Science history: Doctor hypothesizes that 'transmissible proteins' can cause disease, contradicting a 'central dogma' of molecular biology — April 9, 1982
Prion diseases, such as "mad cow," are caused by transmissible proteins that were identified in the 1980s.
Milestone: Prions identified
Date: April 9, 1982
Where: San Francisco
Who: Dr. Stanley Prusiner
On April 9, 1982, a doctor at the University of California, San Francisco, published a paper in the journal Science showing that infectious proteins caused a degenerative nerve disease in sheep. In doing so, he transformed our understanding of how some diseases are transmitted.
Dr. Stanley Prusiner had been studying the disease scrapie, which affects sheep and goats. It had long been a mystery exactly how scrapie was transmitted from animal to animal. He had also long been haunted by seeing a patient die rapidly of the degenerative brain disorder Creutzfeldt-Jakob disease (CJD) a decade earlier. The patient had no fever or other signs of immune system activation nor any evidence of a bacterial or viral infection. Yet Prusiner was told the patient was infected by a "slow virus," or one that progressed on longer time scales than is typically seen with viruses.
"At that time, I was beginning a residency in neurology and was most impressed by a disease process that could kill my patient in two months by destroying her brain while her body remained unaffected by this process," he said in a speech he later gave about his discovery.
In the 1950s, the term "slow virus" had been coined to describe the diseases scrapie in sheep and goats. By the 1960s, scientists had started applying the term to certain human disorders, noting that the disease "kuru" that ravaged the Fore tribe in Papua New Guinea seemed to be transmitted when tribal members ate the brains of those who previously died of the disease.
Research in chimpanzees in the 1960s definitively showed that Creutzfeld-Jakob disease — a fatal, relentless brain disease that seemed to run in families — could also be transmitted by feeding the chimpanzees brain tissue from affected animals. Under a microscope, brain tissues affected by kuru, scrapie or CJD all looked remarkably similar, showing a characteristic "spongiform" appearance. In other words, the brain tissue became riddled with holes, like a sponge.
Yet there was a puzzle: CJD seemed to be passed on in families. So how could viruses or bacteria be both heritable and infectious?
Prusiner was initially studying CJD but switched his focus to scrapie when he looked at data from a team led by Tikvah Alper, a radiobiologist. Alper had found that scrapie could still be transmitted when infected tissue was irradiated with ultraviolet light, which damages DNA.
Get the world’s most fascinating discoveries delivered straight to your inbox.

Dr. Stanley Prusiner discovered the existence of prions.
So Prusiner began studying scrapie in mouse spleens and brains. But he quickly switched to hamsters because they developed disease symptoms within 70 days, as opposed to one to two years for mice. He then systematically worked to isolate and identify the chemical nature of the "infectious agent" driving the disease.
Ultimately, he pinpointed a protein as the culprit.
"Six separate and distinct lines of evidence show that the scrapie agent contains a protein that is required for infectivity," Prusiner wrote in the seminal 1982 study. All of those showed that breaking down the protein structure short-circuited the transmission of scrapie. He went on to show that there was no evidence for any nucleic acids, such as DNA or its cousins, in the samples. He proposed the name "prion" to describe the infectious protein, which he suggested could "code for its own biosynthesis," adding that "this hypothesis contradicts the 'central dogma' of molecular biology."
Prusiner's proposal was not widely accepted at first. But over the next 15 years, scientists elucidated the protein structure of prions and showed that they could take multiple conformations, even when encoded by the same DNA sequence. Researchers also showed how the prion's shape resisted degradation,and that it could "convert" the healthy versions of the protein into the pathological form.
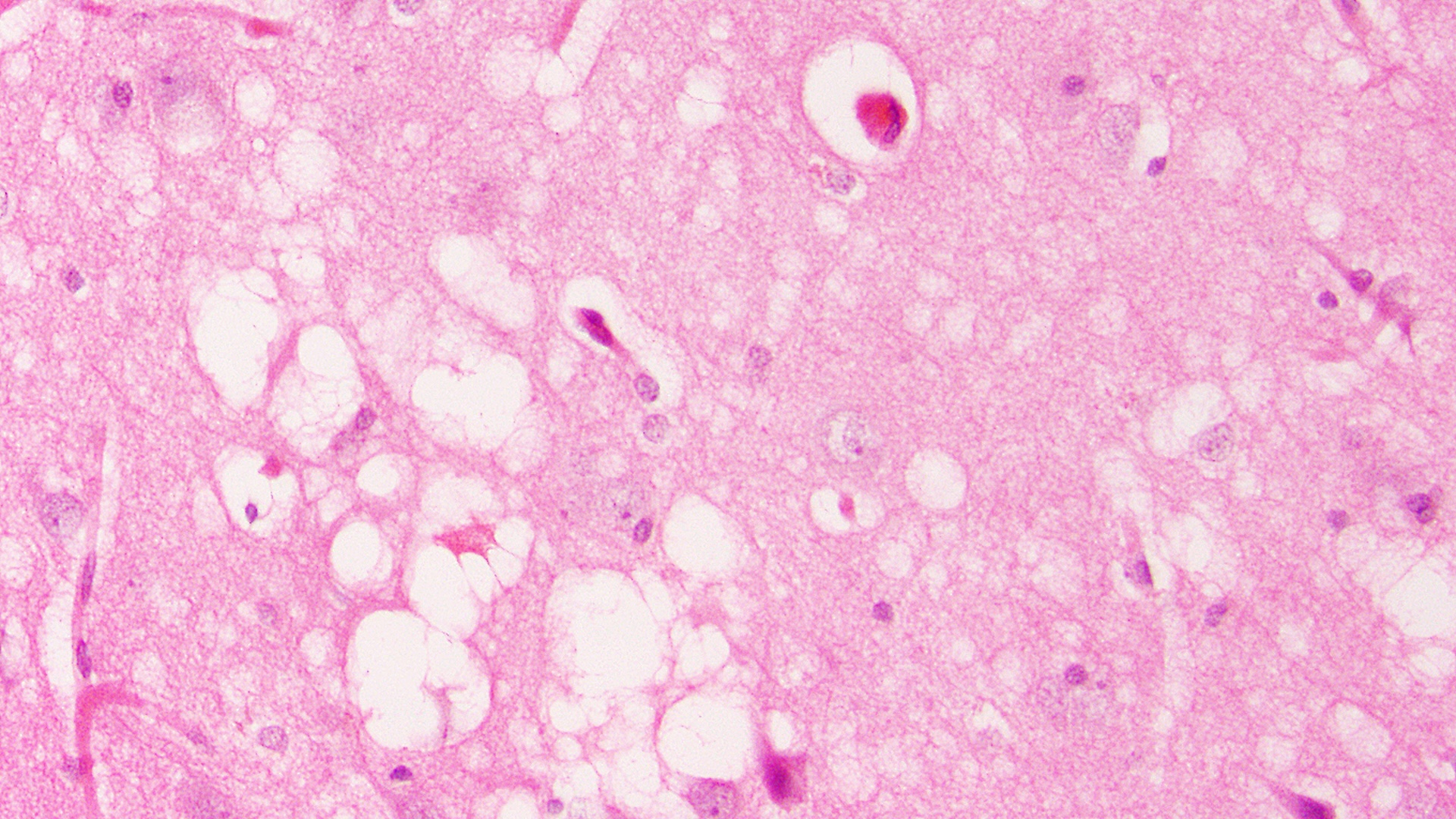
A microscopic image of human brain tissue. The holes in the brain (white spots) are considered the "trademark" of prion diseases.
Follow-up work in familial cases of CJD showed certain genes could also predispose people to the disease and that DNA damage determined how long it took for the disease to incubate.
Prusiner won the Nobel Prize in physiology or medicine in 1997 for his work on prions.
Prusiner's hypothesis was validated when the mad cow disease epidemic struck the U.K. in the early 2000s. Scientists would eventually determine that people became infected after eating beef from cows that had been fed the brain tissue of cows sick with bovine spongiform encephalopathy (BSE). After consuming meat from cows with BSE, humans develop a version of CJD known as "variant CJD."

Tia is the editor-in-chief (premium) and was formerly managing editor and senior writer for Live Science. Her work has appeared in Scientific American, Wired.com, Science News and other outlets. She holds a master's degree in bioengineering from the University of Washington, a graduate certificate in science writing from UC Santa Cruz and a bachelor's degree in mechanical engineering from the University of Texas at Austin. Tia was part of a team at the Milwaukee Journal Sentinel that published the Empty Cradles series on preterm births, which won multiple awards, including the 2012 Casey Medal for Meritorious Journalism.
You must confirm your public display name before commenting
Please logout and then login again, you will then be prompted to enter your display name.

 Live Science Plus
Live Science Plus





