Electrical zaps can 'reawaken' lost neural connections, helping paralyzed people walk again
Scientists identified specific spinal nerve cells that people likely need to regain the ability to walk after paralyzing injuries.

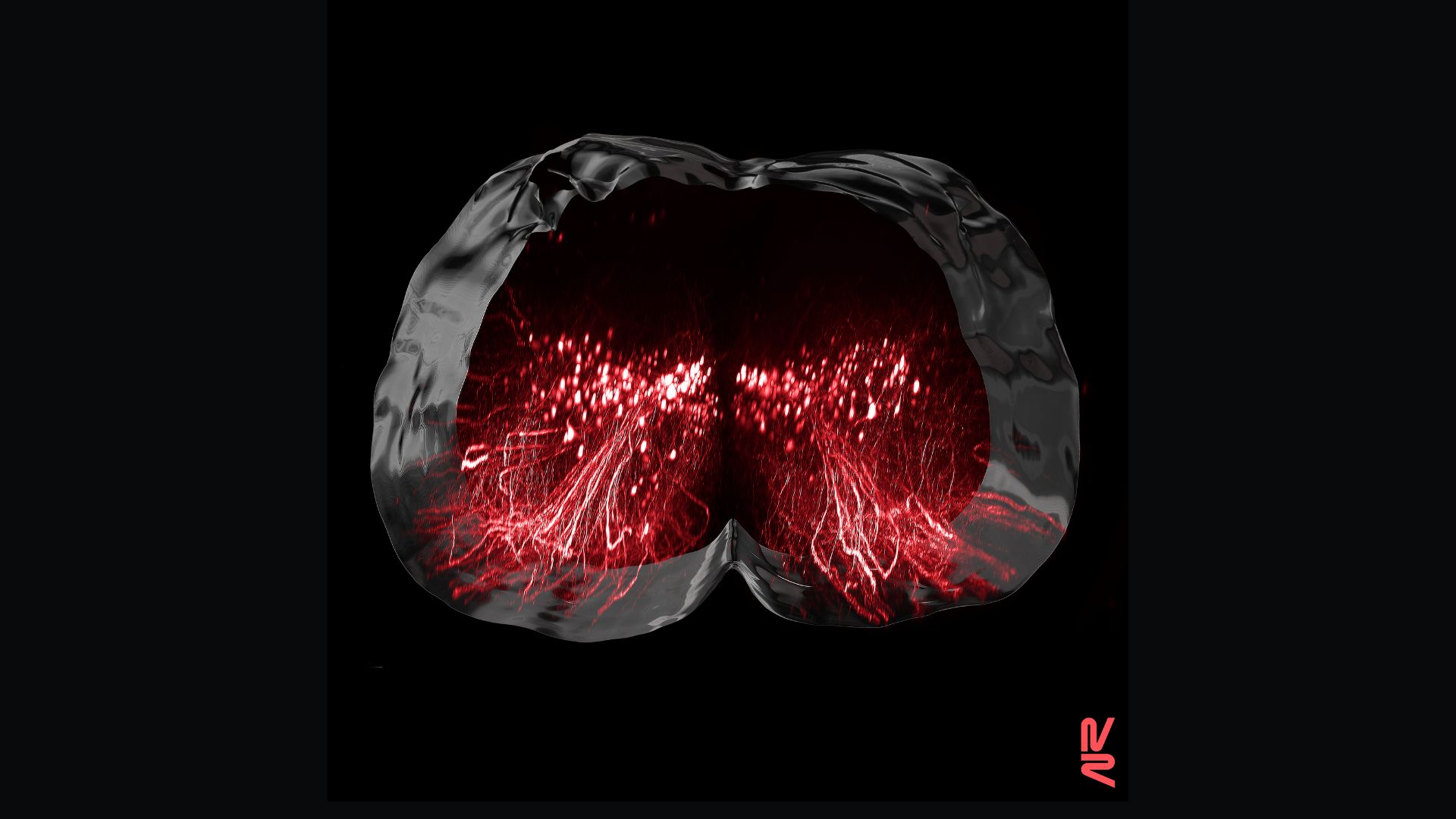
People with paralyzing spinal cord injuries can walk again with the help of medical devices that zap their nerves with electricity. But the designers of these new implants weren't completely sure of how they restored motor function over time — now, a new study provides clues.
The new study of humans and lab mice, published Nov. 9 in the journal Nature, pinpoints a specific population of nerve cells that seems key to recovering the ability to walk after a paralyzing spinal cord injury. With a jolt of electricity, an implant can switch these neurons on and thus jumpstart a cascade of events in which the very architecture of the nervous system changes. This cellular remodel restores the lost lines of communication between the brain and the muscles needed for walking, allowing once-paralyzed people to walk again, the researchers concluded.
Understanding how the nerve-zapping system, called epidural electrical stimulation (EES), "reshapes spinal circuits could help researchers to develop targeted techniques to restore walking, and potentially enable the recovery of more-complex movements," Eiman Azim, a principal investigator at the Salk Institute for Biological Studies in La Jolla, California, and Kee Wui Huang, a postdoctoral fellow in Azim's lab, wrote in a commentary.
Nine people with paralyzing spinal cord injuries participated in the new study. Six were mostly or completely unable to move their legs but retained some feeling in the limbs; the other three participants had no motor control or sensation from the waist down.
Related: The five (and more) human senses
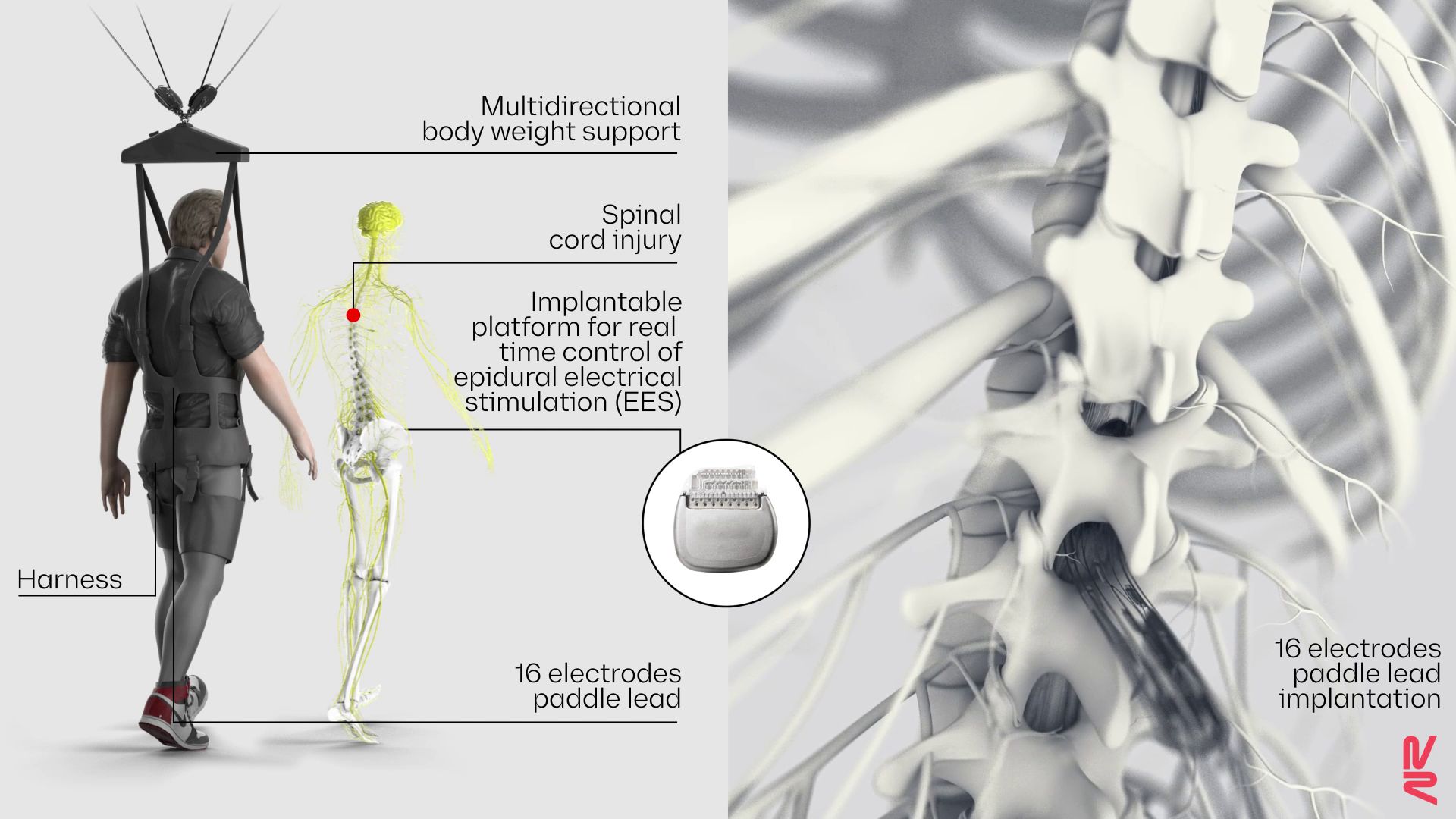
The implant delivers electrical stimulation to the nerves of the lower spinal cord.
The nine participants underwent surgery to have electrodes implanted atop their lower spinal cords, below the muscle and bone but outside the membrane that encases the nervous system. Each participant then trained with their implant for five months. They started out by practicing standing, walking and performing various exercises indoors in a weight-bearing harness, and they eventually graduated to training outdoors with a walker for stability.
These exercises were completed with the EES implant switched on, but in time, four of the nine participants could bear weight and walk with the device switched off, the researchers wrote in their report.
Get the world’s most fascinating discoveries delivered straight to your inbox.
The team also found that, as each participant regained their ability to walk, the overall activity of their spinal cords decreased in response to the EES — what initially looked like a roaring fire of nerve cell activation dwindled down to a smolder. This hinted that the combination of rehab and electrical stimulation was reorganizing the nervous system, such that fewer and fewer cells were needed to complete the same action.
RELATED STORIES
"When you think about it, it should not be a surprise because in the brain, when you learn a task, that’s exactly what you see — there are less and less neurons activated" as you improve, co-senior author Grégoire Courtine, a neuroscientist and professor at the Swiss Federal Institute of Technology, Lausanne (EPFL), told Nature.
The team used rodent-size EES implants to study how this reorganization unfolds in mice with paralyzing spinal cord injuries. The mice completed a course of rehabilitation, similar to the human participants, and throughout, the researchers tracked which of their nerve cells responded to the treatment by changing which genes they had switched on.
This analysis revealed a set of neurons in the lumbar spinal cord that consistently responded to the therapy, even as other neurons became less active. Blocking the activity of these neurons in uninjured mice didn't affect their ability to walk, but in injured mice with paralysis, silencing the cells prevented them from walking again. This suggests that, although other nerve cells might play their own roles in recovery, this particular group is especially important, Courtine told Science.
"The findings are consistent with the idea that certain types of spinal neuron[s] that have lost their inputs from the brain after injury can be 'reawakened' or repurposed to restore movement if they are given the appropriate combination of stimulation and rehabilitation," Azim and Huang wrote. Assuming the findings from the mouse studies carry over to humans, the experiments could lay the groundwork for new-and-improved devices aimed at repairing the spinal cord after injury, they said.

Nicoletta Lanese is the health channel editor at Live Science and was previously a news editor and staff writer at the site. She is a recipient of the 2026 AHCJ International Health Study Fellowship, with a project focused on antibiotic stewardship practices in Japan and the U.S. They hold a graduate certificate in science communication from UC Santa Cruz and degrees in neuroscience and dance from the University of Florida. Beyond Live Science, Lanese's work has appeared in The Scientist, Science News, the Mercury News, Mongabay and Stanford Medicine Magazine, among other outlets. Based in NYC, she also remains involved in dance and performs in local choreographers' work.