Scientists discover on-off switch for bacteria that breathe electricity

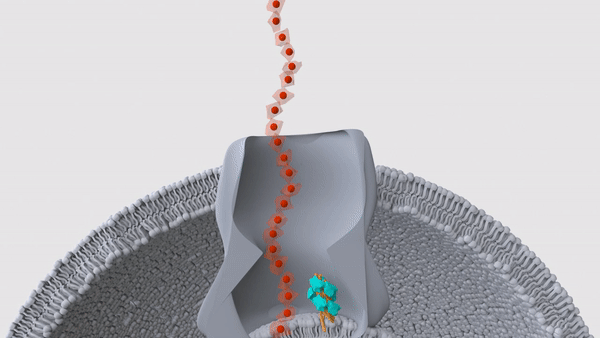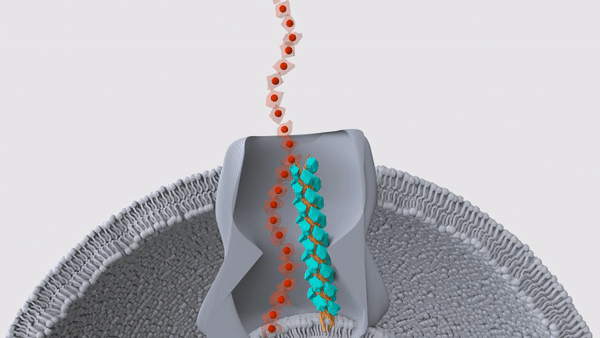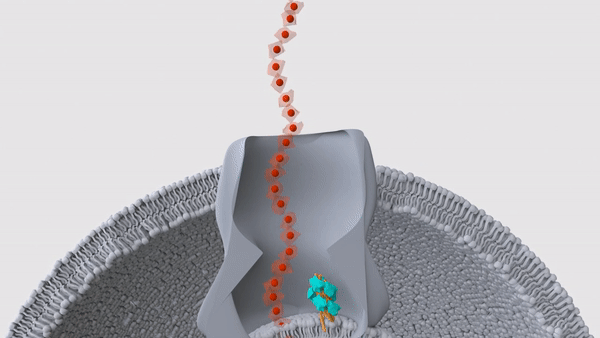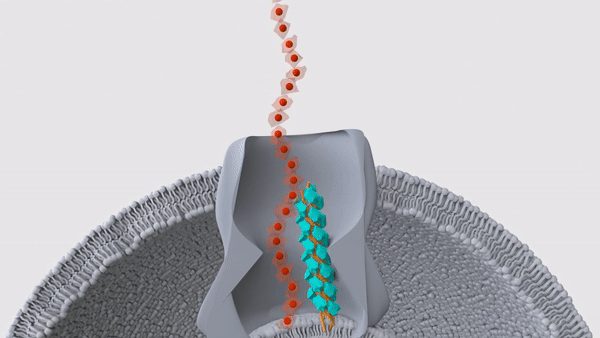
A hair-like structure (light blue) in a Geobacter microbe pushes a nanowire (red) out through the surface of the cell. These nanowires allow the bacteria to "exhale" electricity.
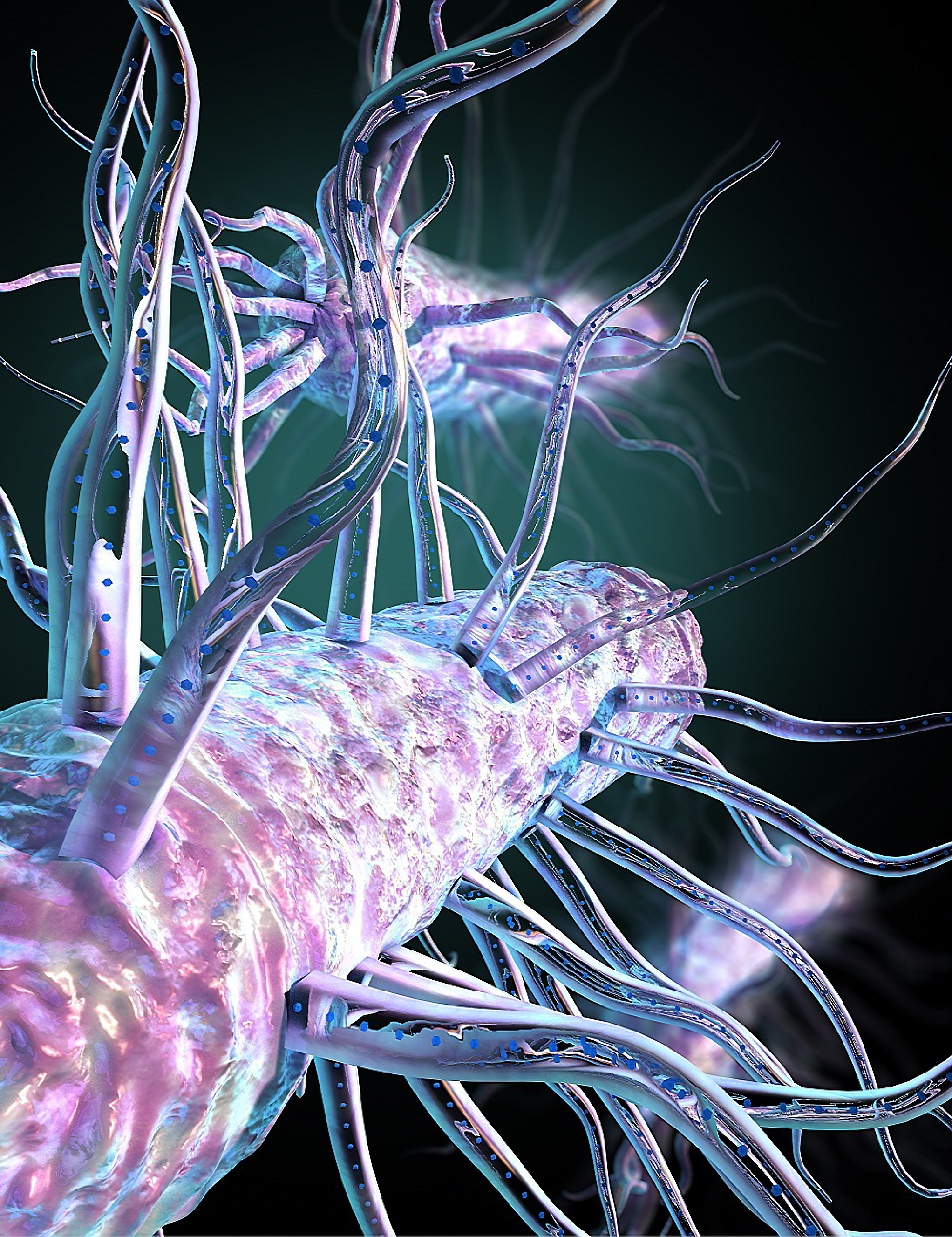
Deep beneath the seabed, teensy bacteria "exhale" electricity through long, skinny snorkels, and now, scientists have discovered how to switch these microbes' electric breath on and off.
These bizarre bacteria rely on two proteins, which band together in a single hair-like structure called a pilus, the researchers reported in a new study, published Wednesday (Sept. 1) in the journal Nature. Many of these pili lie just beneath the bacterial membrane and help push the snorkels out of the cell and into the surrounding environment, thus allowing the microbe to breathe.
This discovery not only reveals something unexpected about the bacteria's biology but could also pave the way for new technologies, from powerful microbe-powered batteries to new medical treatments for bacterial infections, senior author Nikhil Malvankar, an assistant professor of molecular biophysics and biochemistry at Yale University's Microbial Sciences Institute, told Live Science.
Related: Extreme life on Earth: 8 bizarre creatures
The bacteria belong to the genus Geobacter and can be found all across the world, growing deep underground in soils that are totally devoid of oxygen. Humans rely on oxygen to convert food into usable energy and to sop up electrons that are left over from this metabolic process. If the leftover electrons accumulated, they would quickly become toxic to the body, Malvankar said.
Just like humans, Geobacter microbes generate waste electrons during metabolism, but they don't have access to oxygen like we do. So, to get rid of their excess electrons, the bacteria coat themselves in thin, conductive filaments, called nanowires, which can shuttle electrons out of the microbes and to other bacteria or minerals in the environment, such as iron oxide.
These thin nanowires are 100,000 times smaller than the width of a human hair and can transport electrons over huge distances, hundreds to thousands of times the original microbe's body length, Live Science previously reported.
Get the world’s most fascinating discoveries delivered straight to your inbox.
"I cannot breathe oxygen which is like 100 meters [328 feet] away from me," Malvankar said. "And somehow, these bacteria are using these nanowires like a snorkel which is 100 times their size, so that they can keep breathing over such long distances." This impressive feat generates an electric current, as electrons continually flow through the lengthy nanowires.
But although scientists discovered these nanowires in the early 2000s, Malvankar and his colleagues only recently discovered what the cellular snorkels are actually made of. Initially, scientists assumed that the nanowires were pili. This notion seemed to be supported by the fact that, if you delete the genes needed for pili construction from Geobacter bacteria, nanowires no longer appear on their surfaces, Malvankar said.
But there was a problem: Pili proteins don't contain any metals, like iron, that conduct electricity. Malvankar and his team investigated this conundrum in a 2019 study, published in the journal Cell, during which they examined Geobacter bacteria using cryo-electron microscopy (cryo-EM), a technique that involves shining a beam of electrons through a substance to take a snapshot of its component molecules.
Related: The bacteria in your gut produce electricity
Nanowires branch from the surface of Geobacter microbes.
"That's when we realized that there are no pili on the bacterial surface at all," Malvankar said. "That was a big surprise." Instead, the team found that the nanowires were made of proteins called cytochromes, which readily transfer electrons down their lengths and therefore make much better nanowires than pili. In a 2020 study, published in the journal Nature Chemical Biology, the team reported that these cytochrome-based nanowires come in multiple "flavors," which conduct electricity with different levels of efficiency.
But even after the team revealed the chemical makeup of the nanowires, pili proteins still cropped up in their biochemical assessments of the Geobacter bacteria. If the pili weren't conducting electricity, "the real big question was, you know, what do these pili really do? Where are they?" Malvankar said.
In their newest Nature study, the team looked more closely at the structure of these pili by first deleting the genes for nanowires in lab-grown Geobacter sulfurreducens. The pili would usually be blocked in by the nanowires, so without those structures in the way, the hair-like projections sprouted from the surface of the cells. This gave the team a chance to examine the pili with cryo-EM, which revealed the two distinct proteins — PilA-N and PilA-C — within each hair.
The team also ran tests to see how well the pili conducted electricity, and found that "they move electrons 20,000 times slower than OmcZ," the cytochrome protein that forms the most-highly conductive Geobacter nanowires, Malvankar said; "they are just not really made to move electrons."
That said, the pili looked like they might serve a different function, the team noticed. In other bacterial species, some pili sit beneath the cell membrane and move like tiny pistons; this motion lets them push proteins through the membrane, and up and out of the cell. For example, the bacterium Vibrio cholerae, which causes the diarrheal illness cholera, uses such pili to secrete cholera toxin, according to a 2010 report in the journal Nature Structural & Molecular Biology. In a series of experiments, the team determined that the pili in Geobacter fulfill a similar role, in that they help shove nanowires through the microbial membrane.
"We found that the cytochromes are stuck inside the bacteria when the piston protein is not there," Malvankar said. "And when we put the gene back, the cytochromes are able to get out of the bacteria." This, then, was the bacteria's on-off switch, the team concluded.
Looking forward, the researchers plan to investigate how many other types of bacteria build nanowires and use them to breathe electricity. They're also interested in exploring practical applications for the research.
RELATED CONTENT
Researchers have used Geobacter colonies to power small electronics for more than a decade, but as of yet, these bacterial batteries can produce only tiny amounts of power, Live Science previously reported. In past research, Malvankar and his team found that the colonies can be made more conductive under the influence of an electric field, which could help boost the power of these devices; now, the new research could provide scientists another degree of control, by allowing them to switch electricity on or off.
This research could also have applications in medicine and, in particular, in treatments for bacterial infections, Malvankar said. For example, Salmonella manages to outgrow beneficial bacteria in the gut because it can switch from fermentation, which produces energy slowly with no oxygen required, to respiration, which produces energy quickly and usually requires oxygen, Live Science previously reported. In the low-oxygen environment of the intestines, Salmonella uses a compound called tetrathionate as a substitute for oxygen, thus outcompeting beneficial bacteria in the body.
But what if those helpful bacteria could get a leg up? In theory, if you equipped bacteria with nanowires and introduced them into the gut, as a kind of probiotic treatment, they could potentially outcompete harmful pathogens such as Salmonella, Malvankar said. Malvankar and his colleagues are studying this potential course of treatment, but the work is still in its early stages.
Originally published on Live Science.

Nicoletta Lanese is the health channel editor at Live Science and was previously a news editor and staff writer at the site. She is a recipient of the 2026 AHCJ International Health Study Fellowship, with a project focused on antibiotic stewardship practices in Japan and the U.S. They hold a graduate certificate in science communication from UC Santa Cruz and degrees in neuroscience and dance from the University of Florida. Beyond Live Science, Lanese's work has appeared in The Scientist, Science News, the Mercury News, Mongabay and Stanford Medicine Magazine, among other outlets. Based in NYC, she also remains involved in dance and performs in local choreographers' work.

 Live Science Plus
Live Science Plus




