Why Sugars May Be the Body's Superstars
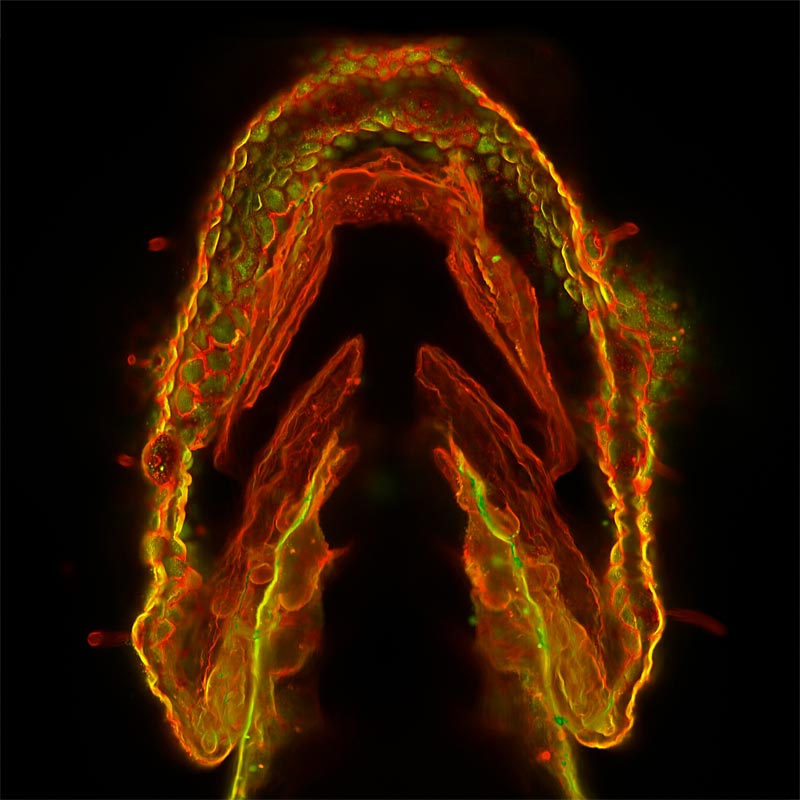
Sugars are well known as an energy source for our bodies. But did you know that sugar chains made within the body, called glycans, play important roles in just about every aspect of how our cells work?
Glycans make sure proteins in the body are folded into the proper shape so they function correctly. They also act as ZIP codes that direct newly made proteins to the right cellular locations. Some glycans on cell surfaces act as anchors that viruses latch onto, while others divert white blood cells to infection sites. Many glycans help relay essential signals needed for the immune system and brain to function.
Levels of Complexity
Article continues belowGlycansconsist of chains of sugar molecules (polymers, or polysaccharides) that are chemically linked. These chains can be linear, but they are often more intricate and include branches. The chains can vary from a few to dozens of sugar molecules that vary in their combinations.
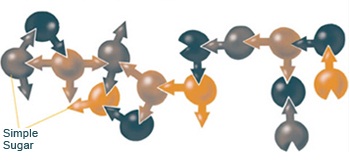
Chemists have struggled to examine these chains in order to get a sense of precisely how glycans work in the cell and how they might be harnessed to treat or prevent disease. For example, it might be possible to block infections by manipulating the glycans on cell surfaces that bacteria and viruses attach to.
Glycans, right and wrong
Glycans don't always act alone. They are most often found attached to proteins or lipids (fats). When that is the case, glycans might just be the largest and most structurally diverse class of molecules in nature.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Glycans are found attached to about half the proteins in the human body. These glycoproteins have important functions, like helping fertilized eggs latch onto the uterine wall so that the egg can implant and develop.
Glycans attached to fats also have many roles, including determining a person’s blood type.
Because of glycans' diverse and critical roles in our bodies, when glycan synthesis goes wrong, the results can be devastating. Most of the time, embryos won't survive if they have genetic errors in the cellular machinery needed to make glycans. In the rare cases that they do survive, the babies are born with virtually every part of their bodies affected, causing developmental, neurological and digestive problems.
With glycans implicated in cancer cell spread, malaria, muscular dystrophy and other health problems—as well as treatments and drug safety—a growing number of researchers supported by the National Institutes of Health are focusing on sugars to treat disease.
One such researcher—glycobiologist Hudson Freeze at the Sanford-Burnham Medical Research Institute in La Jolla, California—has been investigating whether a sugar called mannose could treat disorders caused by a defective gene that is supposed to transport sugars to sites where glycans are synthesized in cells. His therapy helps the malfunctioning transporter bring sugars into the endoplasmic reticulum so they can be used to make glycoproteins.
Sleuthing with sugars
In 2008, a contaminated batch of heparin—a drug composed of long chains of a repeating sugar that functions as an anticoagulant—was sickening and killing people. The search for the harmful contaminant was particularly tricky because it had a structure very similar to heparin's. But Massachusetts Institute of Technology bioengineer Ram Sasisekharan and colleagues found the contaminant.
Sasisekharan's team developed a new test to screen heparin for contaminants, providing the U.S. Food and Drug Administration with an important tool to keep the drug safe.
Another of Sasisekharan's glycan breakthroughs came when he figured out the precise order of heparin's sugar components. This knowledge has opened the door for other researchers to study similar glycans in intricate detail. Sasisekharan has also investigated heparin’s function in the hope of developing safer versions of the drug for patients who experience side effects from the blood thinner.
Learnmore:
This Inside Life Science article was provided to LiveScience in cooperation with the National Institute of General Medical Sciences, part of the National Institutes of Health.
