Why are vinegar and baking soda so good for cleaning?
It's basic (and acidic too).


More and more people are tossing out the harsh chemicals from their daily cleaning routine and instead turning to natural products, such as baking soda and vinegar, to remove grime, disinfect surfaces and leave spaces shiny and clean, according to Reader's Digest. So why are these household items such effective cleaning agents? The answer is pretty basic — baking soda and vinegar lie on opposite ends of the pH scale.
"When you are cleaning using baking soda or vinegar, you are actually doing very complicated manipulations of molecules," said May Nyman, a professor in the department of chemistry at Oregon State University.
Baking soda is the common name for sodium bicarbonate (NaHCO3). Most people probably associate it with cooking, because it makes your cakes and breads big and puffy. Vinegar is a dilute solution of acetic acid (HC2H3O2), produced by bacteria during fermentation.
Related: Does salt make water boil faster?
"Baking soda is the opposite of vinegar. It is harsh like vinegar but dissolves organic matter," Nyman told Live Science. "Like vinegar, it cannot harm you and will not be harmful when cleaning places where you store your food."
Both kitchen ingredients are effective cleaning agents because they are found on opposite sides of the pH scale. pH is a measure of how acidic or basic a substance is, on a scale from 1 (very acidic) to 14 (very basic), with a neutral value at 7. Pure water has a pH of 7. Baking soda has a pH of 9, while vinegar has a pH of 2, according to the U.S. Geological Survey.
Cleaning one-two punch
As a base, baking soda dissolves organic compounds like dirt, grease and other sticky ickies. In addition, the mineral structure of each baking soda particle provides a gentle abrasive to clean without leaving scratches behind. As an acid, vinegar breaks down minerals that form from hard tap water, forming unsightly stains on sinks, tubs and counters.
Get the world’s most fascinating discoveries delivered straight to your inbox.
RELATED MYSTERIES
Combining these two common household substances can produce incredible results in the kitchen, but it's important to not combine them in equal amounts because you need to keep the mixture in either the acidic or basic side of the neutral value. When baking soda is mixed with vinegar, the acid breaks down baking soda, releasing carbon dioxide gas that can help lift dirt from the surfaces being cleaned.
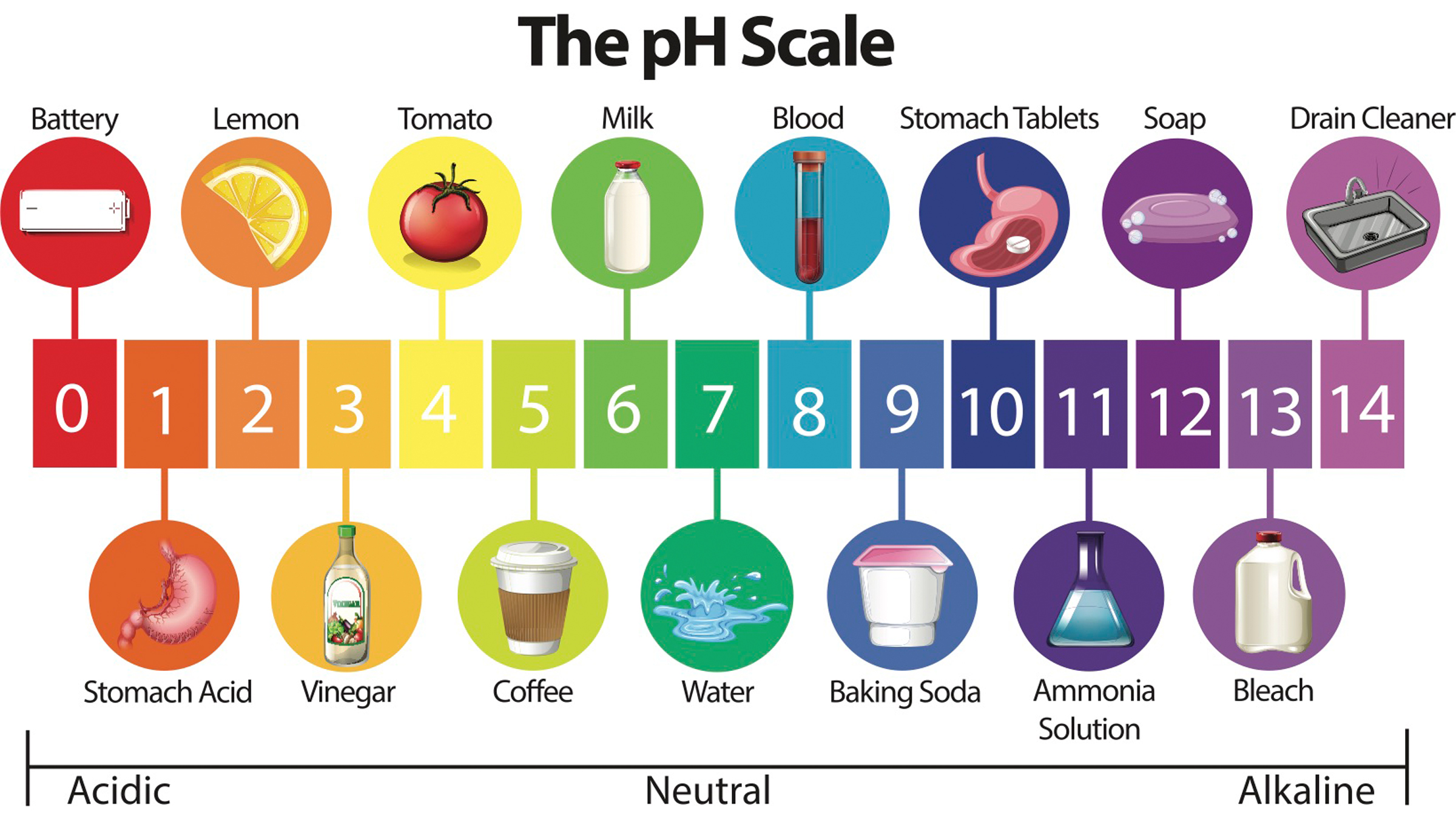
Everyday items on the pH scale.
Here are some recipes to try.
- Freshen your sink by mixing one part of baking soda with two parts of vinegar. This mixture unlocks an effervescent fizz of carbon dioxide that cleans and freshen drains.
- Remove hard water stains by placing a vinegar-soaked towel over the affected area. After a few hours, remove the cloth and scrub the affected area with a paste made of baking soda and water.
- Kill mildew in laundry using baking soda and vinegar. Add a half a cup of baking soda with the laundry detergent to supercharge the cleaning process. Follow this with one cup of vinegar during the rinse cycle to kill bacteria and soften fabric.
- Clean grout by applying a baking soda paste made of baking soda and water. Spray the paste with vinegar before scrubbing the grime away.
Originally published on Live Science.

As a scientist, Stacy Kish has focused her research on Earth science, specifically oceanography and climate change. As a science writer, she explores all aspects of science from mites living books to noctilucent clouds, stretching across the mesopause. She finds every aspect of science intriguing and considers a good day to be one where she learns something new and unexpected. In her free time, she works on perfecting new cake recipes to share with others.
