Pac-Man-shaped blobs become world's first self-replicating biological robots
These bio-bots are made from frog cells.

Tiny groups of cells shaped like Pac-Man are the world's first self-replicating biological robots.
The tiny bots are made from the skin cells of frogs, but they don't reproduce by mitosis or meiosis or any of the other ways cells divide and replicate in normal circumstances. Instead, they build more of themselves from raw materials — free-floating frog skin cells — creating multiple generations of nearly identical organisms.
In action, the bots (dubbed "xenobots" by their inventors), even look like Pac-Man. They move in wild corkscrews and spirals, their open "mouths" scooping the free-floating skin cells into piles. The cells tend to adhere, or stick together, once put in contact with one another, so these piles gradually meld into new, spiraling xenobots.
Though this self-replication is a fairly delicate process, so far possible only in a carefully controlled lab dish, researchers hope it offers new promise for biologically based robots.
Related: 11 body parts grown in the lab
"The ability to make a copy of yourself is the ultimate way to make sure you keep doing whatever it is you do," said Sam Kriegman, a computer scientist and postdoctoral scholar at the Wyss Institute at Harvard University and the Allen Discovery Center at Tufts University.
Bio-bots
Kriegman and his colleagues, including computer scientist Joshua Bongard of the University of Vermont, have been developing the xenobots for years. The bots are made from stem cells taken from frog eggs and are 0.04 inches (1 millimeter) wide or less. When put in contact with each other, the stem cells naturally form spherical blobs covered with tiny, beating cilia, or hairlike structures that can propel the blobs around.
Get the world’s most fascinating discoveries delivered straight to your inbox.
"They're neither a traditional robot nor a known species of animal," Bongard said in a statement when the invention of xenobots was first announced in 2020, Live Science reported at the time. "It's a new class of artifact: a living, programmable organism."
Programming an organism isn't as easy as entering commands into code, though, Kriegman told Live Science. "It's difficult to program something that doesn't have software," he said.
Ultimately, control of the xenobots comes down to control of their shapes. That's where artificial intelligence comes into play. It's not always intuitive what a xenobot will do when you alter its shape, or how to get a desired outcome by sculpting the shape. But computer simulations can run through billions of shape and size options in days or weeks. Researchers can even vary the environment around the simulated xenobots. Promising shapes, sizes and environments can then be tested in the real world.
Biological robots are promising, Kriegman said, because they can self-repair. They're also biodegradable. Left to their own devices, the xenobots run out of energy and begin to degrade within 10 to 14 days. They don't leave microplastics or toxic metals behind, just tiny specks of organic decay. The researchers are working on designs that might allow the xenobots to carry small amounts of material. Potential uses include delivering drugs inside the body or cleaning up toxic chemicals in the environment.
Self-replication
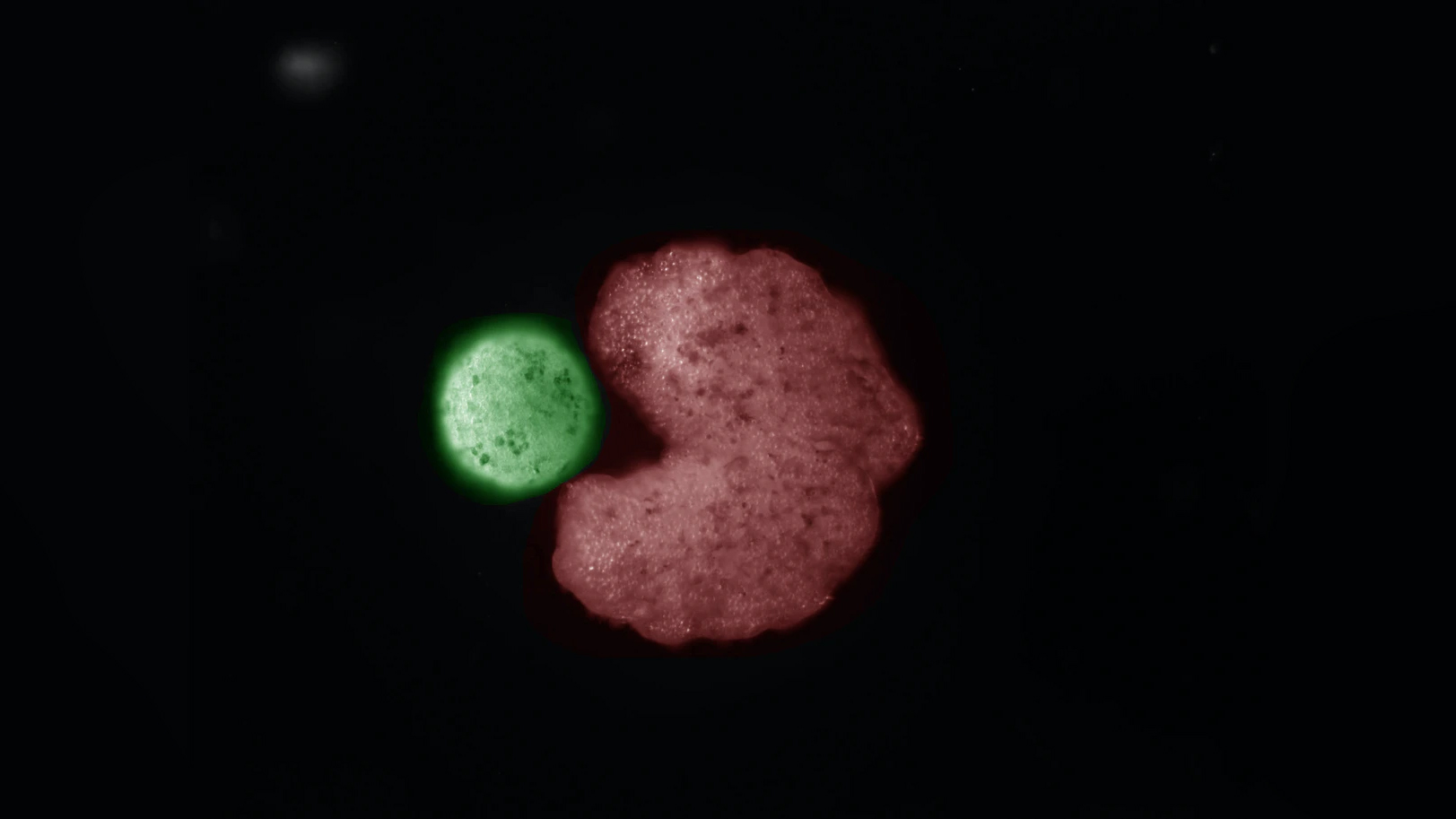
In their typical spherical shape, the xenobots are capable of a limited version of self-replication, the researchers found. When put in a dish full of independently floating frog stem cells, the blobs circle merrily, randomly pushing the free-floating cells into clumps, some of which stick together to form new xenobots. These tend to be smaller than their parents, however, and typically they aren't capable of moving around enough single cells to create yet another generation.
After computer simulations suggested that a Pac-Man shape might be more effective, the researchers tested these C-shaped xenobots in a soup of stem cells. They found that the diameter of the offspring of Pac-Man xenobots was 149% larger than the offspring of spherical xenobots. Thanks to the size improvements, the baby xenobots were able to create their own offspring. Instead of just one generation of xenobot replication, the researchers found they were able to reach three or four.
The system is still quite fragile, and the process of growing the cells and making sure their growth substrate is clean and fresh is tedious, Kriegman said. And not to worry, as there's no concern that these biological robots will replicate out of control and take over the world: "If you sneeze on the dish, you'll ruin the experiment," Kriegman said.
That also means the xenobots aren't quite ready to become working robots. The researchers are still working on testing different shapes for different tasks. Their AI simulation also suggested that varying the shape of the lab dishes the xenobots replicate in might lead to better results, but that still needs to be tested in the real world.
However, there are lessons from the xenobots that could be incorporated into robotics right away, Kriegman said. One is that artificial intelligence can be used to design robots, even robots that can self-replicate. Another is that it makes sense to create robots from intelligent components. Biological organisms are smart all the way down to their component parts, he said: Organisms are made of self-organizing cells, which are made of self-organizing organelles, which are made of self-assembling proteins and molecules. Current metal-and-plastic robots don't work in that way.
"If we could build robots out of intelligent modules, maybe we could create more robust machines," Kriegman said. "Maybe we could create robots in the real world that could self-repair or self-replicate."
Originally published on Live Science.

Stephanie Pappas is a contributing writer for Live Science, covering topics ranging from geoscience to archaeology to the human brain and behavior. She was previously a senior writer for Live Science but is now a freelancer based in Denver, Colorado, and regularly contributes to Scientific American and The Monitor, the monthly magazine of the American Psychological Association. Stephanie received a bachelor's degree in psychology from the University of South Carolina and a graduate certificate in science communication from the University of California, Santa Cruz.