What is the first law of thermodynamics?
The first law of thermodynamics states that energy cannot be created or destroyed, but it can be transferred.

Ashley Hamer Pritchard
Related content
The first law of thermodynamics states that heat is a form of energy, and thermodynamic processes are therefore subject to the principle of conservation of energy. This means that heat energy cannot be created or destroyed, according to Britannica. It can, however, be transferred from one location to another and converted to and from other forms of energy.
Thermodynamics is the branch of physics that deals with the relationships between heat and other forms of energy. In particular, it describes how thermal energy is converted to and from other forms of energy and how it affects matter. The fundamental principles of thermodynamics are expressed in four laws.
"The First Law says that the internal energy of a system has to be equal to the work that is being done on the system, plus or minus the heat that flows in or out of the system and any other work that is done on the system," Saibal Mitra, a professor of physics at Missouri State University, told Live Science. "So, it's a restatement of conservation of energy."
"The change in internal energy of a system is the sum of all the energy inputs and outputs to and from the system similarly to how all the deposits and withdrawals you make determine the changes in your bank balance," Mitra said.
This is expressed mathematically as: ΔU = Q — W, where ΔU is the change in the internal energy, Q is the heat added to the system, and W is the work done by the system, according to Britannica.
History of the first law of thermodynamics
Scientists in the late 18th and early 19th centuries adhered to caloric theory, first proposed by Antoine Lavoisier in 1783, and further bolstered by the work of Sadi Carnot in 1824, according to the American Physical Society. This scientific theory treated heat as a kind of fluid that naturally flowed from hot to cold regions, much as water flows from high to low places. When this caloric fluid flowed from a hot to a cold region, it could be converted to kinetic energy and made to do work much as falling water could drive a water wheel. It wasn't until Rudolf Clausius published "The Mechanical Theory of Heat" in 1867 that caloric theory was finally put to rest, according to the University of Virginia.
Thermodynamic systems
Energy can be divided into two parts, said David McKee, a professor of physics at Missouri Southern State University. One is our human-scale macroscopic contribution, such as a piston moving and pushing on a system of gas. The rest is made up of the things that happen at a very tiny scale where we can't keep track of the individual contributions.
Get the world’s most fascinating discoveries delivered straight to your inbox.
"When I put two samples of metal up against each other, and the atoms are rattling around at the boundary, and two atoms bounce into each other, and one of them comes off faster than the other, I can't keep track of it. It happens on a very small time scale and a very small distance, and it happens many, many times per second," McKee told Live Science. "So, we just divide all energy transfer into two groups: the stuff we're going to keep track of, and the stuff we're not going to keep track of. The latter of these is what we call heat."
Thermodynamic systems are generally regarded as being open, closed or isolated. According to the University of Calgary, an open system freely exchanges energy and matter with its surroundings; a closed system exchanges energy, but not matter, with its surroundings; and an isolated system does not exchange energy or matter with its surroundings. For example, a pot of boiling soup receives energy from the stove, radiates heat from the pan, and emits matter in the form of steam, which also carries away heat energy. This would be an open system. If we put a tight lid on the pot, it would still radiate heat energy, but would ideally no longer emit matter in the form of steam. This would be a closed system. However, if we were to pour the soup into a perfectly insulated thermos bottle and seal the lid, there would be no energy or matter going into or out of the system. This would be an isolated system.
In practice, however, perfectly isolated systems cannot exist. All systems transfer energy to their environment no matter how well insulated they are. The soup in the thermos will only stay hot for a few hours and will reach room temperature by the following day. In another example, white dwarf stars, the hot remnants of burned-out stars that no longer produce energy, can be insulated by light-years of near perfect vacuum in interstellar space, yet they will eventually cool down from several tens of thousands of degrees to near absolute zero due to energy loss through radiation. Although this process takes longer than the present age of the universe, there's no stopping it.
Heat engines
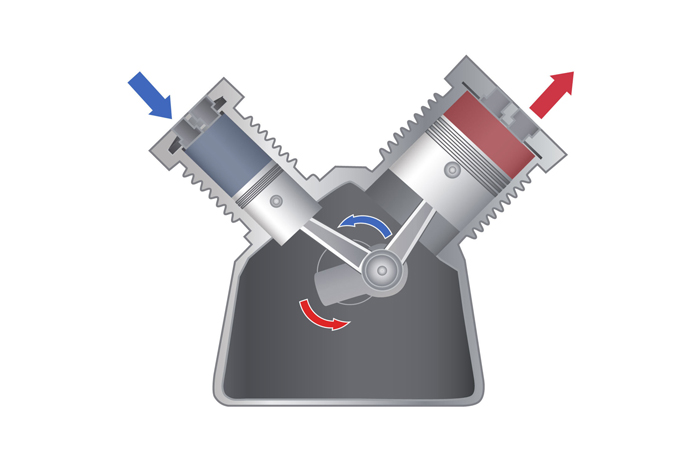
The most common practical application of the First Law is the heat engine. Heat engines convert thermal energy into mechanical energy and vice versa. Most heat engines fall into the category of open systems. The basic principle of a heat engine exploits the relationships among heat, volume and pressure of a working fluid (any substance that flows), typically a gas, according to Georgia State University. Examples of working fluids include steam in a steam engine and hydrofluorocarbons in refrigeration systems.
When gas is heated, it expands; however, when that gas is prevented from expanding, it increases in pressure. If the bottom wall of the confinement chamber is the top of a movable piston, this pressure exerts a force on the surface of the piston causing it to move downward. This movement can then be harnessed to do work equal to the total force applied to the top of the piston times the distance that the piston moves.
There are numerous variations on the basic heat engine. For instance, steam engines rely on external combustion to heat a boiler tank containing the working fluid, typically water. The water is converted to steam, and the pressure is then used to drive a piston that converts heat energy to mechanical energy. Automobile engines, however, use internal combustion, where liquid fuel is vaporized, mixed with air and ignited inside a cylinder above a movable piston, driving it downward, according to The University of Oklahoma.
Refrigerators, air conditioners and heat pumps
Refrigerators and heat pumps are heat engines that convert mechanical energy to heat. Most of these fall into the category of closed systems. When the working fluid, or gas, is compressed, its temperature increases. This hot gas can then transfer heat to its surrounding environment. Then, when the compressed gas is allowed to expand, its temperature becomes colder than it was before it was compressed because some of its heat energy was removed during the hot cycle. This cold gas can then absorb heat energy from its environment. This is the working principle behind an air conditioner, according to Boston University. Air conditioners don't actually produce cold; they remove heat.
A mechanical pump transfers the working fluid outdoors, where it is heated by compression. Next, the heat transfers to the outdoor environment, usually through an air-cooled heat exchanger, which often uses an electric fan to expel heat into the environment. Then, the working fluid is brought back indoors, where it is allowed to expand and cool so it can absorb heat from the indoor air through another heat exchanger.
A heat pump is simply an air conditioner run in reverse. The heat from the compressed working fluid is used to warm the building. It is then transferred outside where it expands and becomes cold, thereby allowing it to absorb heat from the outside air, which even in winter is usually warmer than the cold working fluid. The working fluid typically has a low enough freezing point to keep flowing even in very low temperatures.
Geothermal or ground-source air conditioning and heat pump systems use long U-shaped tubes in deep wells or an array of horizontal tubes buried in a large area through which the working fluid is circulated, and heat is transferred to or from the earth, according to the U.S. Department of Energy. Other systems use rivers or ocean water to heat or cool the working fluid.
Live Science contributor Ashley Hamer updated this article on Jan. 28, 2022.
Additional resources
Here are three other explanations of the first law of thermodynamics:
- Conceptual Academy on YouTube
- George Mason University earth science professor Robert M. Hazen on The Great Courses Daily
- This OpenStax College Physics chapter from the University of Iowa
Bibliography
Britannica, "The first law of thermodynamics," June 1 2021. https://www.britannica.com/science/thermodynamics/The-first-law-of-thermodynamics
Science History Institute, "Antoine-Laurent Lavoisier," December 11 2017. https://sciencehistory.org/historical-profile/antoine-laurent-lavoisier
The American Society of Mechanical Engineers, "Nicolas Léonard Sadi Carnot," April 10 2012, https://www.asme.org/topics-resources/content/nicolas-leonard-sadi-carnot
Rudolfph Clausius, "The Mechanical Theory of Heat." John Van Voorst, 1867.
American Physical Society, "This Month Physics History December 1840: Joule's abstract on converting mechanical power into heat," December 2009. https://www.aps.org/publications/apsnews/200912/physicshistory.cfm
University of Virginia, "Teaching Heat: the Rise and Fall of the Caloric Theory," July 2003. http://galileoandeinstein.physics.virginia.edu/more_stuff/TeachingHeat.htm
University of Calgary Energy Education, "System and Surrounding," September 27, 2021. https://energyeducation.ca/encyclopedia/System_and_surrounding
University of Georgia Hyperphysics, "Heat Engine Cycle," http://hyperphysics.phy-astr.gsu.edu/hbase/thermo/heaeng.html
University of Oklahoma ECourses, "Thermodynamics - Theory." http://www.ecourses.ou.edu/cgi-bin/ebook.cgi?topic=th&chap_sec=08.1&page=theory
Boston University, "Heat engines and the second law," December 10 1999. http://physics.bu.edu/~duffy/py105/Heatengines.html
US Department of Energy, "Geothermal Heat Pumps." https://www.energy.gov/energysaver/geothermal-heat-pumps

- Ashley Hamer PritchardLive Science Contributor

 Live Science Plus
Live Science Plus





