Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
The Food and Drug Administration (FDA) has approved the first automated insulin-delivery system. This represents a step toward a so-called artificial pancreas that could automatically regulate blood sugar levels for people who have diabetes.
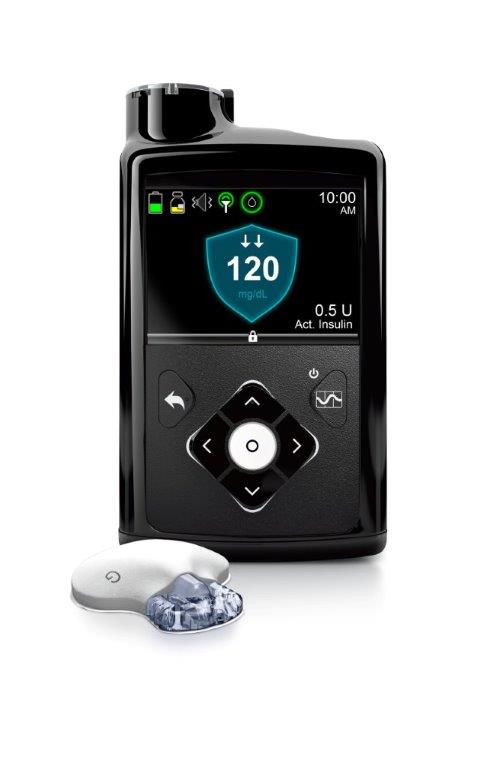
The device, made by the manufacturer Medtronic, has been approved to treat people with type 1 diabetes who are ages 14 and older. The product is set to start shipping in the spring of 2017, according to Medtronic.
"The FDA is dedicated to making technologies available that can help improve the quality of life for those with chronic diseases, especially those that require day-to-day maintenance and ongoing attention," Dr. Jeffrey Shuren, director of the FDA's Center for Devices and Radiological Health, said in a statement. "This first-of-its-kind technology can provide people with type 1 diabetes greater freedom to live their lives without having to consistently and manually monitor baseline glucose levels and administer insulin." [Bionic Humans: Top 10 Technologies]
Article continues belowThough the new product, called the MiniMed 670G, has been dubbed an artificial pancreas, it's a far cry from a truly biological cure for diabetes, as it does not replace the cells destroyed by the body. The device's insulin-delivery system relies on three elements: a coin-size transmitter attached to a tiny needle inserted into the skin that continuously tracks glucose levels, a pump that delivers precise doses of insulin as they are needed through a separate catheter inserted into the skin, and a computer chip that uses data from both to optimize the delivery of insulin on a minute-by-minute basis.
Elusive control
In people with type 1 diabetes, the body mistakes the beta cells in the pancreas, which make insulin, for foreign invaders, and attacks and destroys them. Insulin is the hormone that tells cells in the body to take in sugar from the blood and use it as fuel. Without insulin, blood sugar levels can skyrocket, and lead to a diabetic coma, and the body can waste away burning fat stores and muscle for energy.
To keep blood sugar levels from going too high or too low, people with type 1 diabetes typically monitor their blood sugar continuously throughout the day, either with a continuous glucose monitor or more typically by drawing a small drop of blood from the fingertip. They must inject insulin, either through a pump or a needle, several times a day.
Get the world’s most fascinating discoveries delivered straight to your inbox.
However, the amount of insulin needed on a given day varies unpredictably as a result of many unseen processes occurring in the body. For instance, insulin needs vary depending on what a person ate hours earlier, whether the individual has exercised recently and if the person is having a growth spurt, going through puberty, menstruating, getting sick, or feeling stressed or excited.
As a result, only 28 percent of teens and young adults with type 1 diabetes meet the targets for maintaining the blood sugar levels needed to reduce the risk of complications, according to a 2014 study in the journal Diabetes Care. Over the long term, persistently high blood sugar can damage organs, including the kidneys and heart, and the nerves. On the flip side, delivering too much insulin can induce dangerously low blood-sugar levels, a condition called hypoglycemic shock, which can cause seizures or death.
The 670G aims to avoid some of the variability in blood sugar by using a predictive model. It can estimate where blood sugar levels are likely to be in the next few minutes, and re-evaluate that level every 5 minutes. If blood sugar is plummeting, the system can shut off insulin delivery, or ramp it up to rein in spiking blood-sugar levels.
In clinical trials, the new system reduced A1C, a measure of the average blood sugar over three months, by a significant amount. The most striking improvements were seen in those individuals who had the highest blood sugar levels to start out.
Users of the new system will still have give themselves insulin doses based on the carbohydratecontent of their meals.
A number of other companies — including Beta Bionics, Insulet and Bigfoot Biomedical — are working on their own versions of artificial-pancreas insulin pumps.
Original article on Live Science.

Tia is the editor-in-chief (premium) and was formerly managing editor and senior writer for Live Science. Her work has appeared in Scientific American, Wired.com, Science News and other outlets. She holds a master's degree in bioengineering from the University of Washington, a graduate certificate in science writing from UC Santa Cruz and a bachelor's degree in mechanical engineering from the University of Texas at Austin. Tia was part of a team at the Milwaukee Journal Sentinel that published the Empty Cradles series on preterm births, which won multiple awards, including the 2012 Casey Medal for Meritorious Journalism.
 Live Science Plus
Live Science Plus









