Facts About Einsteinium

Einsteinium, the 99th element on the Periodic Table of Elements, is a synthetic element that is produced in extremely small amounts and with a very short lifetime. If the name seems familiar, it's because it is indeed named after famed physicist Albert Einstein, although he had nothing to do with the element's discovery or research.
History
Einsteinium was discovered during the examination of debris from the first hydrogen bomb test in November 1952, according to Chemicool. A team of scientists from the Lawrence Berkeley National Laboratory, the Argonne National Laboratory and the Los Alamos Scientific Laboratory and led by Albert Ghiorso, an American nuclear scientist at Berkeley, studied the debris collected by drones using chemical analysis. Minuscule amounts of einsteinium-253, an isotope of einsteinium, were discovered (less than 200 atoms, according to an article printed in Nature Chemistry by Joanne Redfern, a British science writer, in 2016). Fermium, the 100th element, was also discovered in the debris.
The results of the test were not published until 1955, according to Peter van der Krogt, a Dutch historian. At the time of the hydrogen bomb demonstration, tensions due to the Cold War were running high and many new discoveries were kept secret. Due to the method of creation and the nature of the new elements, further research of einsteinium continued in silence, according to the Los Alamos National Laboratory.
Article continues belowFurther production and research on einsteinium, as well as fermium, was done at the Oak Ridge National Laboratory in Tennessee, according to Lenntech. The scientists created larger amounts of the element and also developed methods of purifying the einsteinium, as described in a 1978 paper by D.E. Ferguson and presented at a symposium. The paper describes the production of einsteinium and fermium in nuclear reactors by bombarding heavy elements such as uranium and curium with neutrons and having those products undergo radioactive decay. Einsteinium and the other heavy elements were then extracted from a tank filled with a solvent.

Just the facts
- Atomic number (number of protons in the nucleus): 99
- Atomic symbol (on the Periodic Table of Elements): Es
- Atomic weight (average mass of the atom): 252
- Density: Unknown
- Phase at room temperature: solid
- Melting point: 1,580 degrees Fahrenheit (860 degrees Celsius)
- Boiling point: Unknown
- Number of natural isotopes (atoms of the same element with a different number of neutrons): 0. There are also at least 19 artificial isotopes created in a lab, none of which are stable.
Who knew?
- Einsteinium-253 is the product of combining 15 neutrons with uranium-238, which then undergoes seven beta decays.
- Einsteinium-253 has a half-life of 20.5 days, according to the Royal Society of Chemistry.
- Scientists at Lawrence Berkeley National Laboratory, the Argonne National Laboratory, and the Los Alamos Scientific Laboratory published the discovery of einsteinium and fermium on June 9, 1955, for the United States Atomic Energy Commission.
- Einsteinium-252 is the most stable isotope of einsteinium and has a half-life of about 471.7 days, according to the Jefferson Laboratory.
- According to the Royal Society of Chemistry, einsteinium has no uses other than scientific research.
- Einsteinium is an actinide element, according to Lenntech, and is found on the bottom row of the periodic table. Actinide elements are attacked by oxygen, steam and acids, but not by alkali metals, such as lithium, sodium, potassium, rubidium, cesium, and francium.
- According to the Los Alamos National Laboratory, einsteinium was the seventh transuranic element to be discovered. Transuranic elements are artificially made, radioactive elements, according to the U.S. Nuclear Regulatory Commission.
- Enough einsteinium had been collected to be visible to the naked eye by 1961 by researchers in Berkeley, California, according to the Royal Society of Chemistry. The amount weighed approximately ten millionths of a gram (1.0 x 10-5 grams or 3.5 x 10-7 ounces).
- Einsteinium is created in very small amounts from bombarding plutonium with neutrons in a nuclear reactor, according to the Royal Society of Chemistry.
- Einsteinium is soft and silver in color, according to Elements Database.
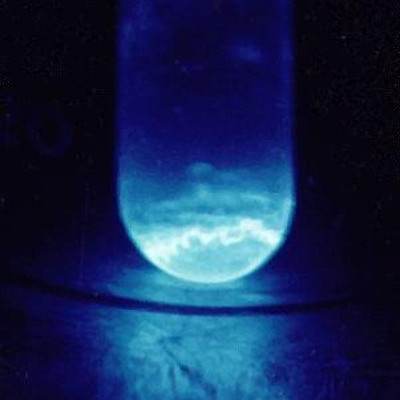
- Einsteinium glows blue in the dark due to the great release of energy as it undergoes radioactive decay, according to Redfern.
- Einsteinium is highly radioactive, according to Lenntech, but as it is not a naturally occurring element, there are no known health risks to the general population. Those who work closely with einsteinium in a laboratory, however, must take precautions to protect themselves from the radiation.
- Due to the rapid decay of einsteinium, it is difficult to study the pure element, according to Redfern. Einsteinium decays into berkelium and californium, causing the nearly all samples of einsteinium to be contaminated.
- Some early reports about the discovery of element 99 unofficially named the element Athenium, for the Greek capital of Athens, according to van der Krogt. The element was officially named for Albert Einstein.
- Einstein was an ironic choice for the name of einsteinium, according to Redfern. Einstein was a pacifist and opposed to the hydrogen bomb.
Current research
There is very little research involving einsteinium. According to Redfern, the main use of einsteinium is to create heavier elements, including mendelevium. Due to the high rate of decay and radioactive nature, there are currently no other uses for einsteinium.
Additional resources
- Peter van der Krogt’s Elementymology website
- Los Alamos National Laboratory
- Images of Elements: Einsteinium
Get the world’s most fascinating discoveries delivered straight to your inbox.

Rachel Ross is a science writer and editor focusing on astronomy, Earth science, physical science and math. She holds a Bachelor of Arts in Philosophy from the University of California Davis and a Master's degree in astronomy from James Cook University. She also has a certificate in science writing from Stanford University. Prior to becoming a science writer, Rachel worked at the Las Cumbres Observatory in California, where she specialized in education and outreach, supplemented with science research and telescope operations. While studying for her undergraduate degree, Rachel also taught an introduction to astronomy lab and worked with a research astronomer.