2 women earn Chemistry Nobel Prize for gene-editing tool CRISPR

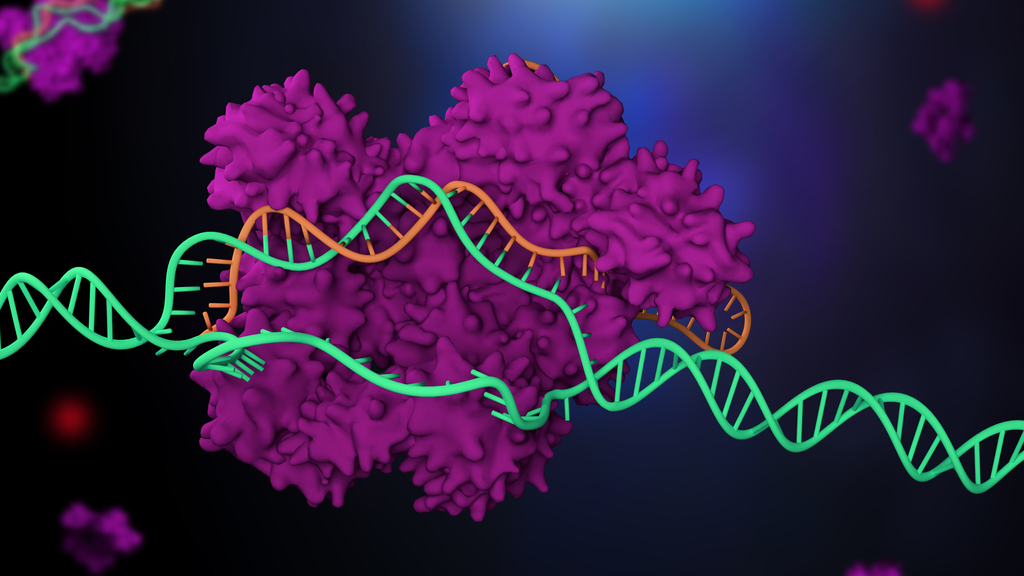
The 2020 Nobel Prize in chemistry went to two women who developed a gene-editing tool called CRISPR-Cas9, which snips DNA like a pair of molecular scissors.
The technique "has not only revolutionized basic science, but also resulted in innovative crops and will lead to ground-breaking new medical treatments," Claes Gustafsson, chair of the Nobel Committee for Chemistry, said in a statement. With the ability to deftly slice specific DNA sequences from the genome, scientists can pinpoint the functions of genes; these discoveries both add to our basic understanding of how those genes work and can have practical applications, such as for growing drought- and pest-resistant crops and developing therapies for cancer and genetic disorders. The genetic cut-and-paste system is also being used in new COVID-19 diagnostic tests.
The Nobel "for the development of a method for genome editing" went to Emmanuelle Charpentier, director of the Max Planck Unit for the Science of Pathogens, and Jennifer Doudna, a professor of biochemistry, biophysics and structural biology of the University of California, Berkeley. This is the first science Nobel to be awarded to an all-female team, according to Science Magazine.
Related: Nobel Prize in Chemistry: 1901-Present
The development of CRISPR-Cas9 began serendipitously when Charpentier was studying the bacteria Streptococcus pyogenes, which causes a range of diseases from tonsillitis to sepsis, according to a statement from the Nobel Committee. The bacteria contain a molecule called tracrRNA, Charpentier discovered, which protects S. pyogenes against infection by viruses, according to a 2011 report in the journal Nature.
It turned out that tracrRNA was just one component in a larger defense mechanism known as the CRISPR/Cas system, which bacteria use to slice and dice the DNA of viruses that try to infect them, Live Science previously reported. After a viral attack, the bacteria incorporate a piece of viral DNA into their own genome; these battle trophies line up in the genome, appearing repeatedly, and are known as "clusters of regularly interspaced short palindromic repeats," abbreviated as CRISPR. These archived genes are thought to help bacteria recognize the viruses and ward off future attacks.
But to first slice up the viral DNA, bacteria use "CRISPR-associated" proteins, called Cas proteins, under the guidance of tracrRNA and other molecules.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Related Content
After her discovery of tracrRNA, Charpentier began collaborating with Doudna, and the two recreated the bacteria's genetic scissors in a test tube. In their seminal work, published in 2012 in the journal Science, they simplified the system into a handy gene-editing tool, capable of targeting and snipping specific DNA sequences from the genome. The tool has since been further refined and used for a wide range of applications, including the recent development of diagnostic tests for COVID-19, BBC reported.
"This discovery, originally derived from a natural defense mechanism in bacteria against viruses, will have untold applications in treating and curing genetic diseases and fighting cancer, as well as impacts on agricultural and other areas," Luis Echegoyen, president of the American Chemical Society, said in a statement. "The future for this technique is indeed bright and promising."
Some scientists expected that biochemist Feng Zhang of the Broad Institute might share the Nobel with Charpentier and Doudna, as shortly after their discovery, he demonstrated that CRISPR also works in mammalian cells, Science Magazine reported. Based on Zhang's work, the Broad Institute received the first patent for the use of CRISPR gene-editing technology in eukaryotes — complex cells with nuclei to hold their DNA — but Charpentier's and Doudna's institutions continue to fight for their own patents, according to The Scientist Magazine.
Originally published on Live Science.

Nicoletta Lanese is the health channel editor at Live Science and was previously a news editor and staff writer at the site. She is a recipient of the 2026 AHCJ International Health Study Fellowship, with a project focused on antibiotic stewardship practices in Japan and the U.S. They hold a graduate certificate in science communication from UC Santa Cruz and degrees in neuroscience and dance from the University of Florida. Beyond Live Science, Lanese's work has appeared in The Scientist, Science News, the Mercury News, Mongabay and Stanford Medicine Magazine, among other outlets. Based in NYC, she also remains involved in dance and performs in local choreographers' work.