What is thermodynamics?
Learn all about thermodynamics, the science that explores the relationship between heat and energy in other forms.

Ashley Hamer Pritchard

Thermodynamics is the branch of physics that deals with the relationships between heat and other forms of energy. In particular, it describes how thermal energy is converted to and from other forms of energy and how thermal energy affects matter.
Thermal energy is the energy a substance or system has due to its temperature — in other words, the energy of moving or vibrating molecules — according to the University of Kentucky. Thermodynamics involves measuring this energy, which can be "exceedingly complicated," David McKee, a professor of physics at Missouri Southern State University told Live Science. "The systems that we study in thermodynamics … consist of very large numbers of atoms or molecules interacting in complicated ways. But, if these systems meet the right criteria, which we call equilibrium, they can be described with a very small number of measurements or numbers. Often this is idealized as the mass of the system, the pressure of the system, and the volume of the system, or some other equivalent set of numbers."
Heat
Thermodynamics, then, is concerned with several properties of matter; foremost among these is heat. Heat is energy transferred between substances or systems due to a temperature difference between them, according to Georgia State University. As a form of energy, heat is conserved — it cannot be created or destroyed. It can, however, be transferred from one place to another. Heat can also be converted to and from other forms of energy. For example, a steam turbine can convert heat to kinetic energy to run a generator that converts kinetic energy to electrical energy. A light bulb can convert this electrical energy to electromagnetic radiation (light), which, when absorbed by a surface, is converted back into heat. Throughout this process, a portion of the energy is lost to entropy.
Temperature
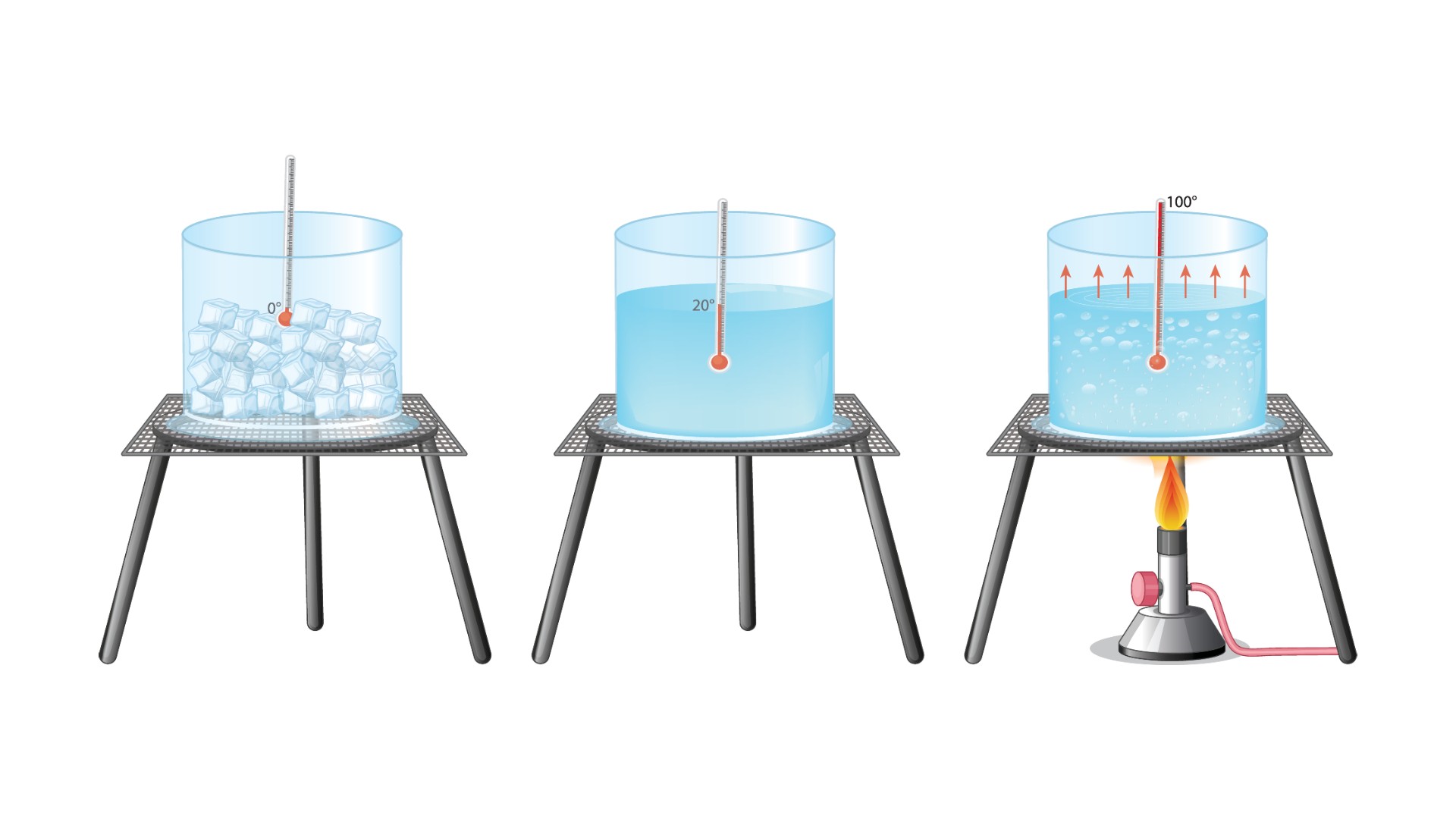
Comparing the freezing and melting points of water (in Celsius).
The amount of heat transferred by a substance depends on the speed and number of atoms or molecules of that substance in motion, according to Georgia State University. The faster the atoms or molecules move, the higher the temperature, and the more atoms or molecules that are in motion, the greater the quantity of heat they transfer.
Temperature is "a measure of the average kinetic energy of the particles in a sample of matter, expressed in terms of units or degrees designated on a standard scale," according to the American Heritage Dictionary. The most commonly used temperature scale is Celsius, which is based on the freezing and boiling points of water, assigning respective values of 0 C and 100 C. The Fahrenheit scale is also based on the freezing and boiling points of water, which have assigned values of 32 degrees Fahrenheit and 212 F, respectively.
Scientists worldwide, however, use the Kelvin (K with no degree sign) scale, named after William Thomson, 1st Baron Kelvin, because it's based on total thermal energy rather than the freezing and boiling points of water, according to the National Library of Scotland. This scale uses the same increments as the Celsius scale; for example, a temperature change of 1 C is equal to 1 K. However, the Kelvin scale starts at absolute zero, the temperature at which there is a total absence of heat energy, and all molecular motion stops. A temperature of 0 K is equal to minus 459.67 F or minus 273.15 C.
Specific heat
The amount of heat required to increase the temperature of a certain mass of a substance by a certain amount is called specific heat, or specific heat capacity, according to Wolfram Research. The conventional unit for this is calories per gram per Kelvin. The calorie is defined as the amount of heat energy required to raise the temperature of 1 gram of water at 4 C by 1 degree.
Get the world’s most fascinating discoveries delivered straight to your inbox.
The specific heat of a metal depends almost entirely on the number of atoms in the sample and the way they bond, not the sample's mass. For instance, a kilogram of aluminum can absorb about seven times more heat than a kilogram of lead. A given mass of water, however, can absorb nearly five times as much heat as an equal mass of aluminum, thanks to the strong forces binding the water molecules, according to Southeastern Louisiana University.
Thermal conductivity

A central processing unit (CPU) is an essential part of a computer's hardware components.
Thermal conductivity (k) is "the rate at which heat passes through a certain cross-section of a specified material," according to Debdatta Ratna's "Thermosets" (Woodhead Publishing limited, 2012). The unit for k is watts (W) per meter (m) per Kelvin (K), according to Swarthmore College. Values of k for metals such as copper and silver are relatively high at 380 and 420 W/m·K, respectively. This property makes these materials useful for automobile radiators and cooling fins for computer chips because they can carry away heat quickly and exchange it with the environment. The highest value of k for any natural substance is diamond at 2,200 W/m·K, according to a 2009 study published in the journal Materials.
Other materials are useful because they are extremely poor conductors of heat; this property is referred to as thermal resistance, or R-value, which describes the rate at which heat is transmitted through the material. These materials, such as fiberglass, goose down, and Styrofoam, are used for insulation in exterior building walls, winter coats, and thermal coffee mugs. R-value is given in units of square feet times degrees Fahrenheit times hours per British thermal unit (ft2·°F·h/Btu), according to OpenStax, an open-source textbook.
Newton's Law of Cooling
In 1701, Sir Isaac Newton first stated his Law of Cooling in a short article titled "Scala graduum Caloris" ("A Scale of the Degrees of Heat") in the Philosophical Transactions of the Royal Society. Newton's statement of the law translates from the original Latin as, "the excess of the degrees of the heat ... were in geometrical progression when the times are in an arithmetical progression." Worcester Polytechnic Institute gives a more modern version of this scientific law as "the rate of change of temperature is proportional to the difference between the temperature of the object and that of the surrounding environment."
This results in an exponential decay in the temperature difference. For example, if a warm object is placed in a cold bath, within a certain length of time, the difference between the two temperatures will decrease by half. Then in that same length of time, the remaining difference will again decrease by half. This repeated halving of the temperature difference will continue at equal time intervals until it becomes too small to measure. At that point, the system is in thermal equilibrium.
Heat transfer
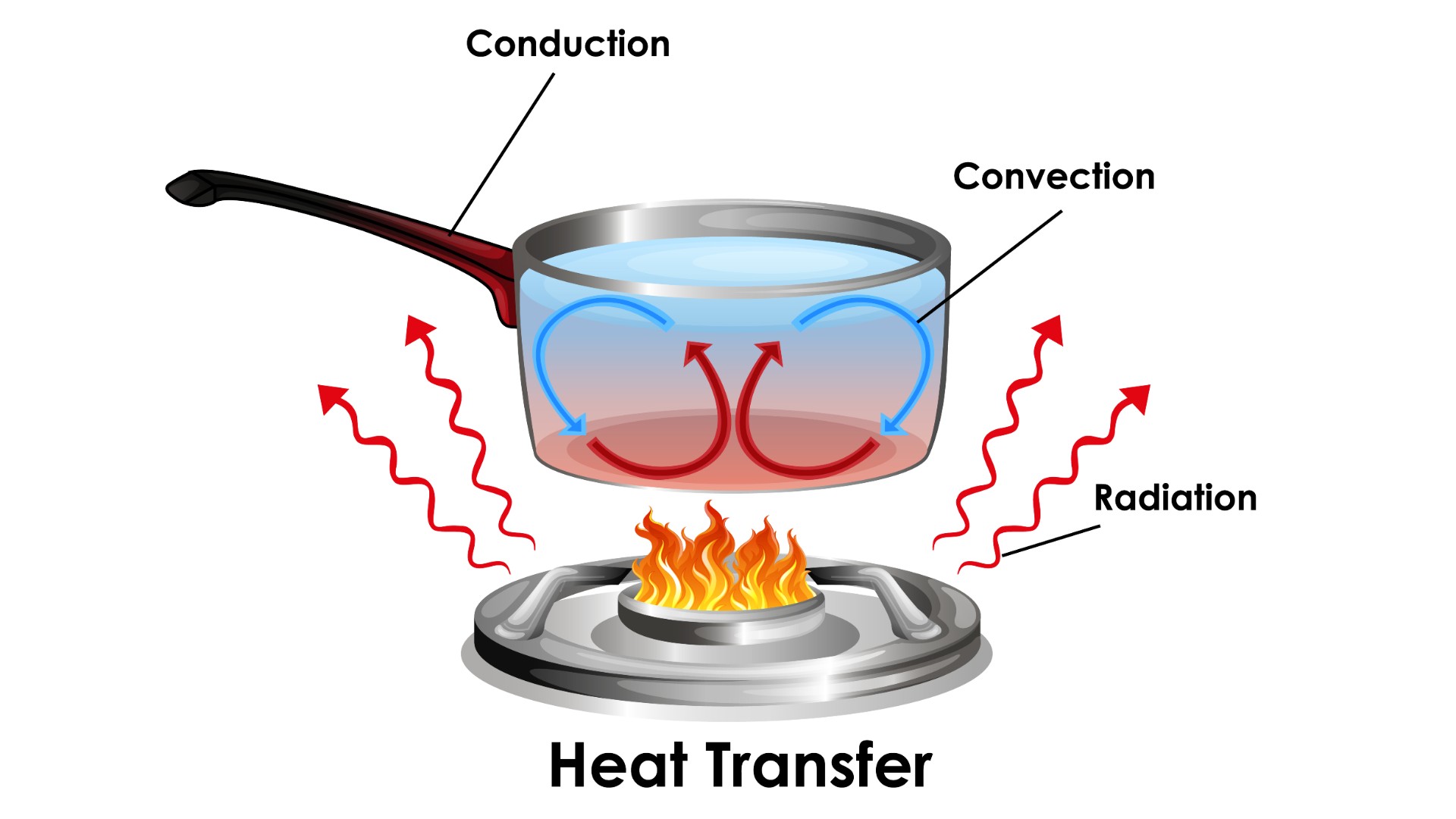
Diagram showing how heat transfer works.
Heat can be transferred from one body to another or between a body and the environment by three different means: conduction, convection, and radiation. Conduction is the transfer of energy through a solid material. Conduction between bodies occurs when they are in direct contact, and molecules transfer their energy across the interface.
Convection is the transfer of heat to or from a fluid medium. Molecules in a gas or liquid in contact with a solid body transmit or absorb heat to or from that body and then move away, allowing other molecules of the fluid to move into place and repeat the process.
Radiation is the emission of energy via electromagnetic (EM) radiation, particularly infrared photons that carry heat energy. All matter emits and absorbs some EM radiation, the net amount of which determines whether this causes a loss or gain in heat, according to Northwestern University.
The Carnot cycle
In 1824, Nicolas Léonard Sadi Carnot proposed a model for a heat engine based on what has come to be known as the Carnot cycle, according to the American Society of Mechanical Engineers. The cycle exploits the relationships among pressure, volume, and temperature of gases, as well as how an input of energy can change form and do work outside the system.
The first step in the Carnot cycle is isothermal expansion, where a gas in a cylinder under pressure from a piston is kept at a constant temperature, but a heat source is brought in contact with the cylinder. To maintain the same temperature, the gas must expand. Next, adiabatic expansion, or expansion without heat transfer to the surrounding environment, removes weight from the piston to allow the gas to expand further, which helps it push on a cylinder to do work. Next, the gas is cooled at a constant temperature and compressed by adding weight to the piston to raise the pressure in the gas, at which point heat transfers from the gas back to the heat source. And finally, adiabatic compression adds more weight to the piston to further increase the pressure of the gas and, therefore, increase its temperature. Then the cycle repeats, according to NASA. This is the basic principle behind heat pumps used for heating, air conditioning, and refrigeration, according to Georgia State University.
Entropy
All thermodynamic systems generate waste heat. This waste results in an increase in entropy, which is a measure of the disorder of a system. Because work comes from ordered molecular motion, entropy is a measure of the energy that's not available to do work, according to Britannica. Entropy in any closed system always increases; it never decreases. Additionally, moving parts produce waste heat due to friction, and radiative heat inevitably leaks from the system.
This makes so-called perpetual motion machines impossible. Siabal Mitra, a professor of physics at Missouri State University, told Live Science "You cannot build an engine that is 100% efficient, which means you cannot build a perpetual motion machine. However, there are a lot of folks out there who still don't believe it, and there are people who are still trying to build perpetual motion machines."
Entropy is also defined as "a measure of the disorder or randomness in a closed system," which also inexorably increases. You can mix hot and cold water, but because a large cup of warm water is more disordered than two smaller cups containing hot and cold water, you can never separate it back into hot and cold without adding energy to the system. Put another way, you can't unscramble an egg or remove cream from your coffee. Entropy, therefore, provides us with an arrow of time: forward is the direction of increasing entropy.
The four laws of thermodynamics
The fundamental principles of thermodynamics were originally expressed in three laws. Later, scientists figured out that a more fundamental law had been neglected, apparently because it had seemed so obvious that it did not need to be stated explicitly. To form a complete set of rules, scientists decided this most fundamental law needed to be included. The problem, though, was that the first three laws had already been established and were well known by their assigned numbers. When faced with the prospect of renumbering the existing laws, which would cause considerable confusion, or placing the pre-eminent law at the end of the list, which would make no logical sense, British physicist Ralph H. Fowler came up with an alternative that solved the dilemma: he called the new law the "Zeroth Law," according to Saint Anselm College. Here are all four laws in brief:
The Zeroth Law of Thermodynamics states that if two bodies are in thermal equilibrium with some third body, then they are also in equilibrium with each other. This establishes temperature as a fundamental and measurable property of matter.
The First Law of Thermodynamics states that the total increase in the energy of a system is equal to the increase in thermal energy plus the work done on the system. This states that heat is a form of energy and is therefore subject to the principle of conservation — that is, it can be neither created nor destroyed.
The Second Law of Thermodynamics states that heat energy cannot be transferred from a body at a lower temperature to a body at a higher temperature without the addition of energy. This is why it costs money to run an air conditioner.
The Third Law of Thermodynamics states that the entropy of a perfect crystal — that is, a substance made up of atoms arranged in a perfectly ordered, symmetrical pattern — at absolute zero is zero. As explained above, entropy is sometimes called "waste energy," or the energy that is unable to do work; and since there is no heat energy whatsoever at absolute zero, there can be no waste energy. Entropy is also a measure of the disorder in a system, and while a perfect crystal is by definition perfectly ordered, any positive value of temperature means there is motion within the crystal, which causes disorder. For these reasons, there can be no physical system with lower entropy, so entropy always has a positive value.
The science of thermodynamics has been developed over centuries, and its principles apply to nearly every device ever invented. Its importance in modern technology cannot be overstated.
Live Science contributor Ashley Hamer updated this article on Jan. 28, 2022.
Additional resources
- The field of thermodynamics has a long history, and this video from CrashCourse provides a tour through its most notable moments and figures.
- Want to see thermodynamics in action? This resource from West Virginia University links to a number of fascinating video examples. Just because thermodynamics is fundamental to science doesn't mean it doesn't have its issues. This article in Physics Today explores why it's been called the "village witch" of physics.
Bibliography
University of Kentucky Department of Physics and Astronomy, "Temperature And Heat — Thermal Energy," https://www.pa.uky.edu/~straley/THE/heat/then4.htm
Carl Nave, "Heat," Hyperphysics, Georgia State University, 2017. http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/heat.html
Carl Nave, "A More General View of Temperature," Hyperphysics, Georgia State University, 2017. http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/temper2.html
American Heritage Dictionary, "Temperature." 2022. https://www.ahdictionary.com/word/search.html?q=temperature
Scottish Science Hall of Fame, "Lord Kelvin (1824-1907)." 2009. https://digital.nls.uk/scientists/biographies/lord-kelvin/
Wolfram Research, "Specific Heat." 2007. https://scienceworld.wolfram.com/physics/SpecificHeat.html
Southeastern Louisiana University, "Thermodynamics Part 1: Work, Heat, Internal Energy and Enthalpy." 2000. https://www2.southeastern.edu/Academics/Faculty/wparkinson/help/thermochemistry/
Debdatta Ratna, "3 - Thermal properties of thermosets," Thermosets: Structure, Properties and Applications, 2012. https://www.sciencedirect.com/science/article/pii/B9780857090867500031
Erik Cheever, "Elements of Thermal Systems," Swarthmore College, 2021. https://lpsa.swarthmore.edu/Systems/Thermal/SysThermalElem.html
Sergey V. Kidalov and Fedor M. Shakhov, "Thermal Conductivity of Diamond Composites," Materials (Basel), December 2009. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5513588/
Paul Peter Urone and Robert Hinrichs, "College Physics." OpenStax, Aug 22, 2016 https://pressbooks.online.ucf.edu/algphysics/chapter/conduction/
Isaac Newton, "VII. Scala graduum caloris," Philosophical Transactions of the Royal Society, 1701.
Worcester Polytechnic Institute, "Newton's Law of Cooling," 1996. http://www.math.wpi.edu/Course_Materials/MA1022A96/lab2/node5.html
Northwestern University, "How does heat move?" https://www.qrg.northwestern.edu/projects/vss/docs/thermal/1-how-does-heat-move.html
Carl Nave, "Carnot Cycle," Hyperphysics, Georgia State University, 2017. http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/carnot.html
NASA, "Carnot Cycle," 2021. https://www.grc.nasa.gov/www/k-12/airplane/carnot.html
Carl Nave, "Heat Pump," Hyperphysics, Georgia State University, 2017. http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/heatpump.html
American Heritage Dictionary, "Entropy," 2022. https://www.ahdictionary.com/word/search.html?q=entropy
Ian T. Durham, "Ralph Fowler," St. Anselm College. 2001. https://mathshistory.st-andrews.ac.uk/Biographies/Fowler/

- Ashley Hamer PritchardLive Science Contributor

 Live Science Plus
Live Science Plus





