
Drug Shortages Threaten US Anti-TB Efforts (Op-Ed)
Erica Lessem, assistant director for TB/HIV at Treatment Action Group (TAG), contributed this article to Live Science's Expert Voices: Op-Ed & Insights.
Shortages of essential medicines increasingly pose a challenge to healthcare in the United States, with the number of shortages nearly tripling from 2007 to 2012. This trend is especially worrisome for infectious diseases like tuberculosis (TB) that represent a public health threat . In fact, a recent U.S. Government Accountability Office report showed that 16 percent of drug shortages occurred among anti-infective drugs.
The United States has been a global leader in its efforts to eliminate TB as a public health problem. Yet mounting problems with accessing life-saving products to diagnose and treat TB threaten the progress made against this communicable disease, which kills as many people worldwide each year as HIV/AIDS.
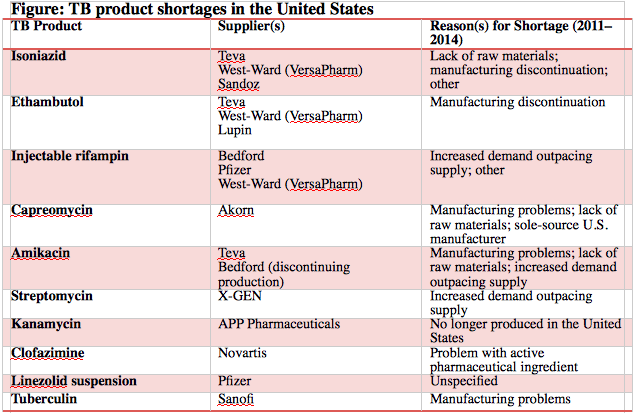
The majority of TB programs across the United States have had difficulty accessing medicines to treat TB — as well as products to diagnose the disease — due to shortages, supply interruptions and high costs.
Supply shortages can cause delays in diagnosing and treating TB , allowing patients to remain sick and infectious longer. These shortages can lead to treatment interruptions or the replacement of medicines with ones that are less effective, or more toxic or difficult to tolerate, giving patients side effects, and potentially driving drug resistance.
And shortages have devastating financial impacts: Recent shortages of tuberculin — the main component of the tuberculin skin test, which is injected into the forearm to test for skin reactions indicative of TB infection — have caused delays in the hiring of health care staff and other professionals who need to demonstrate recent TB tests. The problem extends beyond people, too: Tuberculin shortages have hamstrung the livestock industry. Without tuberculin, the industry cannot conduct the tests required to move herds of animals.
Overall, financially stressed state and local TB-program managers spend inordinate amounts of their limited time and resources addressing the challenges posed by shortages.
Get the world’s most fascinating discoveries delivered straight to your inbox.
But like TB, this plague of shortages is also diagnosable, curable and even preventable.
First, the diagnosis: The unstable supply is largely driven by the small number of manufacturers of TB products. When one company has an issue with production lines, or decides to discontinue a product, there are few, if any other producers ready to compensate with increased production. This small number of manufacturers, in turn, largely results from the challenges of making and selling TB products exclusively for the U.S. market. That market is relatively small and fragmented because it relies on individual, largely uncoordinated procurement from dozens of local and state TB programs.

Next, the cure: A strategic national reserve of TB medicines is essential to mitigating acute shortages. Such a central system could ensure that officials always have a set supply (say, six months' worth) on hand to treat TB should an issue arise. This system could rotate inventory to prevent expiration and waste; the U.S. Centers for Disease Control and Prevention (CDC), or an existing public- or private-sector drug distributor could manage this supply.
Finally, to prevent future shortages, the United States should and can find better ways to pool demand for TB products domestically and internationally. This will encourage a robust supply. Ironically, the United States supports other countries' procurement of quality-assured TB products by funding the Global Drug Facility, but does not rely on this important mechanism for domestic procurement.
U.S. programs should also buy drugs via the Global Drug Facility, with its multiple quality-assured manufacturers, while existing suppliers to the U.S. market should sell their drugs to the global market as well via the Global Drug Facility. This pooling of domestic and global markets would create much more stable demand and incentivize manufacturers to keep producing TB drugs.
To enable such pooling, the U.S. Food and Drug Administration (FDA) can work with the Global Drug Facility and the companies that supply it to facilitate the entry of more global, quality-assured medicines into the United States. The Global Drug Facility can also encourage those companies already registered here to tap into the global market.
TB elimination in the United States is within reach, but will slip beyond this country's grasp if drug shortages continue to challenge patient care. The United States urgently needs important changes to the way it buys and distributes drugs for this important public health problem, and can achieve those changes if the U.S. Congress provides funding and support to empower the CDC and FDA to make them.
The views expressed are those of the author and do not necessarily reflect the views of the publisher. This version of the article was originally published on Live Science.

 Live Science Plus
Live Science Plus





