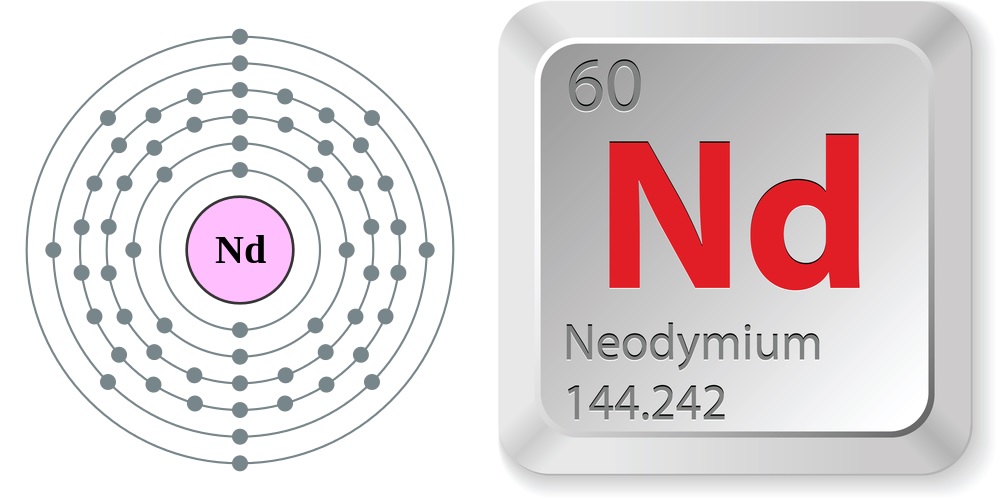
Facts About Neodymium
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
| Atomic Number: 60 Atomic Symbol: Nd Atomic Weight: 144.242 | Melting Point: 1,869.8 F (1,021 C) Boiling Point: 5,565.2 F (3,074 C) |
Word origin: Neodymium comes from the Greek word neos, meaning new, and didymos, meaning twin. The name combined means new twin.
Discovery: This element was discovered in 1841 when Swedish chemist Carl Gustaf Mosander, extracted a rose-colored oxide from cerite, which he named didymium, as it was a twin of the element lanthanum. In 1885, Austrian chemist Carl Auer von Welsbach separated didymium into two new elemental components, neodymium and praseodymium. [See Periodic Table of the Elements]
Properties of neodymium
Article continues belowNeodymium is one of the more reactive lanthanide rare-earth metals and quickly oxidizes in air. The element should be kept under an oil or sealed tightly in a plastic material. The metal has a bright and silvery luster.
Neodymium can be found in two allotropic forms, transforming from a double hexagonal to a body-centered cubic. Naturally-occurring neodymium has seven stable isotopes. Fourteen other radioactive isotopes are known.
Sources of neodymium
Ion-exchange or solvent extraction techniques are two methods used to obtain neodymium from its salts. The element can also be obtained by reducing anhydrous halides such as NdF3 with a calcium metal. Several other techniques to obtain the element are known.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Uses of neodymium
Neodymium colors glass several shades, ranging from pure violet through deep red and warm gray tones. This glass is used in astronomical work to produce sharp absorption bands to calibrate spectral lines.
The element is also a component of didymium, used in welders and glass blowers safety goggles. Some neodymium salts are also used to color enamels. It also makes up nearly 20 percent of mischmetal, a material that is used to make flints for lighters.
(Source: Los Alamos National Laboratory)
 Live Science Plus
Live Science Plus