Pain-Fighting Implant Directly Targets Nerves

In the future, back pain might be treated with implants that deliver drugs right where it hurts, which would make taking pills a thing of the past.
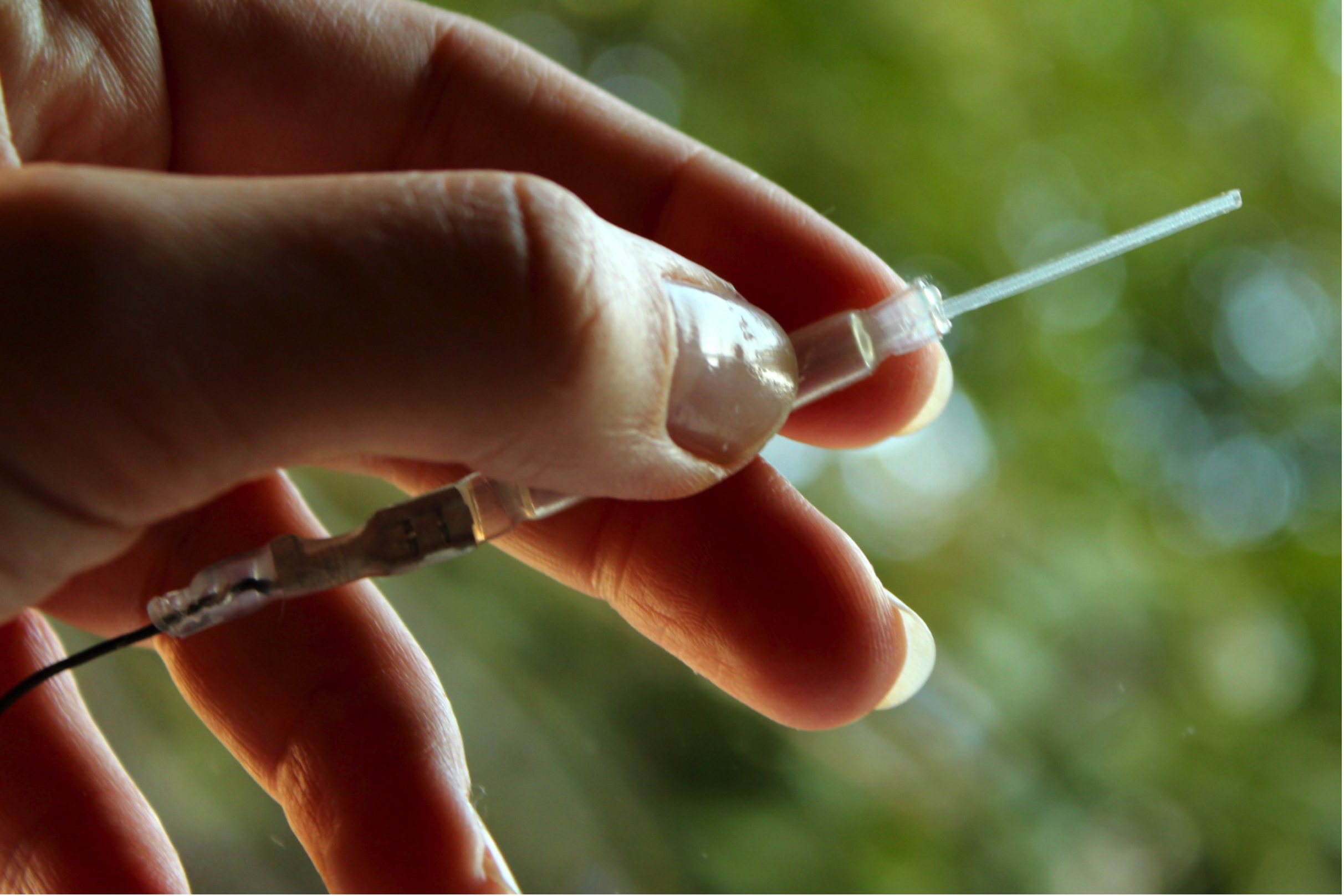
For now, researchers in Sweden have built an implant that treats nerve pain in rats. The implant is a small tube made of polymers with a reservoir for medication; whenever a current is turned on, the device can release a drug onto a nearby nerve.
Nerve pain — also called neuropathic pain — is a common chronic problem, caused by damage to the nerves themselves (rather than injury to the surrounding muscle or bone). For example, some people suffer from debilitating neuropathic pain in the lower back because of compression of the sciatic nerve, a large nerve that runs from the lower back down through the leg.
This type of pain can be treated with painkillers, but the problem is that current ways of taking painkillers release the medicine throughout the body. This means the doses have to be high enough that the amount of drug that reaches the site of the pain is effective, according to the researchers who built the new implant. Such high doses can cause side effects, such as drowsiness, and can even lead to addiction.
A neural implant could change that, because it could deliver drugs to specific nerves and use lower doses, the researchers said. [5 Surprising Facts About Pain]
The goal of making the new implant was to create a permanent solution to nerve pain, said Daniel Simon, assistant professor at Linköping University's Laboratory of Organic Electronics and one of the authors of the new study. "The vision is a real neural prosthesis," he said.
In experiments, the researchers tested out their implant on rats that had damage to their sciatic nerve on one side of the body. The polymer tube released a compound called GABA, which is a neurotransmitter that suppresses nerve function. The tube was designed to have four openings, so it could release GABA at the four junctions where the sciatic nerve connects to the spine.
Get the world’s most fascinating discoveries delivered straight to your inbox.
"We want to deliver GABA right at the point where the nerve enters the spinal cord," Simon said. Many people with neuropathy have pain because their nerves are not getting enough GABA, which is one of the compounds the body uses to regulate nerve response, Simon said.
The researchers then tested the rats' sensitivity to pain by pressing down on their paws. In general, rats already suffering from nerve pain due to a damaged sciatic nerve would be expected to be quite sensitive to more pain from pressure on their paws.
The researchers found that when the implant was turned on, the rats flinched less in response to this pressure on their paws than did control rats that didn't have the implant, or rats with the device turned off.
To make sure that the implant didn't itself cause any injury to the rats, the researchers also tested the animals' nerve function on the uninjured sides of their bodies, and gave some rats a sham operation, in which no implant was used. The implant seemed to cause no problems by itself.
Eventually, the device could be used to deliver drugs other than GABA, the researchers said.
Simon said it will be a long road before this device could become commercially available for use in people. The researchers will first have to show that the device can stay in the body for longer periods than the current study — months rather than weeks. The inventors would also have to demonstrate that it is safe for humans to use and that the drugs can't leak out where they aren't wanted.
Still, the fact that the implant worked in a living animal that was awake was a first, the study authors wrote. Simon noted that human spinal nerves and the spaces between them are also larger. "In some ways, designing for humans is easier," he said.
The research is published today (May 8) in the journal Science Advances.
Follow Live Science @livescience, Facebook & Google+. Originally published on Live Science.


 Live Science Plus
Live Science Plus





