These vegetarian ants have steak knives for teeth, new study finds
Teeth are laced with zinc.


A leafcutter ant (Atta cephalotes) uses its metal-laced mandibles to cut through a leaf.
What do leafcutter ants and scores of middle-school students have in common? A mouth full of metal-laced teeth.
Tiny arthropods such as ants, spiders and scorpions routinely bite, sting or otherwise pierce tough material like wood and skin. It's a remarkable feat, given that humans have trouble chewing through so much as a piece of beef jerky (let alone a hunk of tree bark), even with our strong jaw muscles.
But new research has shed light on what gives one group of leafcutter ants (Atta cephalotes) their biting edge. Using powerful microscopes, scientists have discovered a web of zinc atoms woven into the biological structure of the ants' jaws, lending them the durability of a set of stainless steel knives, the researchers said. This smooth distribution of zinc allows the edge of the ant's teeth to form a fine point — and it keeps them sharp for a long time.
Related: In photos: Trap-jaw ant babies grow up
"The tiny animals who had this material, their muscles are microscopic compared to ours," Robert Schofield, a biophysicist at the University of Oregon and lead author of the study, told Live Science. The trick, he said, is that ants and other metal-mouthed arthropods leverage their sharp chompers to apply precisely the right amount of cutting force to slice through leaves or hide.
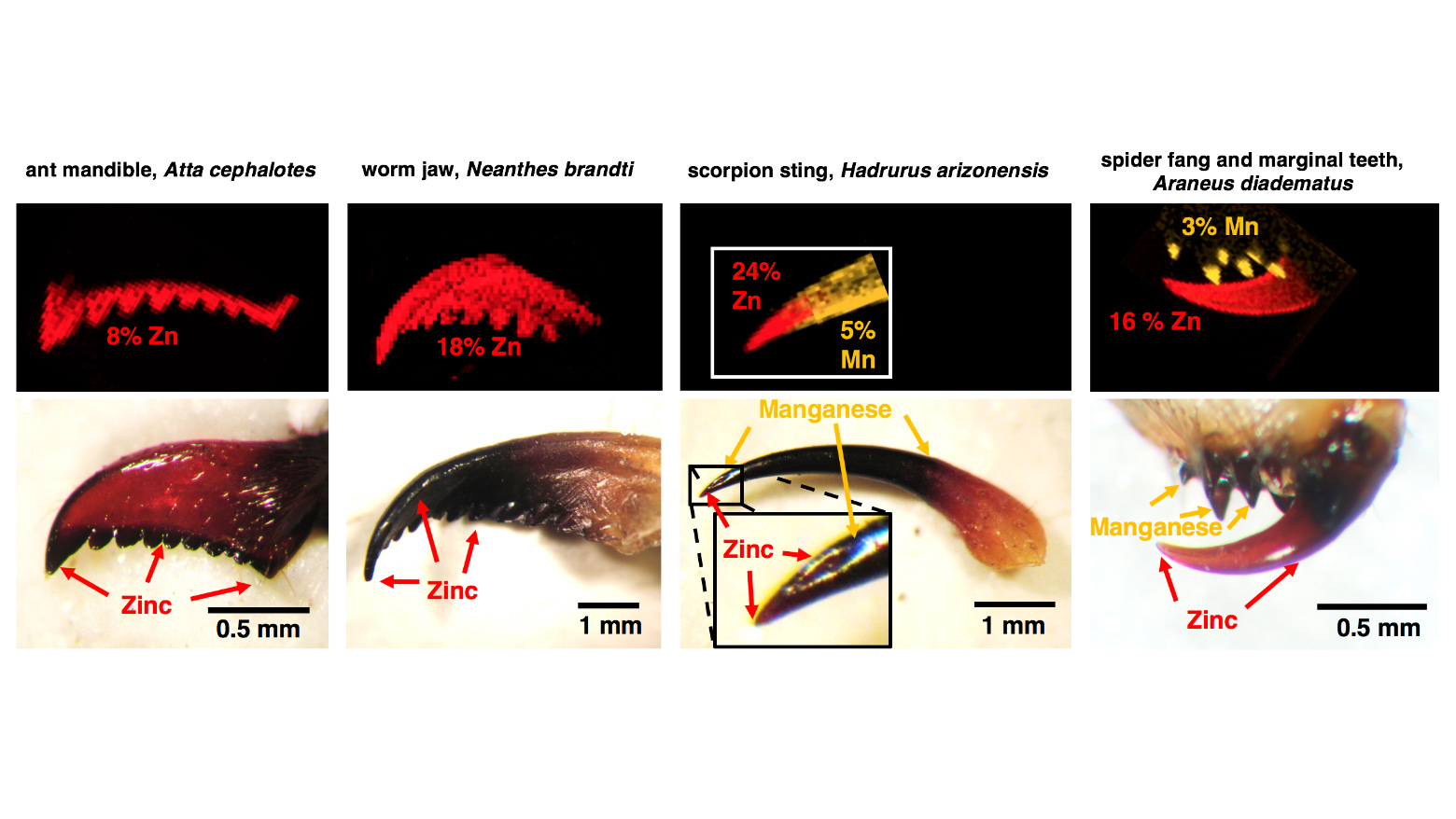
Metals such as zinc (shown in red) and manganese (yellow-orange) are found in the cutting and piercing parts of leaf cutter ants, worms, scorpions and spiders.
Schofield and his team knew from prior research that ant teeth contained a lot of zinc. But they didn't know exactly how those metal atoms were arranged, and how that helped the ants' bite. By examining the material makeup of leafcutter ant teeth under an ion beam microscope before and after biting, the researchers were able to calculate the hardness, sharpness and durability of the teeth.
An ant's jaw, or mandible, differs quite a bit from yours. "Ants don't rely much on mandibles to process food," said Cristian Klunk, an ecologist at the Federal University of Parana, Brazil, who was not involved with the study. But they do use them for pretty much every other task, from defense to home renovation, and so they need to keep them in tip-top shape.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Your teeth are covered in a layer of enamel, a calcium-rich material that is the hardest substance in the human body. If you looked at a bit of enamel under an electron microscope, you would notice calcium and phosphate molecules forming a chunky crystal matrix around carbon, hydrogen, and oxygen atoms. Those crystals are what keep teeth strong — but they're also what prevent them from being razor-sharp.
In contrast, the tiny, serrated "teeth" lining the inside edge of an ant's mandible are coated in a smooth blend of proteins crisscrossed with zinc. This material, known as a "heavy element biomaterial" (HEB), easily matches human tooth enamel for strength. It also makes an ant's tooth much better for slicing and dicing, since the blocky calcium phosphate crystals found in enamel can't form extremely sharp edges — that would be like trying to fashion a knife "out of chunks of gravel", Schofield said. Zinc, however, does not form blocky crystals; instead it stays evenly distributed throughout the protein mixture. That fine consistency allows for the sharp edges of the teeth.
RELATED CONTENT
Metallic reinforcements don't stop with ant teeth. Other invertebrates also weave zinc or a similar metal, manganese, into their tiny tool kits. Schofield and his team found that giant clam worms pack jaws infused with up to 18% zinc. Similarly, scorpion stings and spider fangs employ a mix of zinc and manganese atoms to ensure that these slender, needle-like structures can puncture tough flesh without breaking.
Schofield and his team calculated that the addition of zinc or manganese to an invertebrate's exoskeleton reduced the amount of force they needed to pierce through tough material by 60% on average. "Because the zinc is more resistant to wear," said Schofield, "after a while, it becomes a huge difference."
The research was published Sept. 1 in the journal Scientific Reports.
Originally published on Live Science.

Joanna Thompson is a science journalist and runner based in New York. She holds a B.S. in Zoology and a B.A. in Creative Writing from North Carolina State University, as well as a Master's in Science Journalism from NYU's Science, Health and Environmental Reporting Program. Find more of her work in Scientific American, The Daily Beast, Atlas Obscura or Audubon Magazine.

 Live Science Plus
Live Science Plus




