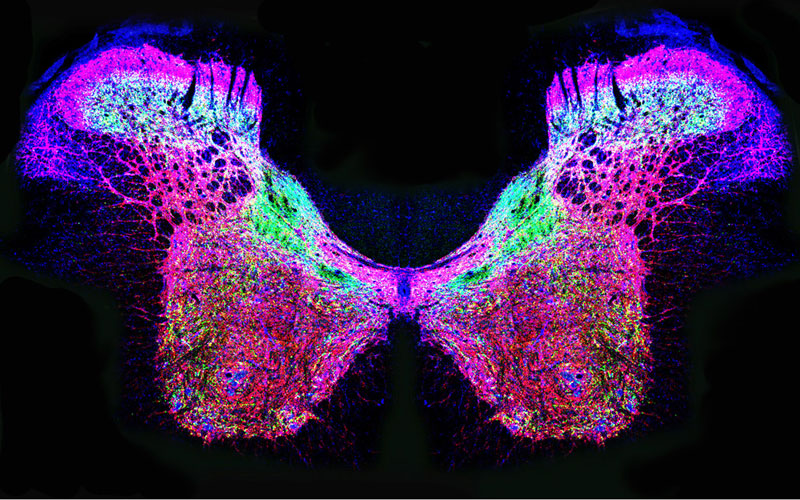
Both glutamate and nitric oxide play an important role in transmitting cardiovascular and respiratory signals between the brain, heart, and lung. This butterfly shaped figure is an image of a rat spinal cord showing the distribution of three types of glutamate and nitric oxide synthesizing enzymes. Understanding the action and interaction of glutamate and nitric oxide in the nervous system could lead to better treatments for cardiovascular diseases such as hypertension and heart failure. This work is supported by NIH funding from the National Heart, Lung and Blood Institute.
Production of New Neurons
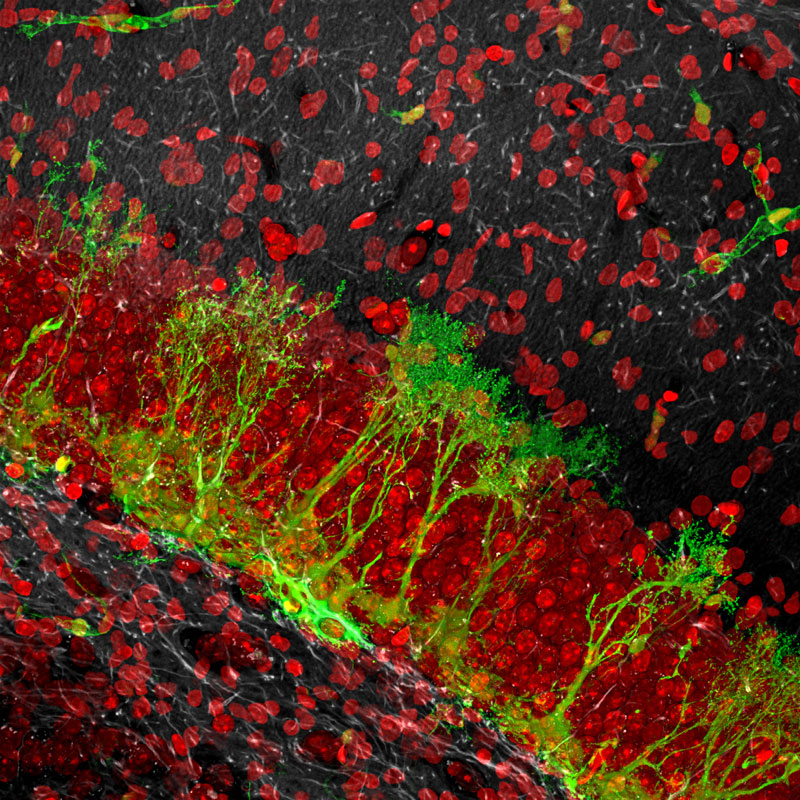
New neurons are produced from neural stem cells in several areas of the adult brain. One such area is in the hippocampus, a brain structure crucial for cognitive function. The number of neural stem cells in the hippocampus decreases over time, possibly contributing to the cognitive impairment associated with aging. When activated by extrinsic stimuli, stem cells divide and generate progenitor cells, which eventually mature into neurons and migrate into the layers above, whereas stem cells themselves undergo additional rounds of rapid divisions and convert into astrocytes, thus leaving the stem cell pool. The image depicts stem cells (green) and neuronal nuclei (red). This research to understand how the brain produces new neurons is supported by NIH through the National Institute of Mental Health and the National Institute of Aging.
Skin Cells Produce Nerve Cells & Psychiatric Insights
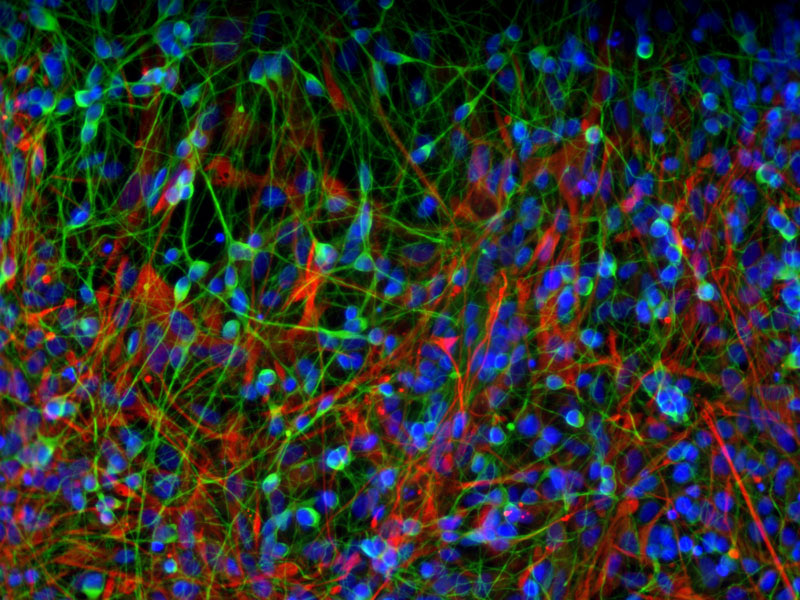
The study of induced pluripotent stem (iPS) cell-derived neurons is a promising new approach to understanding the molecular and cellular underpinnings of schizophrenia, bipolar disorder, and other psychiatric diseases. Researchers are investigating the disease biology of psychiatric disorders by reprogramming patient skin cells to iPS cells and then generating live neuronal cells with the patients' genetic makeup. This image shows a culture of differentiated neuronal cells (green) and neural progenitor cells (red), with the nuclei of the cells in blue. This research is supported by NIH through the National Institute of Mental Health.


 Live Science Plus
Live Science Plus





