What is DNA?
Learn about what DNA is made of, how it works, who discovered it and other interesting DNA facts.

Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
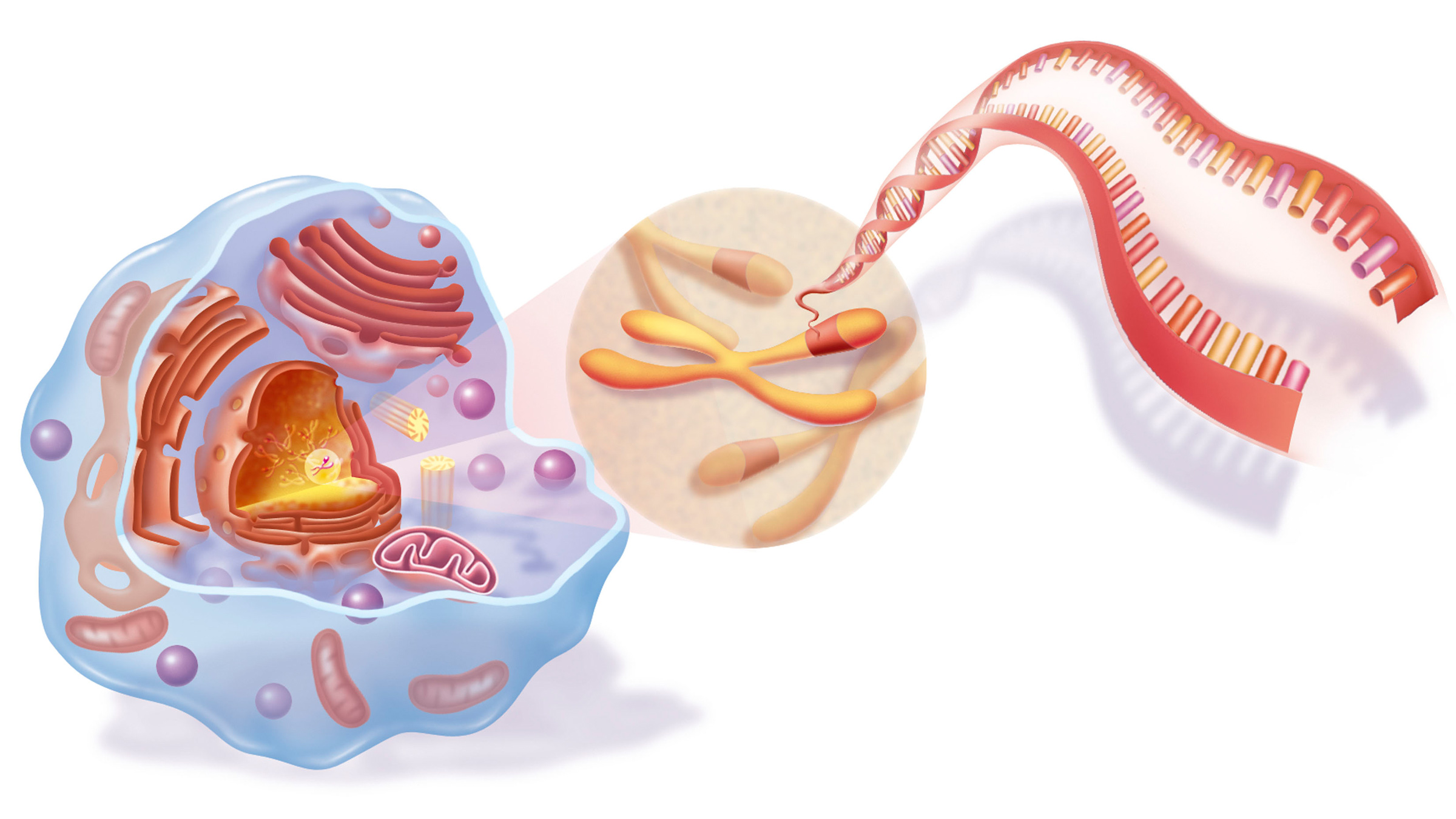
DNA stands for deoxyribonucleic acid, and it's a molecule that supplies the genetic instructions that tell living creatures how to develop, live and reproduce. DNA can be found inside every cell and is passed down from parents to their offspring.
What is DNA made of?
DNA is made up of molecules called nucleotides. Each nucleotide contains three components: a phosphate group, which is one phosphorus atom bonded to four oxygen atoms; a sugar molecule; and a nitrogen base. The four types of nitrogen bases are adenine (A), thymine (T), guanine (G) and cytosine (C), and together, these serve as the "letters" that make up the genetic code of our DNA.
DNA has a structure called a double helix. Nucleotides are attached together to form two long strands that spiral to create this structure. If you think of the double-helix as a ladder, the phosphate and sugar molecules would be the sides, while the base pairs would be the rungs. The bases on one strand pair with the bases on another strand: Adenine pairs with thymine (A-T), and guanine pairs with cytosine (G-C).
Article continues belowHuman DNA is made up of around 3 billion base pairs, and more than 99% of those bases are the same in all people, according to the U.S. National Library of Medicine (NLM).
Related: How accurate are DNA tests?
Similar to the way that letters in the alphabet can be arranged to form words, the order of nitrogen bases in a DNA sequence forms genes, which, in the language of the cell, tell cells how to make proteins. The shorthand for this process is that genes "encode" proteins.
But DNA is not the direct template for protein production. To make a protein, the cell makes a copy of the gene, using not DNA but ribonucleic acid, or RNA. RNA shares a similar structure to DNA, except it contains only one strand, rather than two — so it looks like just one half of a ladder. In addition, while RNA has three of the four nitrogen bases in common with DNA, it uses a base called uracil rather than thymine to pair with adenine.
Get the world’s most fascinating discoveries delivered straight to your inbox.
As a cell prepares to build a new protein, its DNA unzips to expose one strand of the gene with the instructions to build said protein. Then, an enzyme zooms in and constructs a new RNA molecule whose sequence mirrors that of the unzipped gene. This RNA copy, called messenger RNA (mRNA), tells the cell's protein-making machinery which amino acids to string together into a protein, according to "Biochemistry" (W. H. Freeman and Company, 2002).
DNA molecules are long — so long, in fact, that they can't fit into cells without the right packaging. To fit inside cells, DNA is coiled tightly to form structures called chromosomes. Each chromosome contains a single DNA molecule, wrapped tightly around spool-like proteins called histones, which provide chromosomes their structure. Humans have 23 pairs of chromosomes, which are found inside each cell's nucleus, the control center of the cell.
Most chromosomes look like microscopic Xs; that said, humans and most other mammals carry a pair of sex chromosomes that can be either X or Y-shaped, according to the National Human Genome Research Institute. In general, females carry two X sex chromosomes in each body cell and males carry one X and one Y. But there is some natural variation in the number of sex chromosomes people carry — sometimes, there may be extra sex chromosomes, or one might be missing, so other patterns, such as X, XXX, XXY and XXYY, can also occur, Discover reported.
Who discovered DNA?
DNA was first observed by Swiss biochemist Friedrich Miescher in 1869, according to a paper published in 2005 in the journal Developmental Biology. Miescher used biochemical methods to isolate DNA — which he then called nuclein — from white blood cells and sperm, and determined that it was very different from protein. (The term "nucleic acid" derives from "nuclein.")
But for many years, researchers did not realize the importance of this molecule.
In 1952, chemist Rosalind Franklin, who was working in the lab of biophysicist Maurice Wilkins, used X-ray diffraction — a way of determining the structure of a molecule by the way X-rays bounce off it — to learn that DNA had a helical structure. Franklin documented this structure in what became known as Photo 51.
In 1953, Wilkins showed the photo to biologists James Watson and Francis Crick — without Franklin's knowledge. Armed with the information that DNA was a double helix and previous reports that the bases adenine and thymine occurred in equal amounts within DNA, as did guanine and cytosine, Watson and Crick published a landmark 1953 paper in the journal Nature. In that paper, they proposed the iconic double-helix model of DNA as we now know it, with sugar-phosphate sides and rungs made up of A-T and G-C base pairs. They also suggested that, based on their proposed structure, DNA could be copied — and, therefore, passed on.
Watson, Crick and Wilkins were awarded the Nobel Prize in medicine in 1962 "for their discoveries concerning the molecular structure of nucleic acids and its significance for information transfer in living material." Franklin was not included in the award, even though her work was integral to the research.
What is the function of DNA?
Genes encode proteins that perform all sorts of functions for humans (and other living beings).
The human gene HBA1, for example, contains instructions for building the protein alpha globin, which is a component of hemoglobin, the oxygen-carrying protein in red blood cells, according to the NLM. To take another example, the gene OR6A2 encodes an olfactory receptor, a protein that detects odors in the nose, according to the National Center for Biotechnology Information's Gene database. Depending on which version of OR6A2 you have, you may love cilantro or think it tastes like soap, according to a study published in 2012 in the journal Flavour.
Although each one of your 37.2 trillion cells carries a copy of your DNA, not all cells build the same proteins. One reason for this is that molecules called "transcription factors" latch onto DNA to control which genes get switched on and off, and therefore, which proteins get made when, where and in what quantities in each cell, Live Science previously reported. DNA also gets packaged slightly differently in different cell types, and this influences how and where transcription factors can grab onto the molecule.
In addition, epigenetics — which literally means "above" or "on top of" genetics — refers to external modifications to DNA that turn specific genes on or off.
For instance, small molecules called methyl groups can attach to a DNA strand and prevent specific genes from being expressed. Another example of epigenetics is called "histone modification," where changes to the spool-like proteins inside chromosomes can make specific segments of DNA more or less accessible to the proteins that "read" genes. Such epigenetic changes to DNA can be passed onto future generations if the changes occur in sperm or egg cells, Live Science previously reported.
How is DNA sequenced?
DNA sequencing involves technology that allows researchers to determine the order of bases in a section of DNA. The technology can be used to determine the order of bases in genes, chromosomes or an entire genome. In 2000, researchers completed a "working draft" sequence of the human genome, according to the National Human Genome Research Institute, and finished the project in 2003.
DNA testing
A person's DNA contains information about their heritage, and it can sometimes reveal whether they are at an elevated risk for certain diseases.
DNA tests, or genetic tests, are used for a variety of reasons, including to diagnose genetic disorders, to determine whether a person is a carrier of a genetic mutation that they could pass on to their children and to examine whether a person is at risk for a genetic disease. For instance, certain mutations in the BRCA1 and BRCA2 genes are known to increase the risk of breast and ovarian cancers, and a genetic test can reveal whether a person has these mutations.
Genetic test results can have implications for a person's health, and the tests are often provided along with genetic counseling to help individuals understand the results and consequences.
People also use the results of genetic testing to find relatives and learn about their family trees through companies such as Ancestry and MyHeritage.
Additional resources and reading
- Learn about how much of your DNA is just "junk" with SciShow on Youtube.
- Read more about how DNA testing works with Science News for Students.
- Check out renowned science writer Carl Zimmer's 2018 book, "She Has Her Mother's Laugh: The Powers, Perversions, and Potential of Heredity."
Bibliography
Berg, J. M., Tymoczko, J. L., & Stryer, L. (2002). DNA, RNA, and the Flow of Genetic Information. In Biochemistry (5th ed.). chapter, W H Freeman & Co.
Dahm, R. (2005). Friedrich Miescher and the discovery of DNA. Developmental Biology, 278(2), 274–288. https://doi.org/10.1016/j.ydbio.2004.11.028
Eriksson, N., Wu, S., Do, C. B., Kiefer, A. K., Tung, J. Y., Mountain, J. L., Hinds, D. A., & Francke, U. (2012). A genetic variant near olfactory receptor genes influences cilantro preference. Flavour, 1(1). https://doi.org/10.1186/2044-7248-1-22
Incorvaia, D. (2021, September 28). Sex is more complex than a simple binary suggests. Discover Magazine. Retrieved January 14, 2022, from https://www.discovermagazine.com/the-sciences/sex-is-more-complex-than-a-simple-binary-suggests
NIH National Human Genome Research Institute. (2021, February 12). Human genome project timeline of events. Genome.gov. Retrieved January 14, 2022, from https://www.genome.gov/human-genome-project/Timeline-of-Events
NIH National Human Genome Research Institute. (n.d.). Sex chromosome. Genome.gov. Retrieved January 14, 2022, from https://www.genome.gov/genetics-glossary/Sex-Chromosome
U.S. National Library of Medicine. (2020, September 28). HBA1 gene. MedlinePlus. Retrieved January 14, 2022, from https://medlineplus.gov/genetics/gene/hba1/
U.S. National Library of Medicine. (2022, January 5). OR6A2 olfactory receptor family 6 subfamily a member 2 [homo sapiens (human)] - gene - NCBI. National Center for Biotechnology Information. Retrieved January 14, 2022, from https://www.ncbi.nlm.nih.gov/gene/8590
U.S. National Library of Medicine. (2021, January 19). What is DNA?: Medlineplus Genetics. MedlinePlus. Retrieved January 14, 2022, from https://medlineplus.gov/genetics/understanding/basics/dna/
Watson, J. D., & Crick, F. H. (1953). Molecular structure of Nucleic Acids: A structure for deoxyribose nucleic acid. Nature, 171(4356), 737–738. https://doi.org/10.1038/171737a0
Additional reporting by Alina Bradford and Ashley P. Taylor, Live Science contributors, as well as Live Science staff writer Nicoletta Lanese. This page was last updated on Jan. 26, 2022.
Originally published on Live Science.

Rachael is a Live Science contributor, and was a former channel editor and senior writer for Live Science between 2010 and 2022. She has a master's degree in journalism from New York University's Science, Health and Environmental Reporting Program. She also holds a B.S. in molecular biology and an M.S. in biology from the University of California, San Diego. Her work has appeared in Scienceline, The Washington Post and Scientific American.
 Live Science Plus
Live Science Plus