Best fitness trackers 2026: From smart rings to multisport GPS watches
We have tried and tested dozens of fitness trackers, smartwatches and smart rings — these are the top 12 wearables that should land in your cart this spring.

- Best overall
- Best for tracking workouts
- Best cheap fitness tracker
- Best cheap smartwatch
- Best running watch
- Best Garmin alternative
- Best Google Watch alternative
- Best for sleep tracking
- Best screen-free
- Best hybrid smartwatch
- Best for beginners
- Comparison
- Our experts
- Fitness trackers FAQs
- More info
- How to choose
- How we test
- Latest updates
The variety of fitness trackers means there is something for everyone. For sleep tracking, for example, you can't beat an unobtrusive wearable smart ring like the Oura Ring (Gen 4), while for athlete-level workouts, there are plenty of excellent Garmin watches to choose from. Whether you are a beginner looking for one of the best budget fitness trackers or a keen athlete in search of a top-quality running watch, rest assured, there is a fitness tracker that ticks all your boxes.
"Fitness trackers have many useful features and have helped me and my clients make better fitness decisions," Nathan Kennedy, a UK-based personal trainer, told Live Science. If you would like to know more about what fitness trackers do and how they work, scroll down to the FAQs section.
At Live Science, we have spent countless hours testing wearable tracking and fitness devices, assessing their accuracy, features and durability. We have checked whether they work with third-party devices and, more importantly, determined exactly who would benefit most from them. You can read more about our testing protocol in the How we test section below. Unsure what kind of fitness tracker to look for in the spring sales? Check the How to choose section for more useful advice.
The best fitness trackers we recommend in 2026: Fully tested by our expert reviewers
The best fitness tracker overall





Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ You are on a budget: It costs a reasonable $199 and is frequently on sale.
✅ You want long battery life: It can last up to two weeks on a single charge.
✅ You want accurate tracking features: Its heart rate and body composition sensors were top-class in our tests.
Don't buy it if
❌ You are an outdoor adventurer: It may not survive some of the more extreme conditions.
❌ You do not want to pay anything extra: Some minor features are behind a paywall.
The bottom line
🔎 Amazfit Balance: With superb battery life and great tracking features, this lightweight and affordable smartwatch offers real value for money. The only drawback is that some advanced features live behind a paywall.
We have tried and tested plenty of fitness trackers over the last year, but not a single wearable impressed us nearly as much as the Amazfit Balance smartwatch. It is a rare bird: skilfully designed, performing well, and brimming with advanced health-tracking features, but priced at just under $200 (and often on sale, for that matter). You would be hard-pressed to find better value for money.
This sleek wearable not only looks good, feels good and works well, but it is also suitable for a wide range of people, from fitness beginners to budding athletes. The Amazfit Balance, as its name clearly indicates, is more about sleep, post-exercise recovery and general wellness rather than athlete-level exercise performance, and its stats are exceptionally easy to understand and track over time. That said, its fitness-tracking performance would still satisfy most serious exercisers, as we discovered during our full Amazfit Balance review.
This smartwatch accurately measured our heart rate, breathing rate, heart rate variability, and even body composition (!), and it never slid from our wrists or got in the way during intense workouts. We also liked its easy-to-use, colorful display and comfortable, buttery-soft strap. Not to mention, it was one of the lightest and most compact smartwatches we have ever tested.
As we mentioned already, the Amazfit Balance is a particularly good option for those who want to better manage their sleep hygiene and stress levels. This sleek fitness tracker is packed to the brim with wellness apps and health tracking features designed to help you boost your post-exercise recovery and live a more mindful lifestyle. It also comes with a whopping 14-day battery life, dual-band GPS navigation, compass, altimeter, and many other features typically reserved for more premium models.
However, the display in the Amazfit Balance does not feature Gorilla Glass or any other sturdy materials, so it may not be the best choice for those who engage in extreme sports or heavy manual tasks. It may not be the best contender for a reliable outdoor watch. Also, some of its additional features, such as the AI-powered fitness coach chatbot, are locked behind a paywall. These paid-for frills are not essential, though.
For less than $199, the Amazfit Balance offers great value for money, making it our favorite fitness tracker this year.
- Read our full Amazfit Balance review
Best smartwatch for tracking workouts





Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ You are a data-driven fitness enthusiast: It gives you a detailed overview of your exercise performance.
✅ You need accurate navigation tools: This watch boasts reliable GPS tracking and detailed on-watch maps.
✅ You want long battery life: It can last up to 16 days on a single charge.
Don't buy it if
❌ You are on a budget: Priced at around $1,000, it is one of the most expensive options in this guide. If you want something cheaper but of similar quality, consider the older Fenix 7 model.
The bottom line
🔎 Garmin Fenix 8: If you want the best in show of fitness watches, then the Fenix 8 has it all, from a huge array of tracking metrics to a superior battery life. It's certainly not cheap, but we think it's worth the outlay for such a superior product.
A Garmin Fenix 8 is one the best fitness trackers to get if you want the most comprehensive set of built-in exercise features available. These stand out even more when you bring its impressive hardware into the mix. In our eyes, this rugged GPS watch has dethroned its older sibling, the excellent Garmin Fenix 7 Sapphire Solar. They both deserve this spot, though. The Garmin Fenix 8 is slightly more beefed-up and high-tech than its younger sibling, making it more functional for serious fitness enthusiasts— you can take it scuba diving, for example. However, it is also prohibitively expensive. The Garmin Fenix 7, on the other hand, is cheaper by around $450, but quality-wise, these two watches are not too far apart.
That said, we still think this premium wearable is well worth the steep investment. During our Garmin Fenix 8 review, we were thoroughly impressed with its performance and tracking features. This rugged watch can connect to heart rate straps and foot pods, bike turbo trainers, and even Garmin’s bike lights. We liked that you can download continents’ worth of maps to the watch’s internal memory and plan hike, bike and run routes so you can leave your phone at home. Not to mention, its heart rate and GPS accuracy are great, and the presentation of stats like training load and VO2 Max is world-class.
While a perfect choice for hitting your PBs, it is also useful for more mundane activities. While still a sizeable watch, the Garmin Fenix 8 is not as heavy and bulky as its older sibling, making it comfortable to wear all day round. It works well for casual day-to-day activities too. For example, it lets you download music and podcasts from some of the most popular music services, including Spotify and Deezer.
A Garmin Fenix 8 may be a bit much if you just want to track 5km runs a couple of times a week. It is not small and it is not cheap. The bulk is not ideal for sleep tracking, so consider the Garmin Epix 2 or Garmin Forerunner 965 if you like the sound of Garmin’s high-octane style but would prefer something smaller. Looking for something more elegant? The Polar Ignite 3 Titanium may be a better option for you (scroll down to read more).
- Read our Garmin Fenix 8 review
Second-best workout tracker

Best for tracking workouts: Second-best
The Suunto Race 2 may not have the advanced features or top-notch tracking accuracy of the Garmin Fenix 8, but it makes up for these shortcomings with its elegant design, good durability and long battery life. Not to mention, it is cheaper by at least $400 and feels less chunky and intimidating than its pricier counterpart. A solid mid-range pick for fitness enthusiasts.
Pros
- Stylish and comfortable to wear
- Good GPS accuracy
- Durable design
Cons
- No smartwatch features
- Not that user-friendly
- Not suitable for diving
Best budget fitness tracker





Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ Battery life is important to you: The battery life really is as good as it says, with us not having to charge this watch more than once in three weeks.
✅ You want a simple interface: This watch is super easy to navigate around and isn't overly complicated.
✅ You're a beginner to fitness tracking: Thanks to its uncomplicated nature, this Xiaomi band is ideal for those looking for their first fitness tracker.
Don't buy it if
❌ You want reliable location tracking: This fitness tracker doesn't have its own GPS and relies on your smartphone GPS so signal can be lost.
❌ You prefer a larger screen: Although you can see everything you need to on the smaller, slender screen, some might prefer a larger watch face.
The bottom line
🔎 Xiaomi Smart Band 10: With a wealth of workout modes to choose from, 21-day battery life and a sleek design, this fitness tracker is an absolute must-have for those on a tight budget. And it's not just these features that make it worthwhile, but the fact that it accurately captures workouts, albeit not as thoroughly as more expensive models. If basic fitness tracking is what you're after, you can't go wrong with this budget smart band.
When it comes to best budget fitness trackers, two brands are leading the pack: Fitbit and Xiaomi. Having previously tested and reviewed the Xiaomi Smart Band 9, we managed to get our hands on the Xiaomi Smart Band 10 which has a few extra features not previously seen. We thought the Smart Band 9 offered lots, but the 10 keeps some of the best features from its predecessor and adds a few more, such as real-time underwater heart rate monitoring, running courses and sleep improvement plans.
The screen is also slightly larger at 1.72-inch AMOLED display compared to the 1.62-inch display of the Smart Band 9. The unobtrusive nature of this sleek and slender design means you barely realize it's on your wrist during workouts. And, while not a traditional watch face shape, you can still view all the necessary stats easily by scrolling.
We loved the length of battery life we got out of this smart band, with us only having to charge it once in three weeks and even then, it still had 17% battery left. You can choose from 150+ workout modes and can be rest assured your workout will be accurately tracked. The interface is uncomplicated, making it a great option for those looking to track their workouts with ease and for those who want basic stats rather than pages and pages of information post-workout.
And, even though this is a budget fitness tracker, this doesn't mean a lack of features. With 150+ workout modes, running courses, heart rate monitoring, stress level monitoring, blood oxygen levels, sleep tracking and sleep improvement plans (just to name a few), the Xiaomi Smart Band 10 offers a lot for its price point.
We were particularly impressed with the accuracy of the sleep tracking, with asleep and wake times being bang on. Even on nights when we had more disturbed sleep and had checked what time we had woken up, the watch captured these times superbly. For workouts, we found the step counts and average pace for walks to be about right and the same for the BPM, whether resting or exercising. However, for more precise health tracking, you may need to spend more on mid-range trackers like Amazfit Balance or Garmin Vivosmart 5.
- Read our full Xiaomi Mi Smart Band 10 review
Best budget smartwatch





Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ Exercise is a hobby, not career: This watch lends itself best to those who exercise casually or are beginners, rather than athletes.
✅ Versatility is important to you: With some basic smartwatch features and plenty of workouts to choose from, plus some customization options, this watch would suit those looking for versatility.
Don't buy it if
❌ You’re into adventure sports: This smartwatch has a sturdy build, but won’t suffice for adventure sports.
❌ You want a diving watch: The 5ATM water resistance means this watch won’t cope with diving, freediving, or adventure water sports.
The bottom line
🔎 Amazfit Active Max: Ideal for beginners or casual exercisers, this smartwatch is packed with features and comes at an incredibly affordable price — you’d be silly not to get it.
For those who exercise as a hobby, rather than a career, the Amazfit Active Max has just the right level of durability — while it might not have Gorilla Glass or Sapphire Crystal and be made from aluminum alloy rather than titanium, it withstood our testing at the gym with no marks or wear or tear. Overall, we found the design of the Amazfit Active Max to be better than the Amazfit Active — which we voted the best budget smartwatch, previously. With removable straps and customizable watch faces, you can make this watch your own to some extent. However, it currently only comes in one color and size which might disappoint some. That being said, we found this watch comfortable to wear, and it performed well when it encountered rain or sweat, thanks to its 5ATM water resistance.
All fitness trackers now track the basic health stats, such as heart rate variability, sleep, stress, skin temperature and steps. However, this budget smartwatch also has a go at tracking more advanced features such as training load and sleep apnea risk. Not bad for a watch that costs less than $170.
This watch doesn’t fall short on workout tracking options, either, and even includes some more niche examples, such as snow shoveling. While automatic workout detection is available, it can be hard to rely on it as it sometimes guesses wrong. However, for outdoor exercisers such as walkers or runners, there are plenty of features to capture your workouts successfully, such as a compass, altimeter, offline maps and built-in GPS. Side note warning — the GPS is single-band, but this is to be expected at this price point. We found its overall tracking ability to be sufficient — not 100% perfect all the time, but not bad for where it sits in the market. We were pleasantly surprised by its ability to accurately capture heart rate above 150BPM where most smartwatches begin to struggle with accuracy.
And how do you view these health stats? That’s through the accompanying app, Zepp. There is an AI-driven coach within the app that can offer personalized training tips and tips to improve your lifestyle, which is a nice feature for beginners who may want a bit of guidance. The app itself presents the health stats in a digestible format, so anyone new to the world of fitness tracking will be able to understand the data. However, it wouldn’t necessarily suit those who exercise as a career, such as athletes.
- Read our full Amazfit Active Max smartwatch review
Runner-up

Best cheap smartwatch: Runner-up
Want something bigger than the Amazfit Active? Consider the Redmi Watch 5 instead. This Chinese-made smartwatch boasts a bright and colorful 2.07-inch AMOLED display as well as stacks of features, great tracking accuracy and long battery life. That said, its screen can become less responsive when wet, so it may not be a great option for swimming enthusiasts.
Pros
- Up to 24 days of battery life
- 2.07-inch AMOLED display
- Plenty of tracking features
Cons
- Short charging cable
- Can catch on long-sleeved tops
- Screen less responsive when wet
Best running watch





Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ You want good value for money: All the high-end Garmin features and at a reasonable price.
✅ You want long battery life: This watch can last up to 23 days on a single charge.
✅ You want accurate tracking features: It features an excellent GPS, and you can rely on its health sensors too.
Don't buy it if
❌ You are a diver: It is not waterproof enough.
❌ You need an ultra-sturdy watch: Not as durable as more premium Garmin models.
The bottom line
🔎 Garmin Forerunner 965: With an array of Garmin's top tracking features and excellent accuracy, the Forerunner 965 is a fantastic running watch. It's not such a great buy, however, for water enthusiasts as at 5ATM water resistance it doesn't match Garmin's best.
The Garmin Forerunner 965 is an excellent choice for runners thanks to its user-friendly design, excellent navigation, and a wealth of features designed to boost your exercise performance. While it is still relatively pricey (around $500 - $600, depending on the retailer), it is still more affordable than some of the more premium Garmin models. We gave it a strong 4.5 stars in our Garmin Forerunner 965 review and thoroughly recommend it to anyone passionate about running.
The Forerunner watches have won our seal of approval multiple times, even despite the ever-increasing retail prices. We loved the Garmin Forerunner 955, but had to admit it was slightly too expensive for a watch with a plastic shell. The Forerunner 965 has stepped up its design, though — while the main part of the shell is still made from a plastic “resin”, its bezel is now crafted from rock-hard titanium.
The Forerunner 965 screen boasts the biggest OLED Garmin has put into a watch to date, a 1.4in panel of 454 x 454 pixels. The display is also crisp, bright and colorful, making it easy to track your stats and follow the route on the go.
The Garmin Forerunner 965 is packed to the brim with features that can help you level up your running performance. There is a good range of reliable health and activity stats to give you valuable insights into your cardiovascular health, exercise performance and recovery time. The Forerunner 965 is also equipped with excellent navigation systems and detailed maps, so you'll not veer off the track and get lost when running through unfamiliar places. Mind you, this model currently has the 8.8GB Europe Topo maps pack installed, but you can freely install others if you like.
You also have 32GB on which to store any music you might want to carry around with you, smart notifications and handy workout suggestions to spruce up your exercise routine.
That said, the Garmin Forerunner 965 may feel like an overkill for beginner runners. If you are new to this sport, you may want to invest in something simpler and more affordable — such as the Garmin Forerunner 165, for example. Scroll down for more information!
- Read our full Garmin Forerunner 965 review
Budget-friendly alternative

Best budget running watch
One of the handful of trackers to score a full five stars in our tests, the Garmin Forerunner 165 has everything you may need from a beginner-friendly running watch. It is comfortable to wear, jam-packed with tracking features and accurate at measuring your vitals — and it costs less than $200.
Pros
- Bright, easy-to-read display
- Comfortable to wear
- Reliable GPS
- Accurate HR measurements
Cons
- Battery life can drop fast in certain modes
Best Garmin alternative





Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ You are an outdoor exerciser: With comprehensive navigation tools, the Suunto Vertical 2 is ideal for those who exercise outdoors.
✅ You want an adventure watch that’ll keep up with you: With 20 days of battery life in smartwatch mode, or 65 hours in Performance mode, the Suunto Vertical 2 offers good accuracy, durability and features suited to off-grid adventures for those who want to explore.
Don't buy if
❌ You want smart features: This fitness tracker doesn’t have contactless payments or music storage.
❌ You spend more time indoors exercising: If that’s the case, a standard fitness tracker would suit your needs.
The bottom line
🔎 Suunto Vertical 2: This fitness tracker offers outdoor-oriented features which would suit outdoor enthusiasts and exercisers. In strong competition with the Garmin Fenix 8, it offers good accuracy and a stylish yet rugged design, but unfortunately lacks the smart features found on Garmin watches. However, with it coming in at $300 less than the Fenix 8, this is a wonderful alternative for those with a smaller budget.
There is a lot to like about the Suunto Vertical 2, and it’s hard not to compare it to the Garmin Fenix 8. It offers a rugged yet sleek design which is not too bulky on the wrist, especially for an adventure watch. However, it is reasonably heavy, so is definitely more suitable for outdoor use rather than as an everyday watch. Sleep tracking is available on this wearable, but the pure size and weight of this watch may not be comfortable for some, for them to wear it to bed. But, saying that, it is highly durable, which is what you want from an adventure watch.
In terms of the screen, visibility is generally good but it does have auto-dimming in order to preserve battery life, and so it can feel dark at times. However, we never experienced any lag due to temperature changes during our testing. Plus, the touchscreen is responsive but not overly sensitive, so accidental swipes don’t happen.
For outdoor use, the Suunto Vertical 2 has comprehensive navigation tools such as a built-in LED flashlight and advanced offline maps. Plus, the dual-band GPS is reliable, with further features such as breadcrumb tracking, route planning and ‘find back’ (where the watch can help you retrace your steps if you’ve gone off-trail, to guide you back to your starting point), lending this watch particularly well to off-grid explorations.
Even if you’re looking for a more standard fitness tracker rather than an adventure watch or a Garmin alternative, the Suunto Vertical 2 has 115 sports modes as well as continuous heart-rate and blood oxygen monitoring. And don’t forget — sleep tracking.
The Suunto Vertical 2 boasts various battery life lengths depending on the mode you have it in, all of which seem accurate and are quite impressive. The shortest battery life is 20 days when in smartwatch mode. For the highest accuracy in terms of navigation, Performance mode is the go-to, lasting around 65 hours.
Generally, the Suunto Vertical 2 performed well, particularly in terms of heart rate monitoring, navigation and sleep tracking. Where it didn’t shine so much was in terms of calories burned and step count, although these are minor issues in our opinion.
- Read our full Suunto Vertical 2 review
Second-best

Second-best Garmin alternative
Looking for something more suitable for runners and hikers? Meet the Coros Pace Pro. This GPS sports watch offers reliable navigation tools, comprehensive fitness tracking features and impressive battery life, easily making it one of the best Garmin Forerunner alternatives on the market. The downside? Relatively fragile build.
Pros
- Long battery life
- Lightweight design
- Robust workout tracking features
Cons
- Relatively fragile build
- No smartwatch features
- No solar charging
Best Google Pixel Watch alternative




Xiaomi Watch 5
Our expert review:
Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ You need superior battery life: It lasts up to 6 days on a single charge.
✅ You want to save money: It is around $100 cheaper than the Google Pixel Watch 4.
Don't buy if
❌ You want Pixel's health insights: They are not quite as detailed and nuanced.
❌ You need something discreet: It is a relatively big watch.
The bottom line
🔎 Xiaomi Watch 5: Elegant, durable and bursting with smart features, the Xiaomi Watch 5 is a great alternative to the pricier Google Pixel Watch 4.
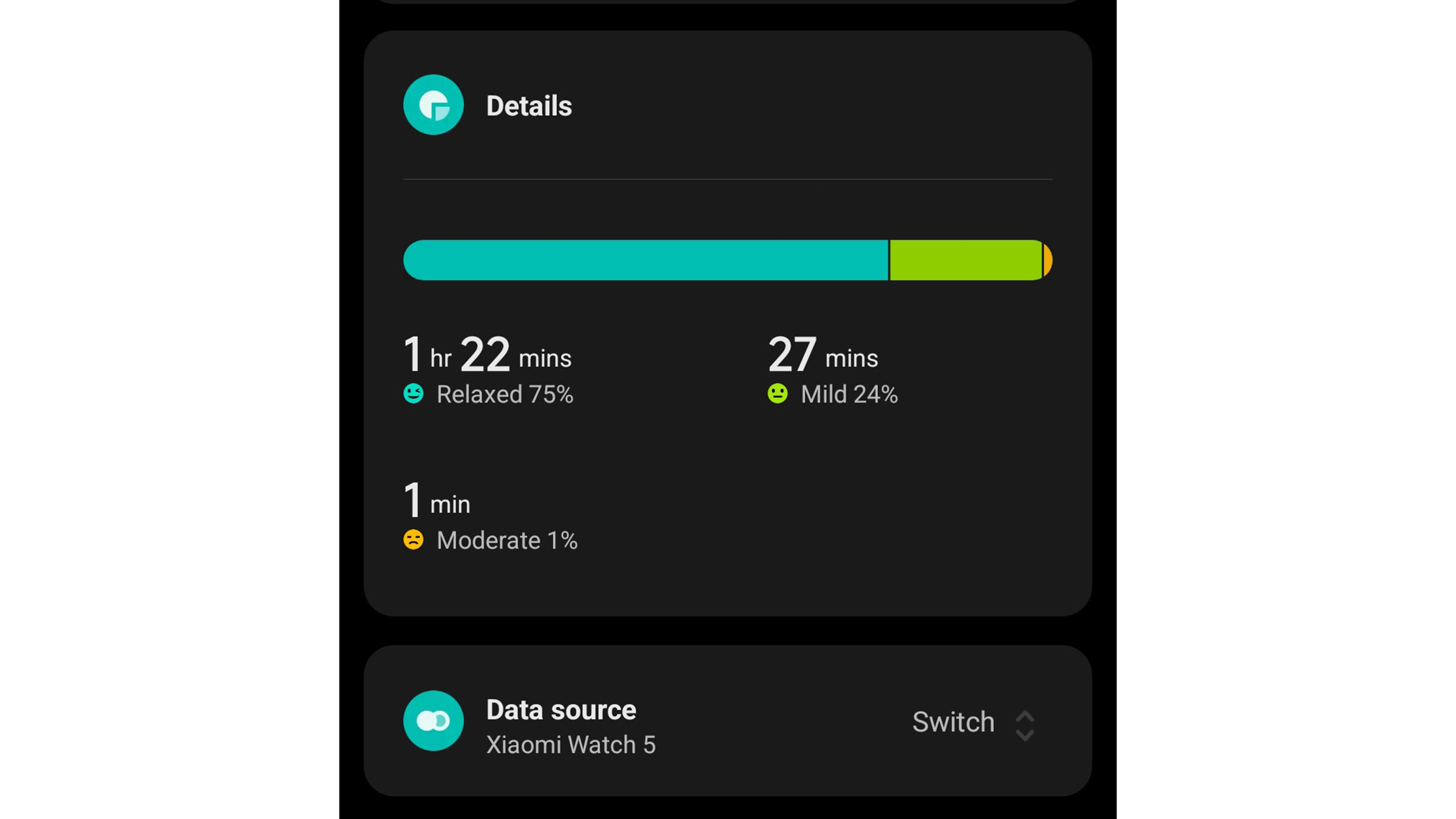
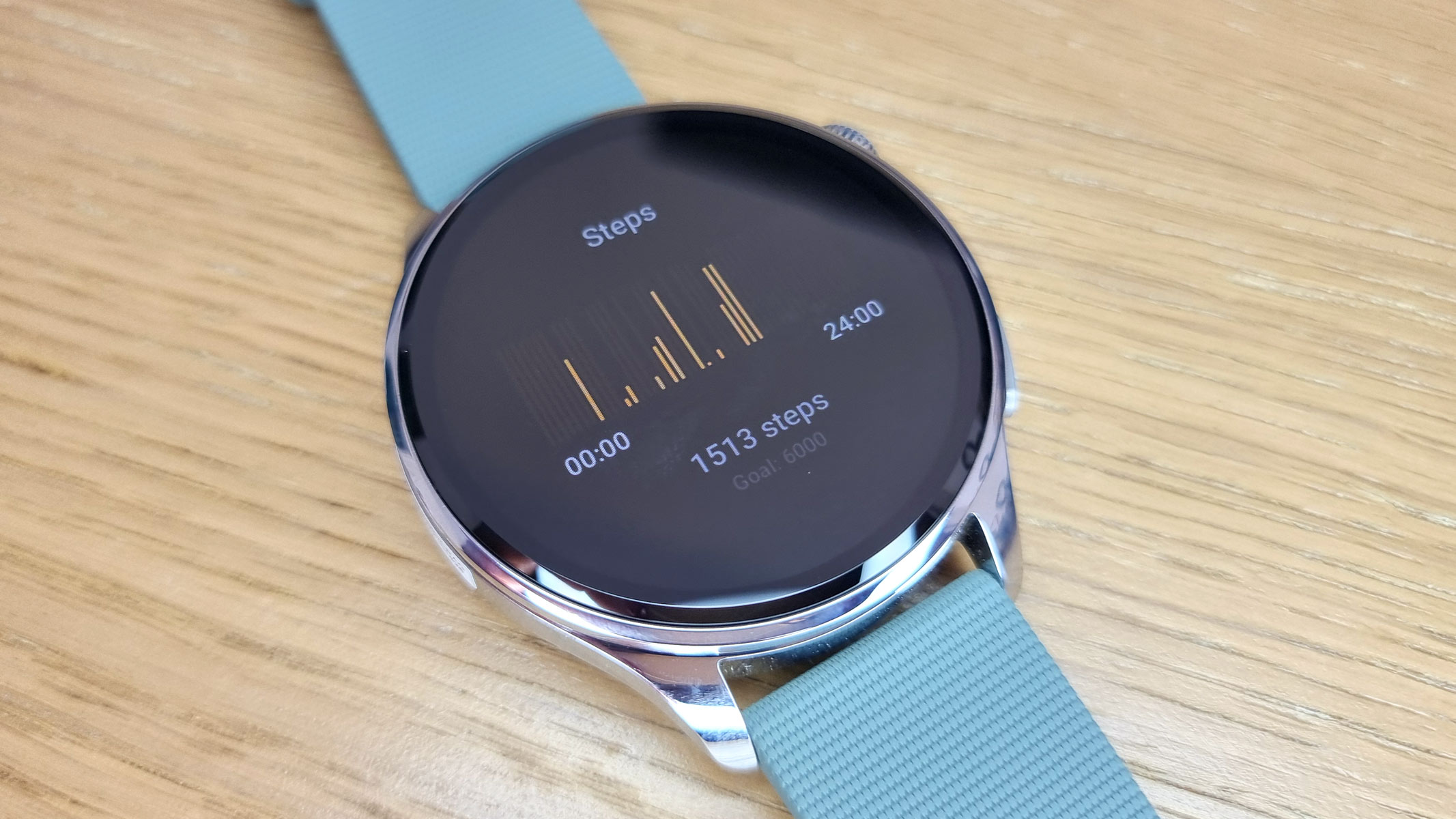
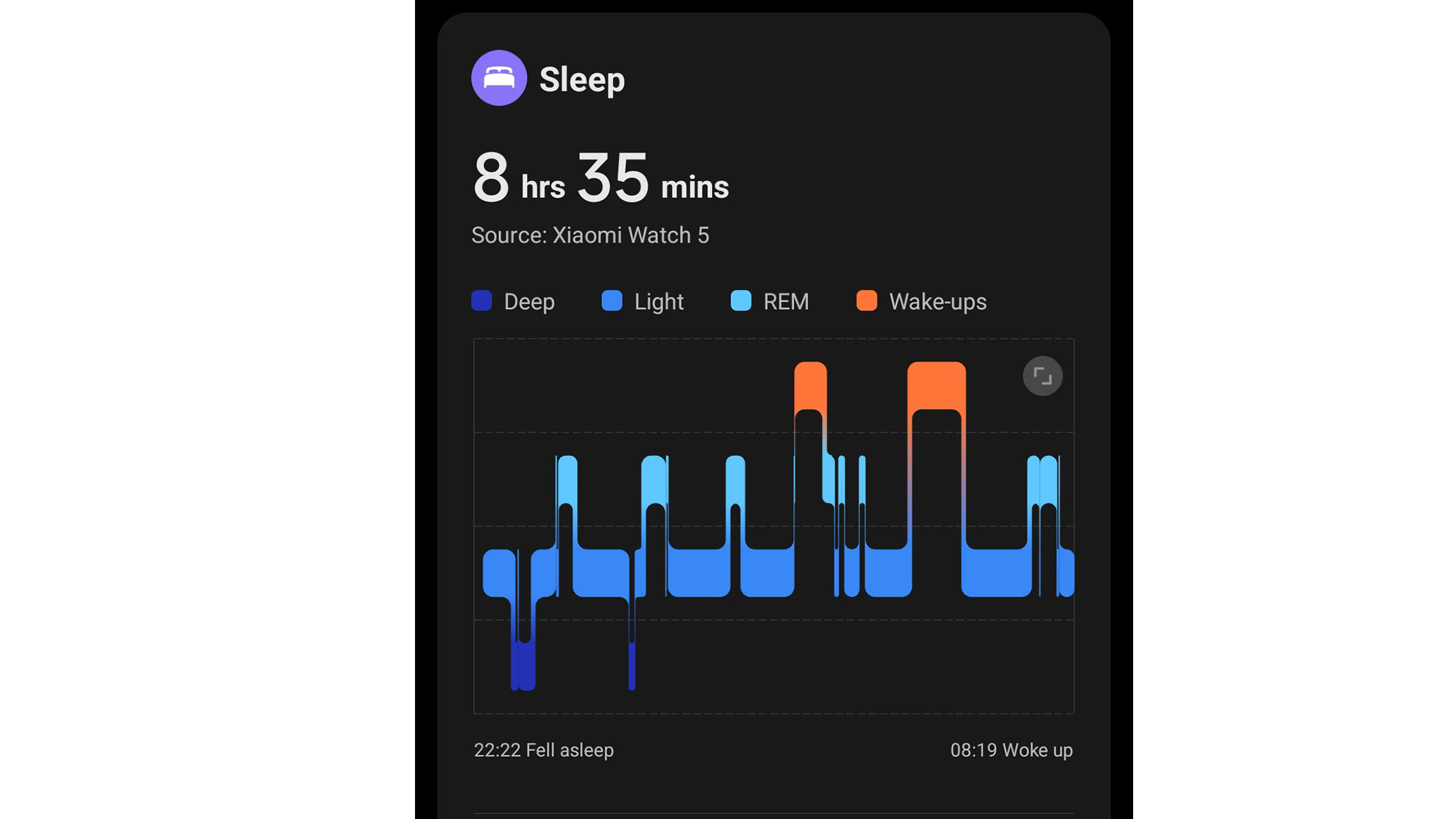
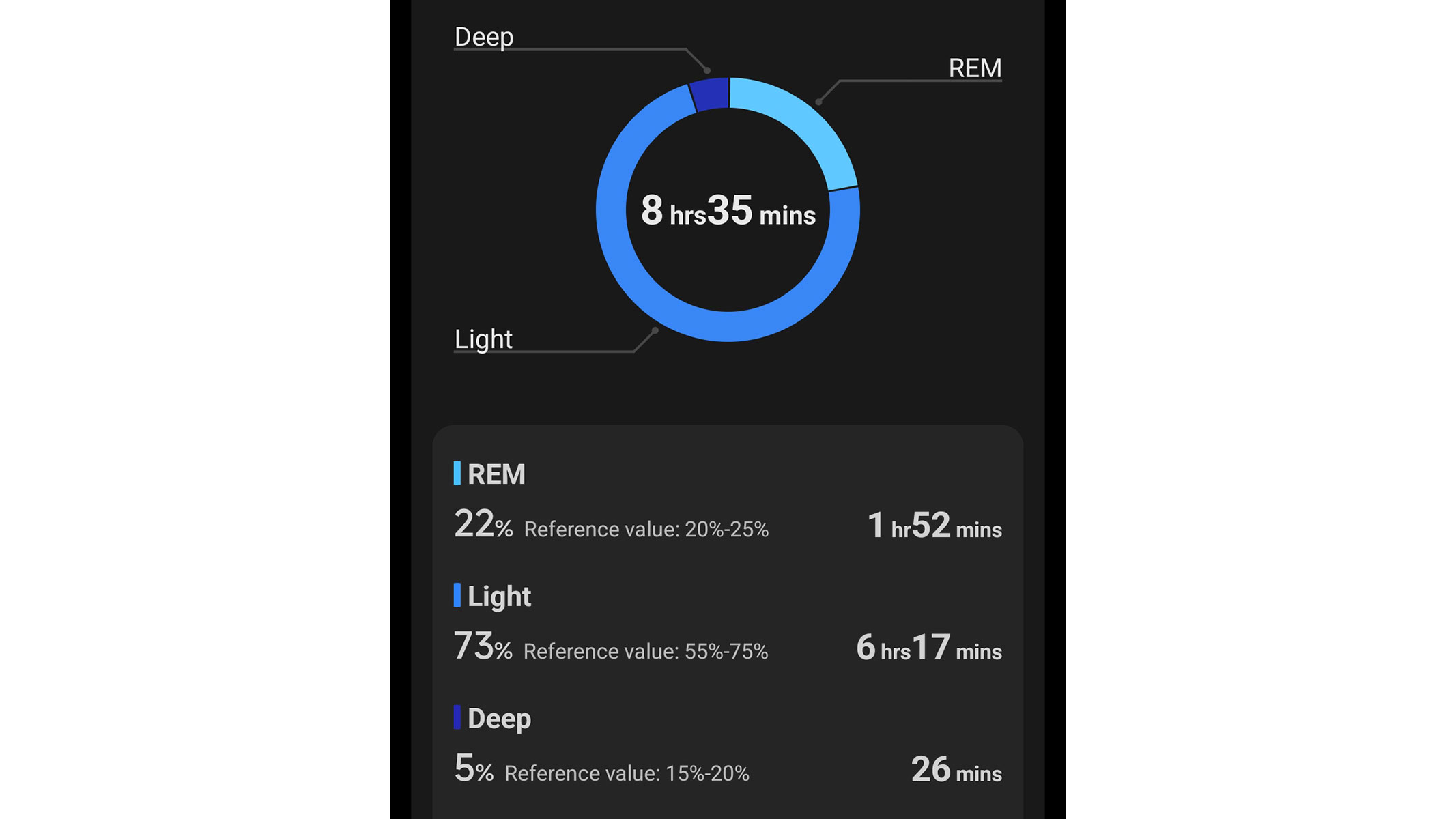
Spooked by the price of the Google Pixel Watch 4? Do not worry, we have found an excellent alternative. The Xiaomi Watch 5 is just as well-built, functional and feature-packed, but costs approximately $100 less. It even has better battery life — this Android smartwatch lasts up to 6 days in smartwatch mode and up to a whopping 18 days in power-saving mode, whereas the Google Pixel Watch 4 tends to run out of battery within two or three days of charging.
The Xiaomi Watch 5 looks great, too. It is sleek, stylish and highly customizable, and its exceptionally large, vibrant AMOLED display is very easy to read. This smartwatch is also exceptionally durable — it is crafted with sturdy stainless steel and comes with a scratch-resistant sapphire crystal glass. We must have dropped or bumped our Xiaomi Watch 5 at least a dozen times, but it has not sustained any visible damage.
Most importantly, the Xiaomi Watch 5 dazzles with extensive functionality and advanced smart features. Just like the Google Pixel Watch 4, it runs Google's Wear OS 6, giving you access to the Google Play Store, Google Maps, Wallet, and many other apps. It also comes with a speaker, microphone and multiple phone integrations. The Xiaomi Watch 5 even offers gesture controls, enabling hands-free navigation and enhancing accessibility. It is an excellent all-rounder.
That said, this smartwatch is not for everyone. The Xiaomi Watch 5 is a relatively large wearable, so those with smaller wrists may find it slightly uncomfortable to wear all day. Clunky navigation is another downside — switching between features and widgets feels quite laborious and takes some getting used to. Health insights also felt a bit basic when compared to the Google Pixel Watch 4. Lastly, the app connectivity feels a bit patchy.
Best for sleep tracking



Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ You want in-depth sleep tracking: It is impressively accurate and insightful.
✅ You need something discreet: No chunky watch case or heavy machinery.
✅ You want long battery life: It lasts up to 8 days on a single charge.
Don't buy it if
❌ You have sensitive fingers: It is bulkier and heavier than a regular ring.
❌ You need highly accurate workout tracking: Some measurements may be imprecise.
❌ You are on a budget: It costs over $350.
The bottom line
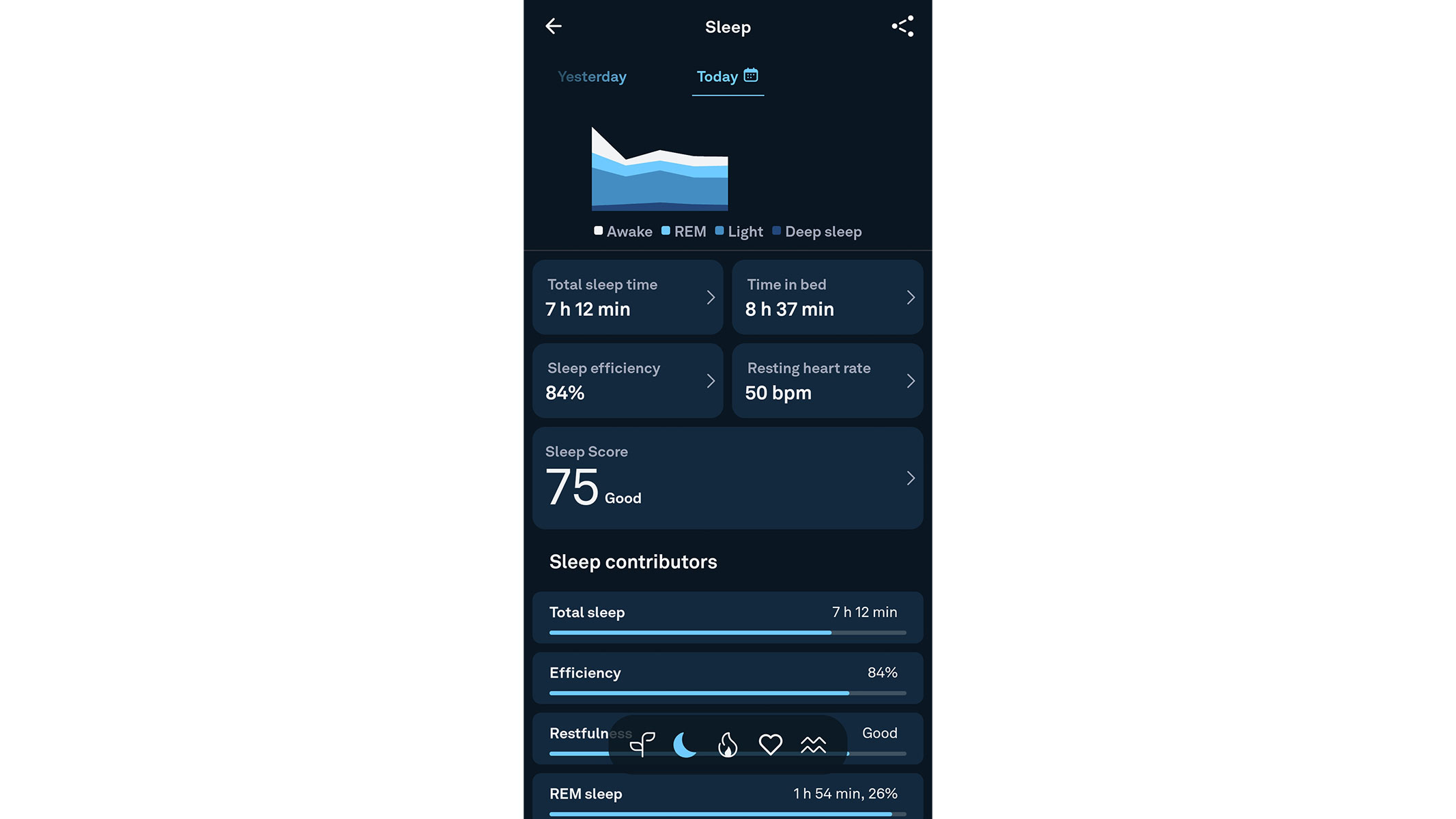
🔎 Oura Ring Gen 3: For those who struggle with wearing a smartwatch all night for accurate sleep tracking, the Oura can't be beaten. It's innovative and 'forget you are wearing it' design, plus impressive battery life, mean you'll get great sleep data. It's less good at other fitness tracking, but there's only so much a ring can do.
The small and discreet smart rings are perfect for tracking sleep, but which one should you pick? While we are big fans of the Ultrahuman Ring AIR and can thoroughly recommend it to anyone who wants to improve their sleep hygiene, it is the Oura Ring Gen 4 that is winning the competition this year. The reason is quite simple. This sleek wearable not only beats other smart rings we tested, but it is also considerably better than the previous model, the Oura ring Generation 3.
We were impressed with the performance of the Oura ring during our four months of testing, especially when it came to sleep tracking and post-exercise recovery assessments. During our Oura Ring Gen 4 review, we found that whenever we woke up feeling groggy and poorly rested, this sleek wearable was able to show us exactly when our rest had been disturbed and how it had affected our REM sleep.
The ring is able to take this data and make suggestions, too. For example, our tester was advised to shift their bedtime to earlier in the day and found that doing so had a positive impact on their sleep duration and consistency.
As the ring is so small, it does not have a screen. Instead, you will have to rely on a companion app to look at most of your health data. We did not find this to be a massive issue, although it does mean there is no easy and quick way to check things like your heart rate when you are in the middle of a workout.
There is an impressive amount of data gathered by the ring. It measures your heart rate at various intervals throughout the day, along with your heart rate variability at night, to help check for potential health problems. And it has a helpful ‘readiness’ feature that will let you know if you should prioritise exercise or rest.
We did find that it is not 100% accurate at tracking all health metrics. For example, we found it massively overestimated calorie burn during our testing period. And its ‘auto-detect’ feature often attributed the wrong exercise to our workout, for example recording a HIIT session as a weights session.
Despite this, we really like it, thanks to its unusual design and brilliant, unmatched sleep-tracking ability.
- Read our full Oura ring Generation 4 review
Runner-up

Best smart ring: Runner up
The stylish Ultrahuman Ring AIR is jam-packed with tracking features, ultra-light and comfortable to wear, and at $349, it is matching the price of the Oura Ring Gen 4. However, its app connectivity could be better.
Pros
- Comfortable to wear
- No app subscription
- Accurate sleep tracking features
- Optional blood glucose tracking
Cons
- Patchy app connectivity
- The outer paint is easy to scratch
- Heart rate can be imprecise during intense exercise
Best screen-free fitness tracker



Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ You are a keen swimmer or triathlete: Its unique design makes it less prone to structural damage.
✅ You want detailed health and fitness tracking: This wearable provides a wealth of in-depth data.
Don't buy it if
❌ You are an endurance athlete: No way to quickly check the heart rate.
❌ You are on a budget: The membership is quite pricey.
The bottom line
🔎 Whoop MG: With its unique design, the Whoop truly is an all-day wearable. And no screen will be a big bonus for those who like to avoid distractions and notifications. It offers impressively detailed health metrics, but there's no way to check your heart rate on the go, and the ongoing membership is quite pricey.
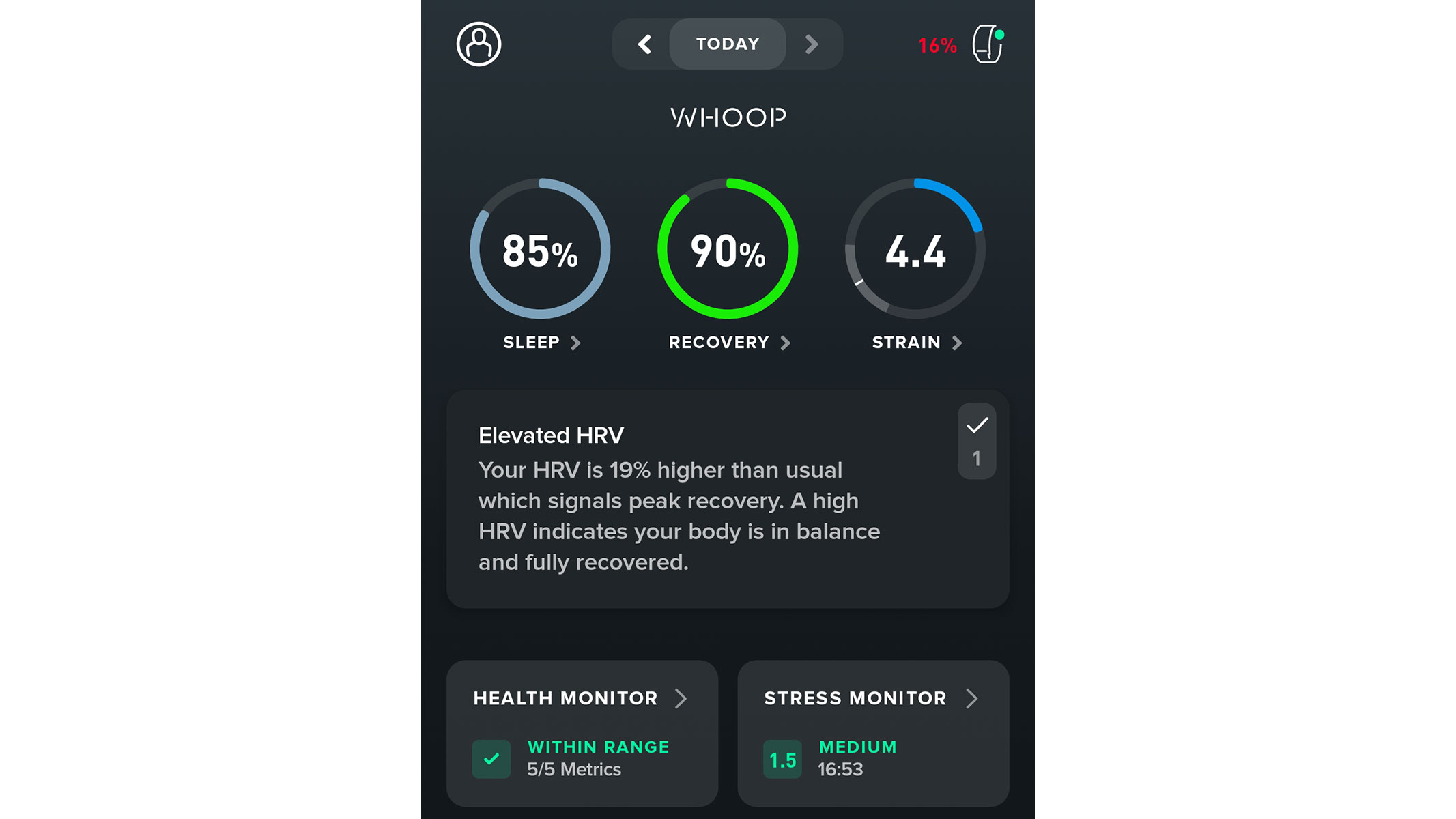
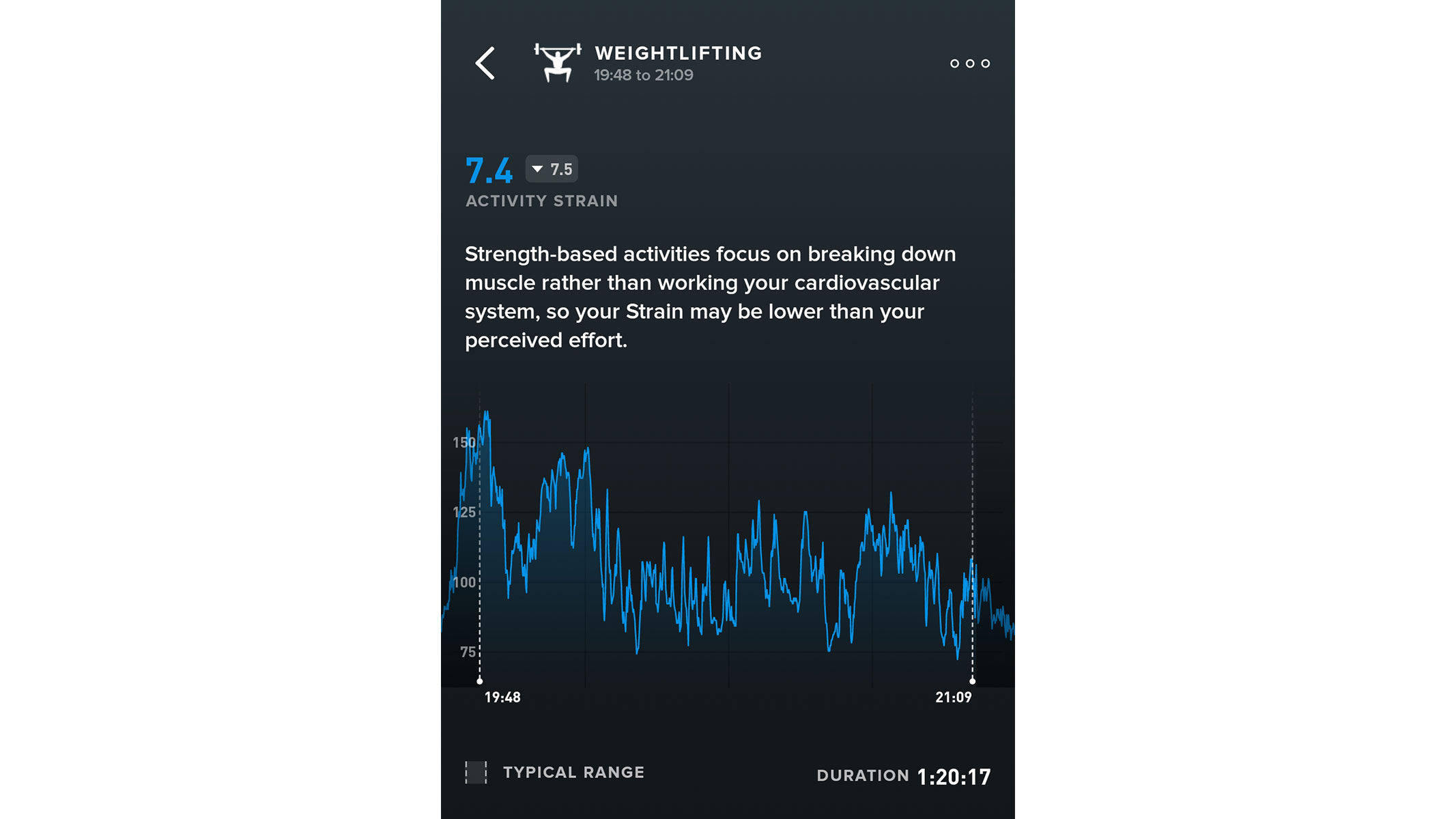
The Whoop MG does not resemble any other fitness tracker on the market — but that is what makes it so special. This peculiar-looking wearable lacks a giant touchscreen or bulky buttons; instead, it comes with a discrete “sensor suite” and a woven, elasticated strap. What you get, however, is continuous monitoring of health metrics including heart rate, skin temperature and heart rate variability, as well as in-depth sleep tracking. This data is then combined and analysed to produce three easy-to-understand daily scores; strain, sleep and recovery.
During our Whoop MG review, we tested it for nearly three months, and found this alternative approach had both benefits and drawbacks. This fitness tracker will not find any devoted fans among endurance athletes, since it does not have a screen, in-built GPS or other features that display stats while running. On the other hand, its discreet, screenless design will appeal to swimmers and weightlifters.
The Whoop MG feels sturdy, durable and shows no signs of damage after being smacked with a dumbbell or submerged in a swimming pool. It really is a game-changer.
The strain, sleep and recovery scores allowed us to get the most out of our training sessions without having to ponder on the individual metrics. But if you like your numbers, you can still get plenty of in-depth data on the Woop's dedicated app. The Whoop MG is a fabulous one-of-a-kind wearable and we can thoroughly recommend it if you are tired of classic fitness watches.
- Read our full Whoop MG review
Best hybrid smartwatch




Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ You need something stylish and discreet: It looks like an analog wristwatch.
✅ You want in-depth insights into your heart health: It tracks ECG and offers cardiologist-approved heart health reviews.
Don't buy it if
❌ You want to improve your exercise performance: It does not offer advanced workout-tracking features.
❌ You struggle with small displays: It can be difficult to read.
The bottom line
🔎 Withings ScanWatch 2: Lightweight, stunningly elegant and jam-packed with health insights, this hybrid smartwatch is a great option for those who want to monitor their heart health closely. Athletes, however, may be disappointed with its limited workout tracking features.
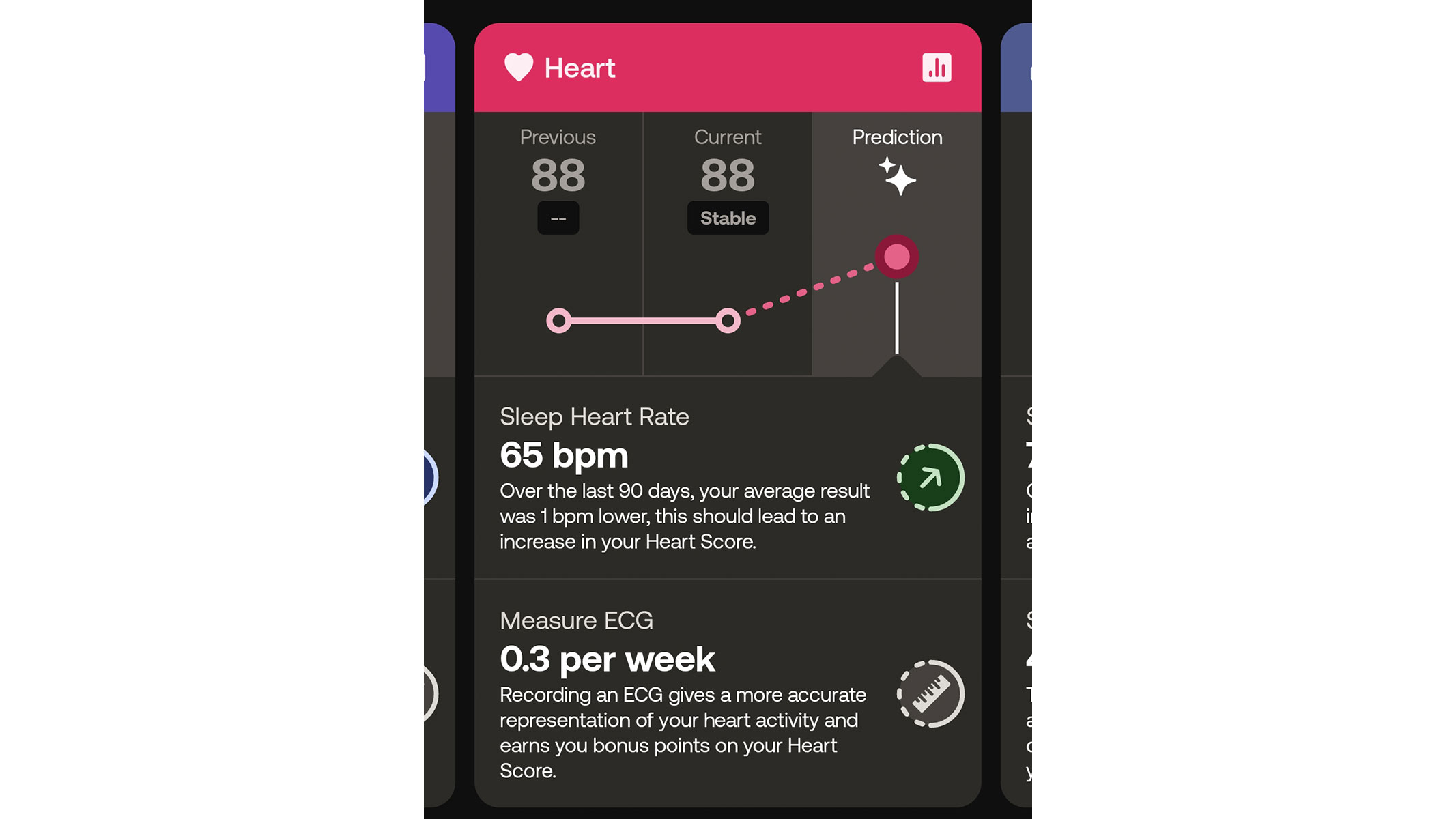
Most fitness trackers are designed to perform, not to look good. If you are tired of chunky, rugged wearables that overload your wrist and ruin your formal outfits, consider a hybrid smartwatch instead. These devices combine smart technology and advanced tracking features with the stylish looks and compact design of a traditional, analog wristwatch. The Withings ScanWatch 2 is a perfect example of a hybrid smartwatch: it is stunningly elegant and exceptionally discreet, but also jam-packed with health and fitness insights.
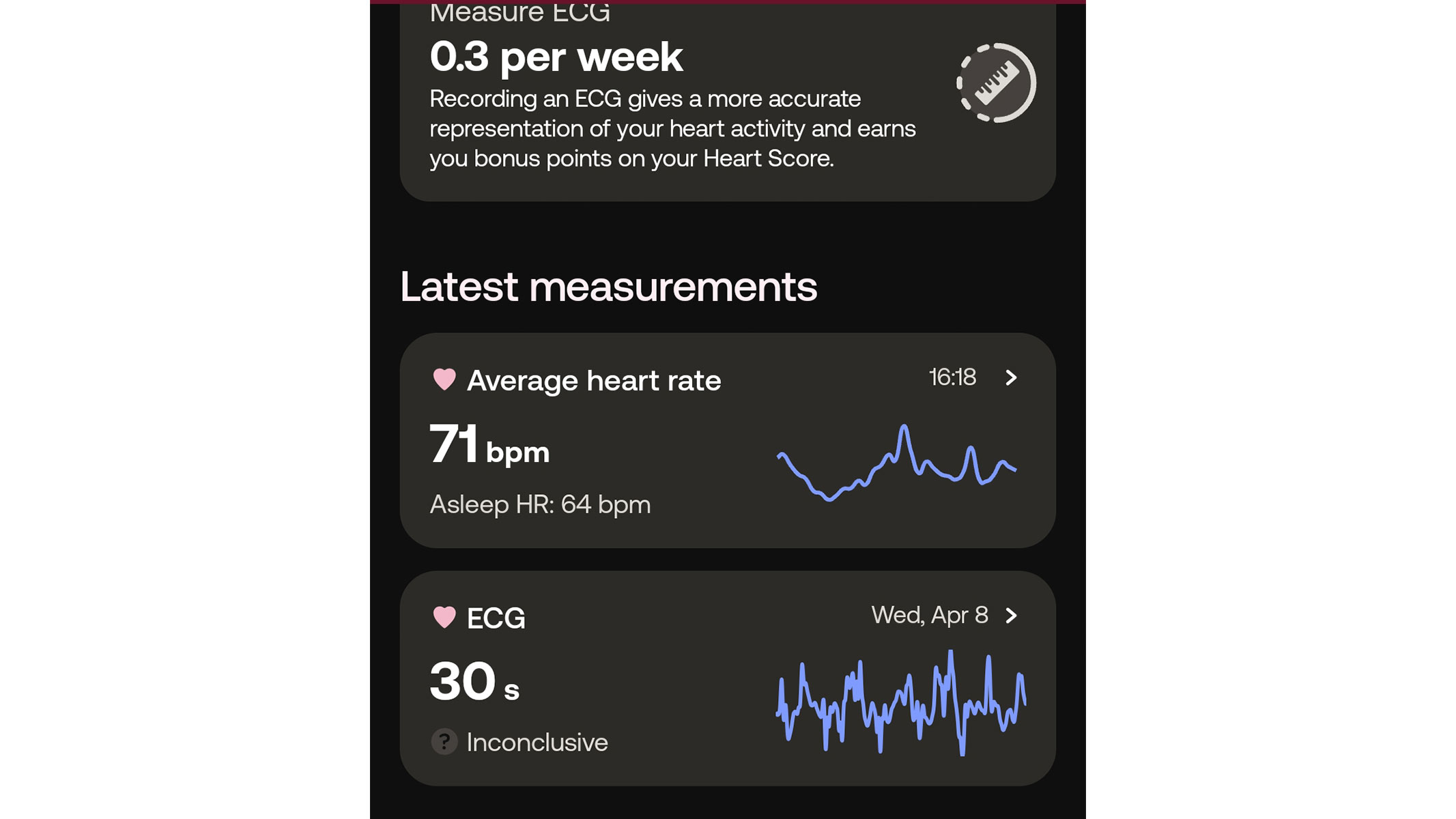
This wearable is a particularly good pick for those who want to keep a close eye on their cardiovascular health. The Withings ScanWatch 2 not only tracks your heart rate and heart rate variability, but also measures your ECG. As such, it is one of the handful of fitness trackers that can detect irregular heart rhythm and alert you to potential signs of arrhythmia and atrial fibrillation. Moreover, it is the only wearable that allows you to request in-depth cardiovascular health reviews conducted by board-certified cardiologists.
That said, the Withings ScanWatch 2 may not be well-suited for athletes focused on improving their exercise performance. It does not offer advanced workout tracking features and has no built-in GPS. We would also not recommend it to those who struggle with tiny displays.
- Read our full Withings ScanWatch 2 review
Best fitness tracker for beginners






Specifications
Reasons to buy
Reasons to avoid
Buy it if
✅ You want to hit your daily step goal: It is the first Garmin watch to feature structured walking workouts.
✅ You want good value for money: Functionality of the high-end Garmin watches, but without the hefty price tag.
✅ You want exercise guidance: It comes with handy exercise animations.
Don't buy it if
❌ You are an advanced exerciser: Its workouts are geared towards people who have never exercised before.
❌ You are a keen swimmer: It may not survive deep dives.
The bottom line
🔎 Garmin Vivoactive 6: Sleek, lightweight and easy to use, this first-ever Garmin watch with structured walking workouts is a great option for exercise beginners. Athletes and runners, however, may not like its limited features.
Many fitness beginners start their fitness journey by hiring a personal trainer, and understandably so. Having someone show you how to perform exercises safely and effectively can help you maximize your results and avoid setbacks. However, fitness coaching can be quite expensive in the long term — and this is where fitness wearables can step in. While they are nowhere near as effective as their human counterparts, smartwatches can act as your pocket-sized personal trainer at a fraction of the cost, helping you pick the right workouts and advising when to push and when to rest. That said, some wearables are more suitable for beginners than others. If you have never exercised before, we would recommend the Garmin Vivoactive 6.
This smartwatch is sleek, intuitive to navigate and easy to understand, and it looks good, too. More importantly, the Garmin Vivoactive 6 offers excellent health tracking and plenty of beginner-friendly features. In fact, it is the first Garmin smartwatch with guided walking workouts. We particularly liked its exercise animations. Since these short clips can be displayed on your watch, they can always tell you how to perform a certain movement, no matter if you are at home, in the gym or attending an outdoor class. During our full Garmin Vivoactive 6 review, we even compared this model to a pocket-sized personal trainer.
We also liked its reasonable price tag — approximately $299.99. While it is not particularly budget-friendly, it is still relatively cheap for a Garmin watch, and the value for money is immense. This watch has the potential to help beginners get a footing in the fitness world, and this is one of the most important points for those who do not feel confident about working out.
- Read our full Garmin Vivoactive 6 review
Best fitness trackers: Comparison
Model | Rating | Screen | Water resistance | GPS | Compatibility | Battery life |
|---|---|---|---|---|---|---|
Amazfit Balance | 4.5 | AMOLED 1.5-inch | 5ATM | Yes | iOS, Android | 14 days |
Garmin Fenix 8 | 4.5 | AMOLED 1.4-inch | 10ATM, dive 40m | Yes | iOS, Android | 16 days |
Xiaomi Smart Band 10 | 4.5 | AMOLED 1.72-inch | 5ATM | No | Android 8.0 or iOS 14.0 and above | 21 days |
Amazfit Active Max | 4.5 | AMOLED 1.5-inch | 5ATM | Yes | iOS, Android | 25 days |
Garmin Forerunner 965 | 4.5 | AMOLED 1.4-inch | 5ATM | Yes | iOS, Android | 23 days |
Suunto Vertical 2 | 4.5 | AMOLED 1.5-inch | 10ATM | Yes | iOS, Android | 20 days |
Oura Ring Gen 4 | 4 | N/A | 10ATM | No | iOS, Android | 8 days |
Whoop MG | 3.5 | N/A | IP68 | No | iOS, Android | 14 days |
Garmin Vivoactive 6 | 4 | AMOLED 1.2-inch | 5ATM | Yes | iOS, Android | 11 days |
Withings ScanWatch 2 | 4 | 0.63-inch OLED | 5ATM | No | iOS, Android | 35 days |
Our expert consultants

Nathan Kennedy is a UK-based certified personal trainer, online fitness coach, and a representative for Bio-Synergy, a sports nutrition brand. He told Live Science about the potential health and fitness benefits of using fitness trackers.

Dr. Alka Patel is a lifestyle medicine physician and longevity expert based in London. She achieved a distinction in the MRCGP (Membership of the Royal College of GPs) and 10 more titles including a Diploma in Lifestyle Medicine. For this piece, Dr. Patel commented on the validity of calorie measurements and sleep tracking features.

Dr. Mark Estes III is a Pittsburgh-based cardiologist and past chair of the American Heart Association’s scientific publishing committee and Council on Clinical Cardiology. For this piece, he commented on the use of fitness trackers for detecting cardiovascular problems.
Best fitness trackers: Frequently Asked Questions
Are fitness trackers worth it?
In short, yes. "Fitness trackers have many useful features and have helped myself and my clients with data collection you can then use to make better fitness decisions," Nathan Kennedy, a certified personal trainer, told Live Science. "Most fitness trackers track number of steps taken, heart rate, and sleep patterns. Some also have connected apps that allow you track your overall progress, set goals as well as offer guidelines to help you achieve your fitness goals."
Many fitness trackers have “stress” monitoring, and guided mindfulness breathing exercises to help you lower that stress level. Even cheap models will also relay notifications received, and this can be a useful if you want to spend less time on your phone after work.
Can fitness trackers make you fitter?
Fitness trackers can help you get fitter and healthier. Studies have shown that using smart wearables can help you increase physical activity levels and improve body composition, according to a 2022 meta-analysis published in the journal Lancet Digital Health.
"Fitness trackers not only encourage exercise and weight loss, but may also help lower blood pressure and cholesterol in people with Type 2 diabetes and other health conditions," Kennedy told Live Science.
Do fitness trackers help with motivation?
In short, yes, they can. "Accountability is much higher when using a fitness tracker as you get the sense personal motivation or if you’re in a group challenge with your friends," Kennedy said. "Trackers can provide feedback to help people monitor their progress toward activity goals and can remind wearers of them. Some trackers come with or integrate with mobile apps that inspire a variety of behavior changes. Ideally, trackers can help users to develop lifestyles that make it easier to integrate physical activity into daily life. The easier it is for the user to get into a lifestyle that works for them the bigger the chance of them achieving their end goal is."
When is Prime Day in October?
Amazon's October Prime Day, called Prime Big Deal Days, is scheduled to take place on Tuesday, October 7 and Wednesday, October 8, 2025. We will be on the lookout for the best deals on fitness trackers (you will find our top picks in our Prime Day smartwatch deal hub). If you are looking for something else, however, there will be plenty more excellent deals in our Prime Day science deals hub.
Everything you need to know about fitness trackers
Are fitness trackers accurate?
Fitness trackers record a bunch of metrics, each of which introduces accuracy headaches. For step counting, how does a tracker separate steps from hand movements while you work sat at a desk? How can a yoga tracking mode really judge calories burnt when it does not know your muscle tension or perhaps even the positions you adopt?
The best fitness trackers now offer good reliability in a few crucial areas, though, heart rate and location — which look at the distances you run or walk.
We find Apple Watches and some of the latest Garmin watches, like the Fenix 7, are top-tier performers for heart rate tracking. Apple led the pack for years, but others are starting to catch up. A 2019 study published in the Journal of Medical Internet Research found the Apple Watch offered “clinically acceptable accuracy” for HR tracking during exercise, a higher standard than it might sound — and that was for an old Apple Watch variant. However, it also found the watch overestimated calories burnt.
Almost all fitness trackers with GPS will provide fairly good distance stats, particularly if you give them time to lock-on properly before starting your session.
A lot of other trackers still struggle a little with heart rate readings, though. Interval training and very high exertion in general can be problematic, although in truth this is not a huge issue for most people. Are you really going to pore over the heart rate graph in your tracker’s app?
SpO2 (blood oxygenation) readings can be more problematic, because we find most wearables can throw out highly suspect results at times. You will often get better results on a second or third attempt, but we frequently see readings that suggest we should head straight to the hospital from high-end trackers.
That said, a study published in Nature concluded the old Apple Watch Series 6 is a reliable enough way to monitor SpO2 in patients with lung conditions.
The best way to approach a fitness tracker’s stats is to compare your exercise results to your own, from previous sessions. Comparing with a friend’s tracker will expose the way different brands’ algorithms color the results.
You should also make sure your tracker’s strap is snug and firmly strapped on before starting a tracked exercise session. This will get you the best results from the heart rate sensor.

Do fitness trackers accurately count calories?
Most fitness trackers can tell you how many calories you burn, and some models can even guide you on the choice of activity that matches your energy requirements. However, these calorie measurements may not be entirely accurate, according to a 2020 systematic review and meta-analysis published in the British Journal of Sports Medicine.
Researchers pulled results from 60 studies and found that, on the whole, fitness trackers tended to underestimate energy expenditure, especially during sedentary and household tasks. They also noted that different wearables can produce vastly different readings.
That is because fitness trackers do not measure calories directly — they use a combination of different data points to assess your approximate energy expenditure. These metrics often include physical activity levels, heart rate and basal metabolic rate (BMR), meaning the minimum number of calories the body needs to maintain normal bodily functions, such as breathing or pumping blood, Dr. Alka Patel, a lifestyle medicine physician and longevity expert based in London, told Live Science.
Most devices calculate your BMR based on height, weight and age, and then put this information into a complex algorithm. This means that calorie values obtained by a fitness tracker may not be as accurate as those obtained through validated clinical tests. However, these devices may still be useful for monitoring calorie trends, meaning changes in energy expenditure over time, Patel said.
To make sure your fitness tracker is as accurate as possible, keep your device close to the body, Patel noted. Most wearables use optical sensors, meaning they use light to detect changes in blood flow under the skin. “If you're wearing a loose-fitting bracelet or loose-fitting ring, the readings are not going to be accurate,” she said.
Can fitness trackers measure blood pressure?
Right now there’s only one mainstream consumer wearable that has true standalone blood pressure monitoring, the Huawei Watch D. And it is not widely available.
Samsung has offered blood pressure readings in its top watches since 2020’s Galaxy Watch 3. However, the feature is only unlocked in certain countries/markets, and is much more limited than the Huawei Watch D’s version.
Where the Huawei Watch D adopts the method used by blood pressure cuffs with a specialist strap that inflates, the Galaxy Watch uses the heart rate reader on the back to evaluate pulse transit time. Fitbit is researching a similar method, using pulse arrival time.
However, such methods that do not demand bespoke hardware do need to be regularly recalibrated using a traditional blood pressure cuff. Samsung says results should be taken with one every four weeks, for example.
Rumors of a blood pressure feature for Apple Watches have circulated for a while, but the latest report suggests we won’t see it released until September 2024. However, you can use an Apple Watch to log blood pressure readings taken elsewhere, in the Apple Health app. Apple sells Withings’s BPM Connect smart blood pressure cuff at the Apple Store.
Blood pressure via a wearable? It’s not quite there yet. And as the Huawei Watch D is expensive, hard to get hold of, and something we have not tested, you are better off with a more traditional cuff-style design for now.

Can fitness trackers detect sleep apnea?
Sleep apnea is a disorder that causes repeated bouts of breathlessness or shallow breathing during sleep. Around 30 million people in the US may have this condition, but the vast majority of cases are not diagnosed and treated, according to the American Medical Association. That is because the key symptoms of sleep apnea — gasping for air, loud snoring and lapses in breathing — occur throughout the night, and people with this disorder are not awake when they happen.
So given that many fitness trackers come with sleep tracking features, can smart wearables detect sleep apnea?
In short: not yet. Devices currently available on the market are not accurate enough to provide a valid diagnosis of sleep apnea. However, fitness trackers have potential to be used in the future, according to a 2020 review published in the Journal of Thoracic Disease.
Fitness trackers provide several data points that, when combined and interpreted together, can indicate whether someone has sleep apnea, Dr. Alka Patel, a lifestyle medicine physician and longevity expert based in London, told Live Science. People with this condition often sleep for fewer hours and have worse quality of sleep than individuals without sleep apnea. For example, they tend to spend less time in deep (non-REM) sleep, the stage of sleep critical for growth and recovery, she said.
During an episode of sleep apnea, the heart rate and oxygen saturation levels may drop, and then increase and recover after the episode is over. People with sleep apnea also tend to toss and turn in their sleep. All of these parameters can show up on a fitness tracker. However, it is for a healthcare professional to put these metrics together, interpret them and then confirm the sleep apnea diagnosis with clinically-approved tests, Patel said.
That is because commercially available fitness trackers often under- or overestimate sleep duration and may not accurately distinguish whether someone is awake or not. They may also fall short of providing reliable cardiovascular measurements or accurately detecting sleep stages, the 2020 review authors noted.
If you are concerned about the readings on your fitness tracker, it is best to consult your doctor.
Can fitness trackers detect heart problems?
Fitness trackers can tell you a lot about the inner workings of your heart, such as the frequency of your heartbeat. Some devices can even help you choose the best activities to boost your cardiovascular health. But can fitness trackers detect heart problems, too?
The answer to this question is not straightforward.
“There have been many advancements in the technology used in fitness trackers, and the ability for people to track their heart rate is helpful, but it shouldn’t be a replacement for medical care,” Dr. Mark Estes III, a Pittsburgh-based cardiologist and past chair of the American Heart Association’s scientific publishing committee and Council on Clinical Cardiology, told Live Science by email.
Smart wearables tend to use photoplethysmography, a technology that employs optical sensors to detect blood flow and determine heart rate. If you look at the underside of a smartwatch, you might see a bright light emitted by one of these sensors. Studies have shown that heart rate measurements picked up by fitness trackers tend to be relatively accurate. Still, they are not as precise as electrocardiogram (ECG) ― a recording of electrical signals in the heart obtained by placing electrodes on the skin, which is used to diagnose conditions like coronary heart disease and arrhythmias — according to a 2020 meta-analysis published in the Journal of Sports Sciences.
The accuracy of heart rate readings also varies between different devices, and may depend on activity. For example, fitness trackers tend to be more precise at measuring heart rate during sleep than during high-intensity exercise, such as running.
Lastly, heart rate alone may not be enough to identify a cardiovascular condition, Peter Charlton, a British Heart Foundation Research Fellow in the Department of Public Health and Primary Care at the University of Cambridge, told Live Science.
However, certain smart wearables may be able to detect signs of atrial fibrillation, a condition in which the heart beats too quickly, too slowly or has an irregular rhythm.
“Some fitness trackers can identify an irregular heart rhythm, and their accuracy is sufficient to warrant a further clinical investigation,” Charlton said. But again, wrist-based devices cannot fully substitute for an ECG test, he cautioned.
For example, a 2022 study published in the journal Circulation found that some Fitbit devices were highly accurate at predicting whether someone may have atrial fibrillation. However, these fitness trackers could not identify the condition during exercise. That is because photoplethysmography sensors could only detect heart rhythm irregularities during periods of inactivity, the study authors noted.
The takeaway? “If you receive any alerts from your fitness tracker about possible abnormalities or experience any other symptoms like dizziness or shortness of breath, it’s important to contact your doctor for further testing,” Estes said.

Why is GPS important in fitness trackers?
GPS is arguably the most powerful tool for fitness trackers. It’s why runners, keen walkers and cyclists should make sure their next fitness tracker has it.
GPS triangulates your position with the help of satellites. One of these satellite arrays is called “GPS”, but today’s best watches also support others like GLONASS and Galileo. Each of these satellite arrays is maintained by a different country, or set of countries.
By tracking your location, fitness trackers can produce maps of your runs, hikes and rides. These make historical records much more useful, so you can actually remember which workout an entry refers to.
Accurate distance data is also important because it affects so many other readings. If the recorded distance is too low, the tracker will think you are slower than you are. And this in turn could affect fitness metrics like VO2 Max. It works the other way around too, of course — too long a recorded distance and the tracker will say you are faster than you really are.
We always recommend a full GPS watch to runners in particular, but there is a second type of GPS. It’s Connected GPS. This is where the tracker does not have its own GPS chip, but can take GPS data from your phone, over Bluetooth.
Many cheaper trackers without GPS have Connected GPS. And if you plan on running with your phone anyway, to listen to music or podcasts, it’s a decent stand-in.
With no GPS at all, your tracker has to extrapolate distance using its motion sensors. It can count your strides and evaluate the pace of them to differentiate between running and walking. This can work pretty well with a good algorithm running behind the scenes, but GPS is better. And considering how cheap Connected GPS wearables get, we don’t recommend entirely non-GPS watches to many folks these days.
How to choose the best fitness tracker for you
How to chose the best fitness tracker
Choosing the best fitness tracker is first and foremost about deciding what you want to use it for. If you want a lifestyle device that will measure sleep, resting heart rate and take a broad view at your health, then you will need a different device to something that is running-specific. So first, decide what you will be primarily using it for.
This will also impact your budget. If you want something that will primarily act as a step counter and track your heart rate, then a simple, budget-friendly tracker will suit your needs perfectly.
If you do a lot of running or biking, however, you will probably want a device that has built-in GPS to track your pace and distance covered as well as heart rate and more. You may also want a tracker that will actually suggest runs and cycle workouts for you, optimising to your own personal metrics like how well you have recovered from your last workout.
If you are more than an occasional swimmer, you may also want a tracker that will count laps for you, and of course that is robustly waterproof.
The most up to date smartwatches have fitness-tracking metrics built in, and can also do things like control your music and help you reply to messages you receive on the go. These can be extremely useful allowing you to even pay for purchases with the same device that is tracking your run. However, if. you want deep dive metrics that go into detail about your chosen sport, these general smart watches may not be for you. Any decision you make will come with trade-offs, whether it is in battery life or in metrics offered, so with our help you can identify the tracker that ticks all your personal boxes and does everything that you need it to do.
Garmin vs Fitbit: which is better?
Garmin vs Fitbit, which should you choose? Generally, Garmins are for fitness nerds, Fitbits are for those who would prefer a tracker with a more friendly face. That has been the traditional take on these two brands, and it still mostly holds up in 2025.
Garmin’s trackers tend to offer significantly better core stat accuracy and reliability than Fitbits. The latest models also have more advanced GPS navigation. However, Fitbits tend to have a more deliberate sense of style, and their prices don’t reach anything like the heights of the top Garmin trackers. The Fitbit Sense 2 and Charge 6 also have a couple of features not seen in Garmin rivals, such as ECG/EKG hardware to monitor sinus rhythm.
Fitbit vs Apple Watch: which is better?
Fitbit vs Apple Watch, which is better? Fitbit makes a wider array of wearables than Apple. Conversely, all Apple Watches look roundly similar aside from how much of the front is covered by a screen. The Fitbit Versa and Sense watches get much closer to the Apple Watch style. However, even these last significantly longer than Apple’s watches off a charge.
This makes Fitbits better for sleep tracking, and Fitbit has one of the best sleep-tracking features on the market. On the other hand, an Apple Watch will have a better heart rate sensor than a Fitbit’s. It also has the best app ecosystem of any watch.
How we tested these fitness trackers




Our reviewers are seasoned fitness professionals and sports enthusiasts who are deeply aware of all the benefits that a reliable tracker can bring to your health and fitness. Most of them own several different models and wear them interchangeably, based on how and where they intend to use them. You can trust their judgments — our expert reviewers have tested plenty of wearable tech over the years and can tell if something does what it says on the tin, whether it is a rugged GPS smartwatch for high-octane outdoor adventures or a compact fitness band for tracking general fitness.
While we may have our favorite picks, we pride ourselves on being as objective as possible in our reviews. We test all fitness trackers to the exact same standard, using the same procedures. After spending at least a week (and often months) with each testing unit, we rank it across the following categories:
1) Design. We look into the tracker's size, weight, appearance and overall functionality. We assess whether it is comfortable to wear (day and night), sufficiently customizable (for example, can you remove its straps? Is this model available in different sizes?) and compatible with various outfits. We also assess its durability based on the materials it is made of and inform of any scuffs and scratches our fitness tracker sustained during testing.
2) Display. We assess the heart of a fitness tracker — its display. We look into the size, brightness and resolution of the screen, and make note of any protective measures (scratch-resistant Gorilla glass, for example) and design tweaks that can enhance user experience. We check whether it is easy to read and navigate in various conditions (at night, in direct sunlight, swimming, etc.) We also assess the touchscreen's responsiveness and report any lags or issues we experienced during testing.
3) Features. We give our verdict on the functionality of the fitness tracker in question. How many health metrics and activities does it track? How in-depth are these tracking features? Does this fitness tracker come with any smart features like voice control and compatibility with third-party devices? Can it store music, pay for your groceries or guide you on the hiking trail? Also, can it provide personalized health advice, and if so, is it useful and easy to understand?
4) Performance. We put the wearable through a range of activities to test its battery life, general tracking accuracy and app connectivity. We wear it during the day, while doing various strength and cardio workouts, and during sleep, regularly switching between different settings. Lastly, we assess the validity of its heart rate tracking features using a chest-strap heart rate monitor (such as the Polar H9), and (if applicable) compare its GPS readings to a more high-end navigation system.
Verdict. Finally, we give our answer to the key question: should you buy this fitness tracker? After testing, we calculate a final score out of five and advise on the pros and cons of the tested fitness tracker.
Latest updates
May 19, 2026: We added the Withings ScanWatch 2 as the best hybrid smartwatch. April 14, 2026: We added the Xiaomi Watch 5 as the best Google Watch alternative.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Andrew Williams is a freelance journalist based near London. He has written about tech for over a decade, contributing to sites such as WIRED, TechRadar, TrustedReviews, Wareable, Stuff, T3, Pocket-lint and many others. When he's not covering fitness tech, he writes about mobile phones and computing, as well as cameras.
- Chris McMullenContributing writer
- Kate Carter
- Harry BullmoreFitness writer

 Live Science Plus
Live Science Plus







