3D scans reveal that beetles have secret pockets on their backs
Here's how the bacteria stay with the beetle through its entire life cycle.

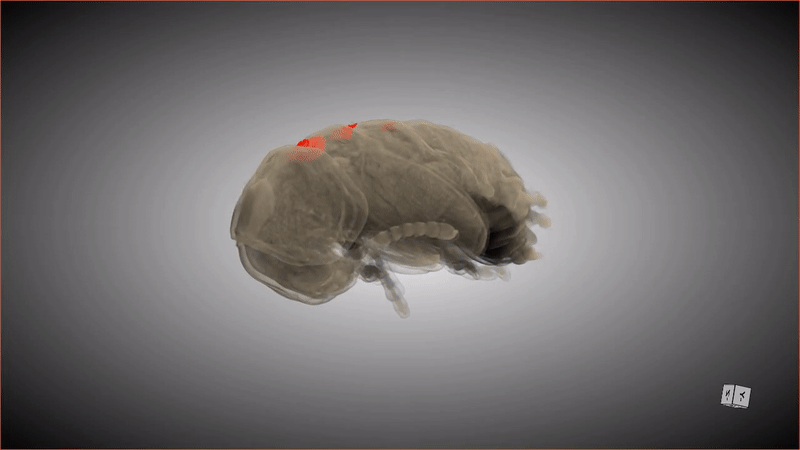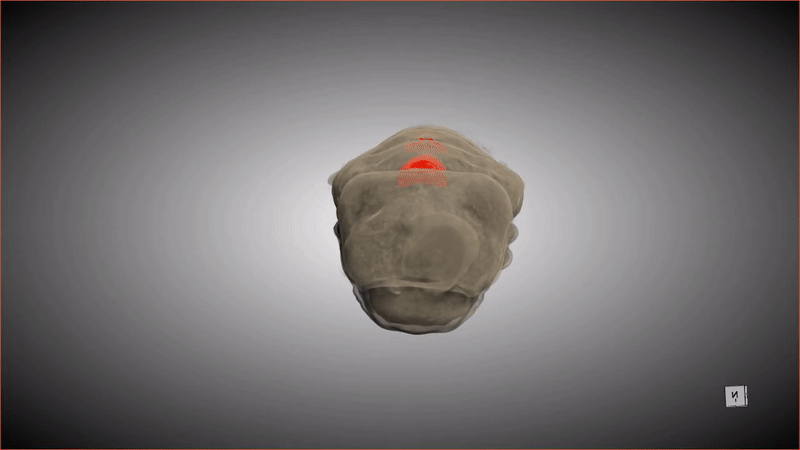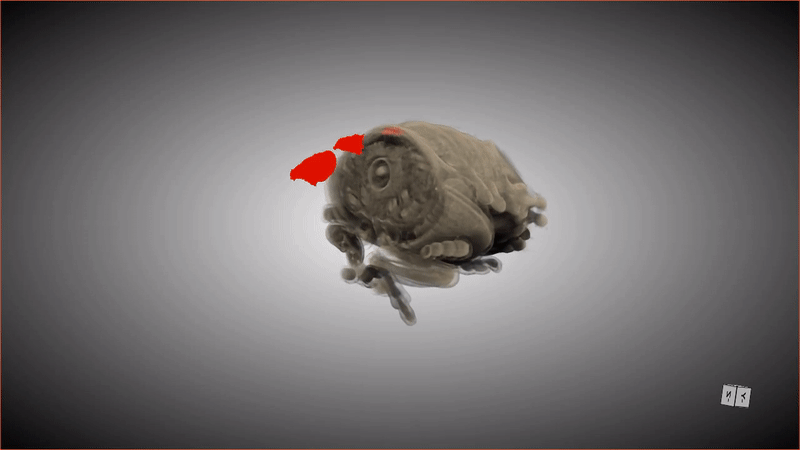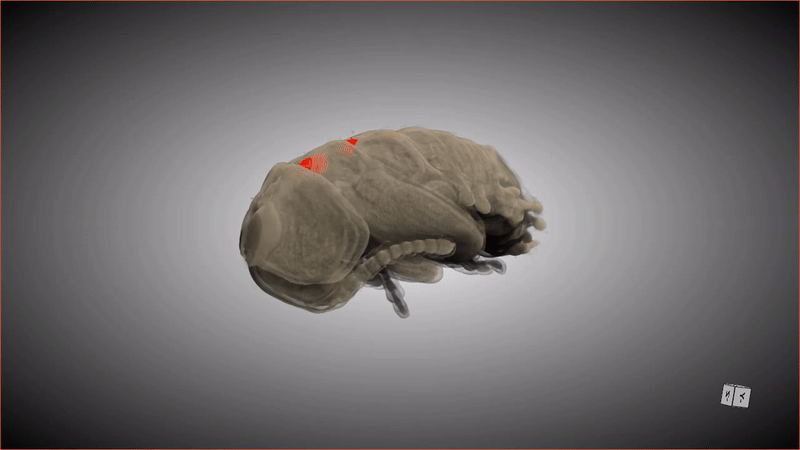
If you were to compliment a female Lagria beetle on her pupa shell, she'd probably respond, "Thanks, it has pockets!"
These special pockets hold an important treasure: symbiotic bacteria that keep the pupa (and the larva that precedes this stage) safe from potentially deadly fungus. When the adult beetle emerges from the pupa, friction from the metamorphosis shoves the bacteria from these pockets into glands in the beetle's abdomen.
Although scientists have known about the pockets and the bacteria (primarily a strain of bacteria called Burkholderia) that populate the abdomen glands of beetles in the genus Lagria, it was unknown how the two were connected, or how the bacteria ended up in the beetles' abdomens in the first place. Recently, a group of researchers revealed the missing steps of this symbiotic relationship by observing what happened when an adult beetle emerged from its pupa, scanning the pupa and creating digital 3D models. The new research was published Tuesday (Aug. 30) in the journal Frontiers in Physiology.
"Fungi are really the major natural enemies of most insects, so they are a threat, especially if [the insects] live in leaf litter" like Lagria beetles do, study co-author Laura Victoria Flórez Patino, a biologist and postdoctoral researcher at the University of Copenhagen in Denmark, told Live Science. So it's important to understand how these beetles can hold on to the protective bacteria throughout the insects' entire life cycle, according to the study.
Related: This beetle can walk upside down on the underside of a pool of water (video)

The Lagria genus contains around 800 beetle species that are distributed all over the world. Only adult female beetles carry symbiotic bacteria, which secrete chemicals that are toxic to fungus. In adult beetles, the bacteria live inside special glands near the insects' oviducts, where eggs emerge. As the beetle lays eggs, the bacteria are deposited onto the egg surfaces; the bacteria transfer to their new beetle hosts when the eggs hatch and remain with them through adulthood.
But Lagria beetles experience some extreme changes in their lives. They emerge from their eggs as wormlike larvae and then grow into immobile, egglike pupae. Inside the pupae, the beetle larvae "get rid of a lot of what they have inside [their bodies] and rebuild," Flórez said. At the end of the pupal stage, adult beetles emerge fully formed.
Get the world’s most fascinating discoveries delivered straight to your inbox.
So how do the bacteria stay with the beetle through these major life changes?
To tackle this question, researchers covered newly pupated beetles with tiny, fluorescent plastic beads measuring just 1 micron thick. (For comparison, a human hair has a thickness of 17 to 180 microns.) After the beetles emerged from their pupae, all of the beads — stand-ins for symbiotic bacteria — collected at the tip of the beetles' abdomens. The researchers suspected that friction from a beetle rubbing against the pupa shell as it climbed out pushed the bacteria toward the abdomen. Once the bacteria were in their new home on the beetle bodies, they were ready to be deposited onto the next generation of Lagria beetles.
Related stories
But where did these helpful bacteria come from? When you think of symbiotic bacteria, the bacteria in your gut, or in the digestive systems of other animals, might come to mind. Flórez suggested that Lagria beetles evolved an external symbiotic relationship with these bacteria because of all the changes that happen inside the pupa. "If you are a symbiont that's inside and highly integrated, you might be lost" as the larval beetle breaks down and rebuilds its entire body, Flórez explained.
Lagria beetles aren't the only insects that evolved this adaptation. Leaf-cutter ants also carry symbiotic bacteria in exoskeleton pockets, and there's a species of weevil that carries yeast inside special pockets, Flórez said.
Although this new research illuminates how the bacteria get from the pupa pockets onto the beetle's abdomen, the study authors have a few more questions, Flórez said. For example, further research could reveal how the bacteria actually get inside the beetles' glands, how the bacteria protect the beetles against fungi, and if bacteria offer protection against animal predators, too.
Originally published on Live Science.

JoAnna Wendel is a freelance science writer living in Portland, Oregon. She mainly covers Earth and planetary science but also loves the ocean, invertebrates, lichen and moss. JoAnna's work has appeared in Eos, Smithsonian Magazine, Knowable Magazine, Popular Science and more. JoAnna is also a science cartoonist and has published comics with Gizmodo, NASA, Science News for Students and more. She graduated from the University of Oregon with a degree in general sciences because she couldn't decide on her favorite area of science. In her spare time, JoAnna likes to hike, read, paint, do crossword puzzles and hang out with her cat, Pancake.

 Live Science Plus
Live Science Plus





