What Is a Polymer?

Polymers are materials made of long, repeating chains of molecules. The materials have unique properties, depending on the type of molecules being bonded and how they are bonded. Some polymers bend and stretch, like rubber and polyester. Others are hard and tough, like epoxies and glass.
Polymers touch almost every aspect of modern life. Chances are most people have been in contact with at least one polymer-containing product — from water bottles to gadgets to tires — in the last five minutes.
The term polymer is often used to describe plastics, which are synthetic polymers. However, natural polymers also exist; rubber and wood, for example, are natural polymers that consist of a simple hydrocarbon, isoprene, according to Encyclopedia Britannica. Proteins are natural polymers made up of amino acids, and nucleic acids (DNA and RNA) are polymers of nucleotides — complex molecules composed of nitrogen-containing bases, sugars and phosphoric acid, for example.
Chemical reactions
Hermann Staudinger, a professor of organic chemistry at the Eidgenössische Technische Hochschule (University of Applied Sciences) in Zurich, is the father of modern polymer development. His research in the 1920s led the way to modern manipulations of both natural and synthetic polymers. He coined two terms that are key to understanding polymers: polymerization and macromolecules, according to the American Chemical Society (ACS). He was awarded a Nobel Prize in Chemistry in 1953 "for his discoveries in the field of macromolecular chemistry."
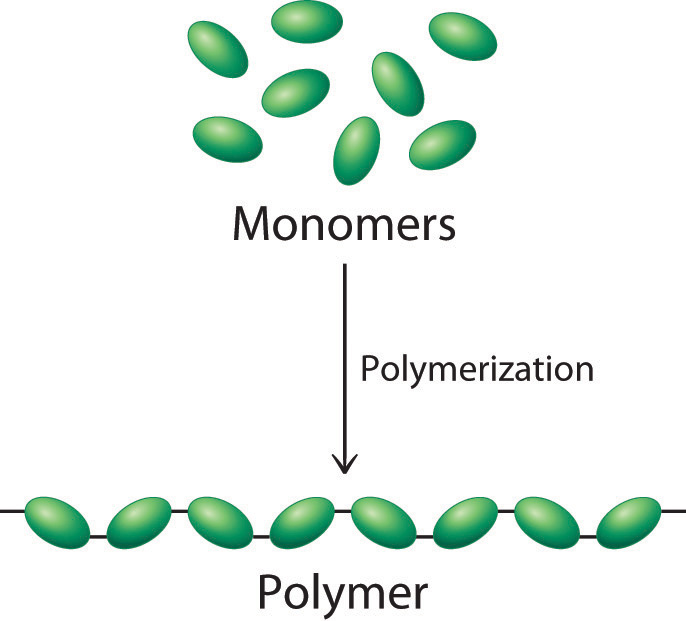
Polymerization is the method of creating synthetic polymers by combining smaller molecules, called monomers, into a chain held together by covalent bonds, according to ThoughtCo., an online educational resource. Various chemical reactions — those caused by heat and pressure, for example — alter the chemical bonds that hold monomers together, according to Scientific American. The process causes the molecules to bond in a linear, branched or network structure, resulting in polymers.
These chains of monomers are also called macromolecules. Most polymer chains have a string of carbon atoms as a backbone. A single macromolecule can consist of hundreds of thousands of monomers, according to the Polymer Science Learning Center.
Uses of polymers
Polymers are used in almost every area of modern living. Grocery bags, soda and water bottles, textile fibers, phones, computers, food packaging, auto parts, and toys all contain polymers.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Even more-sophisticated technology uses polymers. For example, "the membranes for water desalination, carriers used in controlled drug release and biopolymers for tissue engineering all use polymers," according to the ACS.
Popular polymers for manufacturing include polyethylene and polypropylene. Their molecules can consist of 10,000 to 200,000 monomers.
Future of polymers
Researchers are experimenting with many different types of polymers, aiming to further medicine development and enhance products we already use.
For example, carbon polymers are being developed and enhanced for the automotive industry.
"Carbon-fiber-reinforced polymer (CFRP) composites — also called carbon-fiber laminates — are the next-generation materials for making cars lighter, more fuel efficient and safer," according to a 2016 Live Science column by Nikhil Gupta, an associate professor, and Steven Zeltmann, a student researcher, both in the Composite Materials and Mechanics Laboratory of the Mechanical and Aerospace Engineering Department at New York University Tandon School of Engineering. "Carbon laminate is extremely strong and stiff because of its woven layers of nearly pure carbon fibers bonded together by a hardened plastic, such as epoxy resin." [The Carbon-Fiber Future: It's About More Than Speed]
Polymers are also being used to enhance holograms. Scientists at the University of Pennsylvania created a hologram on flexible polymer material called PDMA that was embedded with gold nanorods, according to a study published online in early 2017 in the journal Nano Letters. This new hologram device can hold several images instead of just one.
"The question we asked is, 'Can we encode multiple bits of information in a hologram?'" Ritesh Agarwal, research leader and professor of materials science and engineering at the University of Pennsylvania, told Live Science. "It's an important piece of work, because it's the first time someone's shown you can record multiple holographic images, and by just stretching the polymer, you can basically change the image."
Artificial skin made of a silicone polymer may be the future of anti-aging efforts. In the form of two creams, the polymer may be able to tighten a person's skin, reduce the appearance of wrinkles and diminish under-eye bags, according to a study published May 2016 in the journal Nature Materials. Such artificial skin may also be used to help those with skin conditions, such as eczema, or be used as sun block.
"We are excited about it; it is a brand-new material," study co-author Robert Langer, a professor at the Massachusetts Institute of Technology, told Live Science.
Additional resources


 Live Science Plus
Live Science Plus





