Giant viruses spew their DNA through a 'stargate.' Now, scientists know what triggers them.
Scientists combined several techniques to study the massive viruses in action.


Giant viruses, which measure about 10 times the size of a typical cold virus, infiltrate cells and inject their DNA through a special portal known as a "stargate." Now, detailed new images have revealed what conditions prompt this stargate to open and drive the viruses to infect.
Viruses, giant or otherwise, lack the machinery required to make copies of their DNA; the microbes are essentially just a coil of genetic material tucked inside an envelope, called a capsid. In order to survive, viruses must sneak inside a host cell, hijack the machinery within and set up a so-called viral factory to produce new viruses. Giant viruses have a special portal for this job: the stargate.
Shaped like a splayed starfish with five legs, the stargate lies on the surface of a virus and remains sealed during much of its life cycle. But once inside a host cell, each leg of the stargate "unzips", allowing viral genetic material to slip through the resulting hole, said senior author Kristin Parent, an associate professor of Biochemistry and Molecular Biology at Michigan State University. The new study, published May 8 in the journal Cell, demonstrates that the stargate opens in response to acidic, salty and high-temperature environmental conditions.
Related: Going viral: 6 new findings about viruses
When the stargate opens, other proteins sneak out along with the viral DNA, Parent told Live Science. "We can identify which [proteins] are actually coming out of the capsid during that opening event," she said. Parent and her team plan to probe how these proteins work and what roles they play in giant virus infection, in order to better understand these massive and mysterious microbes.
Big and mysterious
Scientists discovered giant viruses relatively recently; the first study describing them was published in 2003, Science magazine reported.
In that report, the enormous microbes were isolated from water samples collected in the early 1990s, before scientists could closely examine the viruses' genetic code, Parent said. At the time of collection, researchers assumed that the microbes must be bacteria, due to their great size. Giant viruses measure more than 0.00001 inches (300 nanometers) in diameter, or about 10 times larger than your average rhinovirus, which can cause the common cold, according to a statement.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Mistaken for big bacteria, giant viruses were essentially "discovered ten years before [scientists] knew what they were looking at," Parent said. Once the technology became available, researchers dug up the samples and found that the large microbes lacked ribosomal RNA — a key molecule that allows bacteria to build their own proteins, which viruses cannot do on their own.
Since their initial discovery, giant viruses have been recovered from melting permafrost in Siberia, the depths of the Antarctic ocean and highly alkaline soda lakes, as well as less exotic environments, said Chantal Abergel, research director of the Structural and Genomic Information laboratory at the French National Centre for Scientific Research, who was not involved in the study. The viruses have mostly been found to infect amoebas and phytoplankton, but laboratory studies suggest that they can also infect animal cells, including rodent and human cells. However, "no direct link between GVs and human disease has yet been established," the authors noted.
Related: The 12 deadliest viruses on Earth
"You can find them in all kinds of environments," Abergel said. Most likely, the viruses went overlooked for decades because scientists filtered out large particles from their water samples when searching for smaller viruses, Abergel added. And besides being big, giant viruses boast many unique features that set them apart from any smaller virus seen before, she added.
For example, Parent and her coauthors studied several giant viruses that look like 20-sided dice, including the mimivirus, Antarctica virus, Samba virus and Tupanvirus. The structure and outer shells of these viruses "are very complex, as never seen before in the virosphere," study author Jônatas Abrahão, an associate professor of virology at the Universidade Federal de Minas Gerais in Brazil, told Live Science in an email. The stargate found on the surfaces of these viruses particularly fascinates scientists "due its beauty and symmetry," and the fact that no smaller viruses contain such a structure, Abrahão said.
But until now, "how to open the stargate was a mystery," he added.
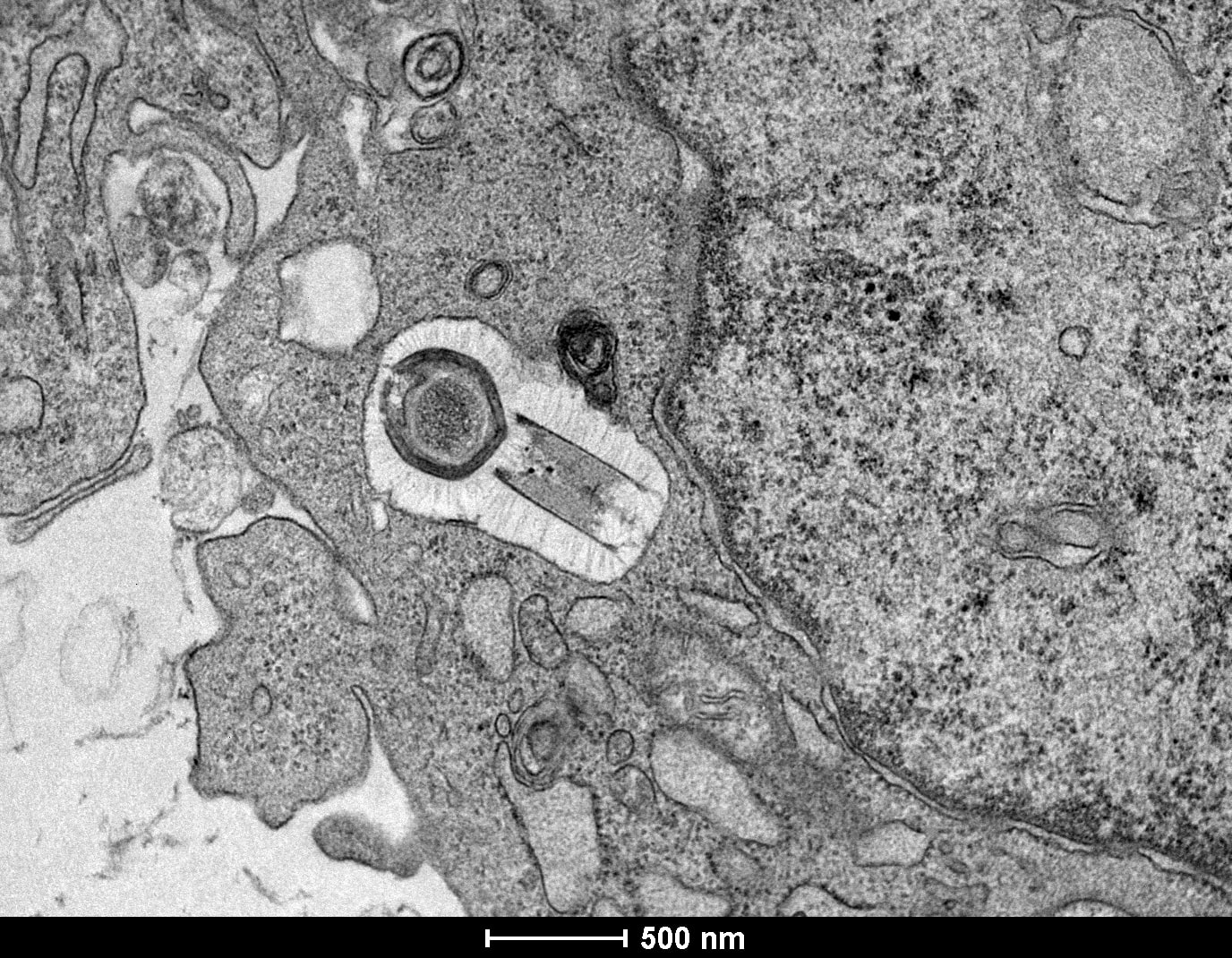
An image of of a Tupanvirus particle (dark round shape in the center) infecting an amoeba
Opening the stargate
Before the stargate opens, giant viruses get "gobbled up by cells" in a process called phagocytosis, Parent said. While small viruses like influenza fuse their fatty membrane with that of the cell they're infecting, giant viruses enter cells by being swallowed whole, hard outer shell and all.
Once inside, the giant viruses open their stargates and release their infectious "seed," Abergel said. "This is an entire structure that is downloaded from the plasmid," or circular ring of viral DNA, into the cytoplasm, or the water solution of proteins and salts that surrounds the organelles in eukaryotic cells. This process jumpstarts infection, she said.
In previous studies, researchers captured snippets of this process by cutting infected cells into thin sections and studying what could be found inside, Parent said. But unless someone happens to capture that "one-in-a-million" snapshot of the stargate opening, the slice-and-shoot technique makes it difficult to discern one stage of infection from the next, she added.
To overcome this obstacle, Parent and Jason Schrad, a graduate student in her lab, devised a strategy to trigger giant virus infection outside of a cell and image each step of the process.
After isolating the viruses, the team subjected each sample to different chemical and environmental treatments in an attempt to mimic the conditions that might trigger infection inside an actual cell. For example, after being gobbled by a cell, the viruses sit in a membrane-bound bubble called a vacuole, which tends to be very acidic (low pH). The team then placed the treated viruses under a cryo-electron microscopy (EM) microscope, which transmits beams of negatively charged particles through the sample to capture atomic-resolution images. They also scanned the samples with a scanning electron microscope to take detailed images of the viral surface.
The team found that three conditions reliably caused the stargate to unzip: low pH, high salt concentrations and high temperatures, up to 212 degrees Fahrenheit (100 degrees Celsius).
Low pH or high salt, in isolation, "cracked" open the stargate but did not open the structure completely, the authors noted. The addition of extreme heat opened the portal more, but, it's unlikely that boiling temperatures could be found in a real cell, Parent said. More likely, high temperature mimics the effects of something else that happens "in the context of a host," such as the presence of a specific enzyme, she said.
Related: Magnificent microphotography: 50 tiny wonders
Under many conditions, giant viruses resist opening their stargates, "and yet, as soon as you have the proper key, they open up very nicely," similar to a tough seed breaking open in response to water, Abergel said. In previous work in her lab, Abergel predicted that low pH and high salt concentrations might cause the stargate to open. "It's very good to see the model demonstrated experimentally," she said of the Cell study.
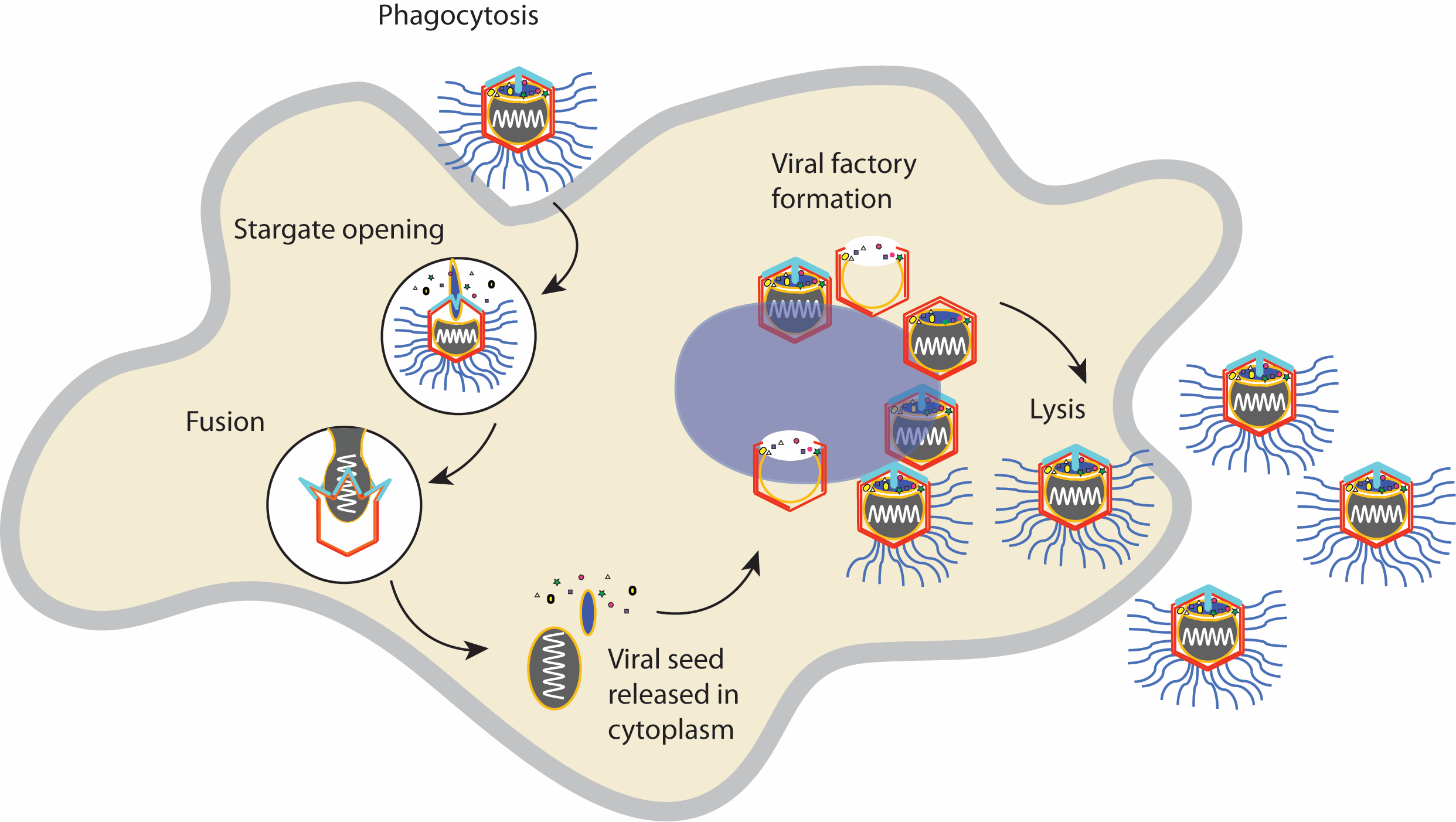
Cartoon of a Samba virus infection a cell, starting with phagocytosis and ending with new viruses escaping the cell.
Still more mysteries to solve
After coaxing the stargate to open, the team went on to study what proteins slipped out of the virus along with its genetic material. Focusing on the Samba virus and Tupanvirus, specifically, they used a technique called mass spectrometry to deduce the structure of different proteins.
"The shape of the viral proteins and the way that they work tend to be conserved across very distantly related [viruses]," Parent said. In future studies, the authors aim to determine how these giant virus proteins function, in part, by "[chopping] up the protein sequences into little pieces" and comparing their structure to other known proteins.
"The proteins released during [giant virus] uncoating are likely related to initial steps of infection, including shutdown of host response and viral factory assembly," Abrahão said. "However, many viral proteins released from the capsid are unknown and their functions remain to be investigated."
The 20-sided viruses included in the study represent just one class of giant virus, but the same research methods could be used to study many other species, "with genomes and particles never described before," Abrahão said. Giant viruses contain genes and proteins that "do not resemble anything in the cellular world," or even other viruses, Abergel added. Future studies could reveal when and how these viruses first evolved, and why they differ so drastically from more familiar forms of life.
- 6 superbugs to watch out for
- 5 ways gut bacteria affect your health
- Genetics by the numbers: 10 tantalizing tales
Originally published on Live Science.
OFFER: Save 45% on 'How It Works' 'All About Space' and 'All About History'!
For a limited time, you can take out a digital subscription to any of our best-selling science magazines for just $2.38 per month, or 45% off the standard price for the first three months.

Nicoletta Lanese is the health channel editor at Live Science and was previously a news editor and staff writer at the site. She is a recipient of the 2026 AHCJ International Health Study Fellowship, with a project focused on antibiotic stewardship practices in Japan and the U.S. They hold a graduate certificate in science communication from UC Santa Cruz and degrees in neuroscience and dance from the University of Florida. Beyond Live Science, Lanese's work has appeared in The Scientist, Science News, the Mercury News, Mongabay and Stanford Medicine Magazine, among other outlets. Based in NYC, she also remains involved in dance and performs in local choreographers' work.

 Live Science Plus
Live Science Plus





