Celiac disease: Causes, symptoms and treatments
Celiac disease causes severe digestive symptoms and results from an immune reaction to dietary gluten.


Celiac disease, also called celiac sprue, is a condition that affects mostly the small intestine, although it can have consequences throughout the body. People with celiac disease experience digestive symptoms and potential long-term tissue damage as a result of the immune system attacking the inner lining of the small intestine.
In nearly all cases, this immune reaction is triggered by the ingestion of gluten, a group of proteins present in grains such as wheat, rye and barley.
Related: What is gluten?
What causes celiac disease?
Celiac disease occurs mostly in people who have a genetic predisposition to the condition, due to abnormalities in the human leukocyte antigen (HLA) genes that are located on chromosome 6. These genes code for HLA proteins, whose function is to bind bits of infectious pathogens, like viruses, and alert the immune system to the invaders' presence. Mutations in these genes can cause the body to mistake its own tissues as a threat and attack them.
Two HLA gene abnormalities, called HLA-DQ2 and HLA-DQ8, are linked to celiac disease, such that the presence of one or both of them makes a person genetically predisposed to the disease. These individuals experience a hypersensitive reaction to the presence of gluten in the gastrointestinal tract, meaning their immune systems respond inappropriately and excessively to the substance.
In celiac disease, the hypersensitivity is classified as a type IV hypersensitivity reaction, or delayed hypersensitivity. Type IV hypersensitivity is mediated by the interaction of immune cells called T lymphocytes, monocytes and macrophages, which, in turn, trigger additional immunological events. In celiac disease, this immunological cascade occurs in the small intestine and changes the histology (the microscopic anatomy) of the inner lining of the organ, leading to symptoms and complications.
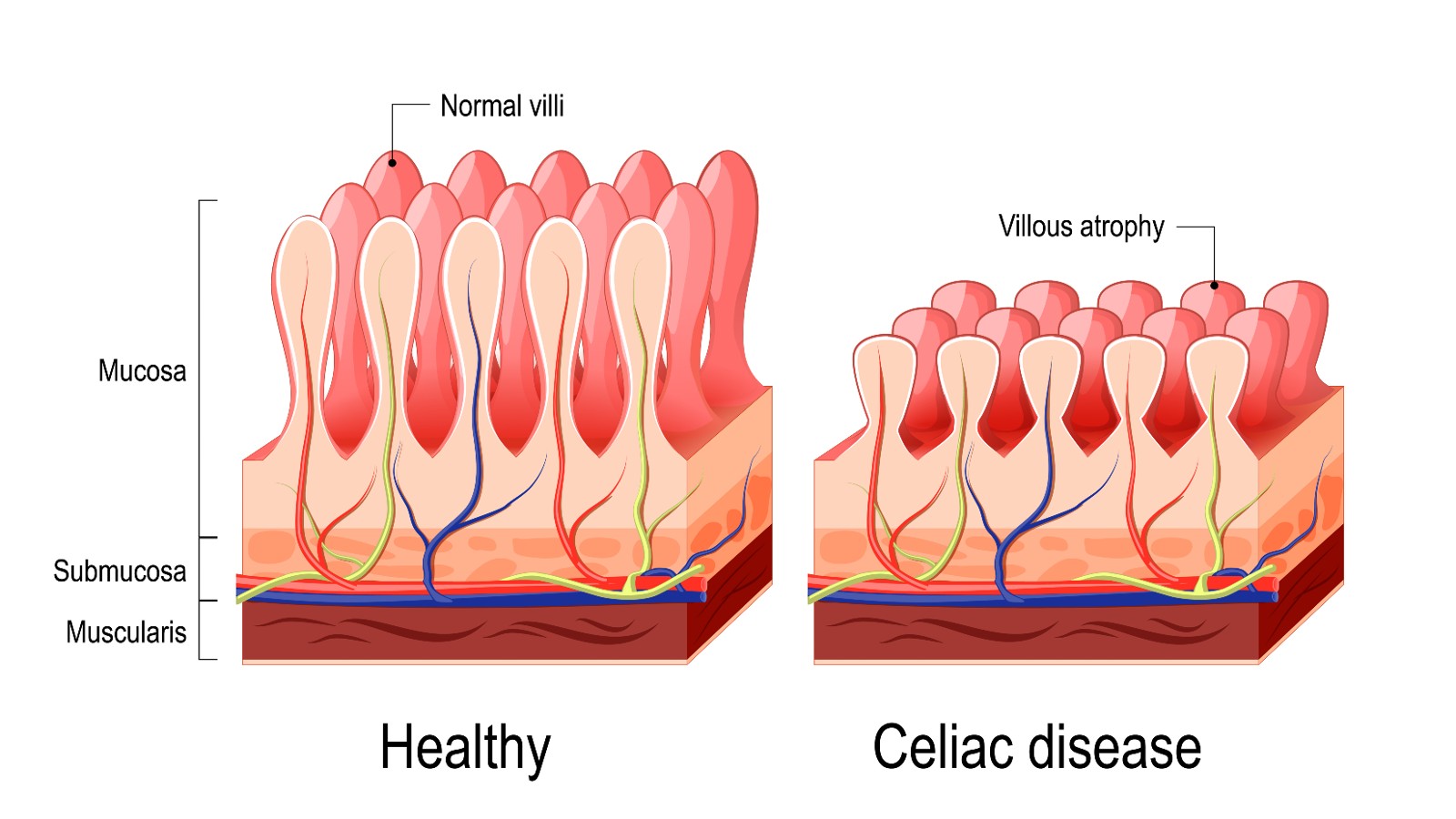
The inside of the small intestine is lined with tiny hair-like projections called villi. This diagram compares healthy villi compared with those that have atrophied, or eroded away, due to celiac disease.
Typically, these changes in the small intestine reverse, and the symptoms abate, when gluten is removed completely from the diet. But in rare cases, known as refractory celiac disease, the disease process can continue even in the absence of gluten, due to the celiac immune process going into a kind of autopilot.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Meanwhile, the disease process in the small intestine leads to the malabsorption of food and the release of mucus, and often blood, into the gastrointestinal tract. All of this produces diarrhea, gas and bloating, nausea and, over the long term, malnutrition.
Celiac disease risk factors
The prevalence of celiac disease depends greatly on a genetic predisposition involving the HLA-DQ2 and HLA-DQ8 genes, but there is notable variation among countries and ethnic groups.
In North America, celiac disease affects about 0.71% of people, or about 1 in 141. The disease is a little more common in white people, with 1 case per every 100 individuals. In Europe, about 1 in 100 people has celiac disease, but the prevalence is higher in certain countries, notably Finland, where 2.4% of people have the disease.
Celiac disease is also about as common in India, North Africa and the Middle East as it is in Europe.
While the aforementioned genetic factors constitute risk factors, people are not often tested for the presence of the HLA-DQ2 and HLA-DQ8 gene variants. Thus, practically speaking, the main risk factor for celiac disease is having a first-degree relative (parent, sibling, child) who is known to have the condition. Additionally, type 1 diabetes has been found to be a potential risk factor for celiac disease.
What are the symptoms of celiac disease?

Abdominal pain is one symptom of celiac disease.
People with celiac disease experience a constellation of symptoms, including the following:
- Diarrhea
- Abdominal pain
- Nausea and vomiting
- Bloating and gas
- Constipation
- Fatigue
- Weight loss
- Constipation
In people diagnosed with celiac disease, both the short- and long-term symptoms tend to be very severe. However, it is not uncommon for people with a family history of celiac disease to experience any of the above symptoms at milder levels, even if diagnostic tests, including serology tests, do not show indications of celiac disease.
Abatement of the above symptoms following the withdrawal of gluten from the diet can support a diagnosis of celiac disease, but such a diagnosis cannot be made based solely on how a person responds to a gluten-free diet. One major reason for this is that foods that lack gluten also lack various other components that could potentially cause gastrointestinal distress. Such components include fiber, as well as fermentable oligosaccharides, disaccharides and monosaccharides and polyols (FODMAPs), which will be discussed later in this article.
How is celiac disease diagnosed?
Diagnosing celiac disease can be complex, but it should begin with a thorough patient history and physical examination. A combination of any of the following suggests celiac disease:
- A long history of diarrhea
- Tummy pain
- Bloating
- Sores in the mouth
- Weight loss
- Bleeding of the GI tract, due to immune attack in the lining of the small intestine
- Bruising and bleeding outside the GI tract, due to nutritional deficiencies
- Bone fractures, which are made more likely by malnutrition
In many cases, celiac disease can be difficult to confirm, because other serious gastrointestinal conditions, such as inflammatory bowel disease and ulcers, cause similar symptoms. These other diseases can be ruled out through additional testing, which can include the checking of stool samples for signs of blood, as well as imaging tests.
If a patient does not suffer from malnutrition, doctors should consider that the person's gastrointestinal problems might be the result of a very common condition called irritable bowel syndrome, which frequently results from intolerance to one or more FODMAPs. One particular FODMAP to which people are often intolerant is lactose, the kind of sugar that's in milk, but there are other types of FODMAP intolerance that cause dietary distress following the ingestion of other things, such as beans and grains.
Although dietary fiber is healthy because it helps move food and waste through the gastrointestinal tract and can have beneficial effects on cholesterol levels in the blood, eating a lot of high-fiber foods also can also produce gas and gastrointestinal distress.
A family history of celiac disease in one or more close relatives points to the possible presence of celiac disease. After a doctor takes this history, the next step is to perform "serology testing," meaning doctors will screen a patient's blood samples for various antibodies. Classically, serology testing centered on what doctors call anti-gliadin antibodies (AGA), which react to gliadins, a component of gluten. These antibodies come in two flavors: IgA and IgG.
One way to check for celiac disease is to screen a patient's blood for various antibodies.
If a patient tests positive on one or more of these tests, experiences gastrointestinal distress, and has a family history of celiac, doctors may diagnose the disease without further tests.
In many cases, however, the diagnosis will not be made until there is also proof of abnormal histology in the lining of the small intestine. This requires a test called endoscopy, in which a gastroenterologist uses an instrument to look at the inside of the small intestine from the inside and to obtain a sample called a biopsy. Positive serology plus a positive biopsy confirms a celiac disease diagnosis.
Sometimes people will be referred for an endoscopy even if their serology testing is negative. In cases when the serology is negative and yet the endoscopy demonstrates histology resembling that of celiac disease, such patients will be diagnosed with a different intestinal disorder.
While genetic testing can reveal the HLA-DQ2 and HLA-DQ8 gene variants, whose presence correlates extremely well with celiac disease, in practice, celiac diagnosis is usually made on the basis of serology and endoscopic biopsy. On the other hand, because nearly everybody with celiac disease has one or both of the aforementioned gene variants, doctors often use genetic testing to rule out celiac disease. For example, in pediatrics, when a sibling or parent of a child is found to have celiac disease, that child might be offered genetic testing. If it comes out negative, the child can avoid going through the usual celiac workup.
Complications of celiac disease
If not recognized and treated, celiac disease can lead to severe malnutrition due to an inability of the small intestine to absorb nutrients. In particular, this can include deficiencies of micronutrients, such as iron, magnesium, vitamin B12, folic acid, vitamin D, zinc, calcium, niacin and riboflavin.
Deficiency of calcium and vitamin D can lead to osteopenia and osteoporosis, in which bone loses density and becomes more susceptible to fractures. Iron deficiency leads to iron deficiency anemia, while B12 deficiency and folate deficiency each lead to another type of anemia called megaloblastic anemia. B12 deficiency also causes very serious neurological problems. Celiac disease also may raise the risk of other medical conditions, including lymphoma and fertility problems.
Additionally, changes to the intestinal lining in celiac disease can cause lactose intolerance. As noted above, this is an inability to digest milk sugar (lactose). Since lactose intolerance is very common outside of celiac disease, this particular complication can complicate the diagnosis of celiac disease.
Treatment of celiac disease

Gluten-free flours can be made from chickpeas, rice, quinoa, almonds, corn, oatmeal and buckwheat (which, despite its name, isn't a wheat product and doesn't contain gluten).
When celiac disease is diagnosed, the first treatment attempted is a gluten-free diet. This diet must include fiber as well as certain micronutrients, such as folic acid and other B vitamins, which are present in whole grains. If implemented correctly, a gluten-free diet works in almost all cases, but the caveat is that such a diet is not always easy to implement.
Studies have revealed that it is not uncommon for people on gluten-free diets to still unwittingly ingest some amount of gluten. Occasionally, this results from patients being ill-informed about dietary sources of gluten, but there may be a societal factor at play, particularly when it comes to cross-contamination in restaurants. One 2019 study suggested that approximately one-third of food items designated as gluten-free on restaurant menus actually contained substantial quantities of gluten.
Related stories
Given this reality, in cases when patients don't improve on a gluten-free diet, they may have to work with a dietitian to confirm that their diet is truly gluten-free before receiving a diagnosis of refractory celiac disease. It's also important for doctors to check that the patient doesn't have another gastrointestinal condition alongside celiac disease, as this could also explain the lack of response to a gluten-free diet.
As noted above, there are some rare cases in which celiac disease does not resolve after a switch to a gluten-free diet. The reasons for this are not fully understood but may involve the immune system continuing to attack the small intestinal lining in the absence of gluten. Alternatively, the disease may have progressed to a point at which intestinal damage is not reversible.
To treat these tough cases, scientists are working on new treatments, including regimens that may involve gradual, controlled exposure to progressively larger amounts of gluten-like protein. This treatment is aimed at deconditioning the immune system from its harmful response.
This article is for informational purposes only and is not meant to offer medical advice.

David Warmflash is a medical researcher, astrobiologist, science communicator, and author, located in Portland, Oregon. He holds an MD from Tel Aviv University Sackler School of Medicine and has conducted research in astrobiology, space biology, and space medicine during research fellowships at NASA's Johnson Space Center, the University of Pennsylvania, and Brandeis University, and in collaboration with The Planetary Society and the Israeli Aerospace Medicine Institute.

 Live Science Plus
Live Science Plus





