New immunotherapy could make blood more 'youthful,' mouse study hints
In mice, a new type of immunotherapy appeared to partly turn back the clock of "immune aging."

Scientists reversed some signs of immune aging in mice with a new treatment that could one day potentially be used in humans.
The new immunotherapy works by disrupting a natural process by which the immune system becomes biased towards making one type of cell as it ages.
The mouse study is an "important" proof-of-concept, but it's currently difficult to gauge the significance of the findings, Dr. Janko Ž. Nikolich-Zugich, a professor of immunobiology at the University of Arizona who was not involved in the research, told Live Science in an email. More work is needed to see how well the therapy shifts the immune system into a more youthful, effective state.
All blood cells, including immune cells and the red blood cells that carry oxygen around the body, start life as hematopoietic stem cells (HSC) in the blood and bone marrow, the spongy tissue found within certain bones. HSCs fall into two main categories: those destined to become so-called myeloid cells and those that will develop into lymphoid cells.
Myeloid cells include red blood cells and immune cells belonging to our broadly reactive first line of defense against pathogens, including cells called macrophages that trigger inflammation. Lymphoid cells include cells that develop a memory of germs, such as T and B cells.
As we age, the HSCs slated to become myeloid cells gradually increase in number and eventually outnumber the lymphoid stem cells. This means we can't respond to infections as well when we're older as when we're young, and we're more likely to experience chronic inflammation triggered by increasing levels of myeloid cells that trigger inflammation.
Get the world’s most fascinating discoveries delivered straight to your inbox.
In the new study, published Wednesday (March 27) in the journal Nature, scientists developed an antibody-based therapy that selectively targets and destroys the myeloid HSCs, thus restoring the balance of the two cell types and making the blood more "youthful." The antibodies latch onto the targeted cells and flag them to be destroyed by the immune system.
The authors injected the therapy into mice aged 18 to 24 months, or roughly the equivalent of being between 56 and 69 years old as a human.
They then extracted HSCs from the mice after treatment and analyzed them, revealing the rodents had a smaller percentage of the myeloid HSCs than untreated mice of the same age.
This effect lasted for two months. Compared with untreated mice, the treated mice also produced more naive T cells and mature B cells. These cells can go on to form memory cells, which are directly involved in the immune attack; in the case of the B cells, they can form antibody-producing plasma cells.
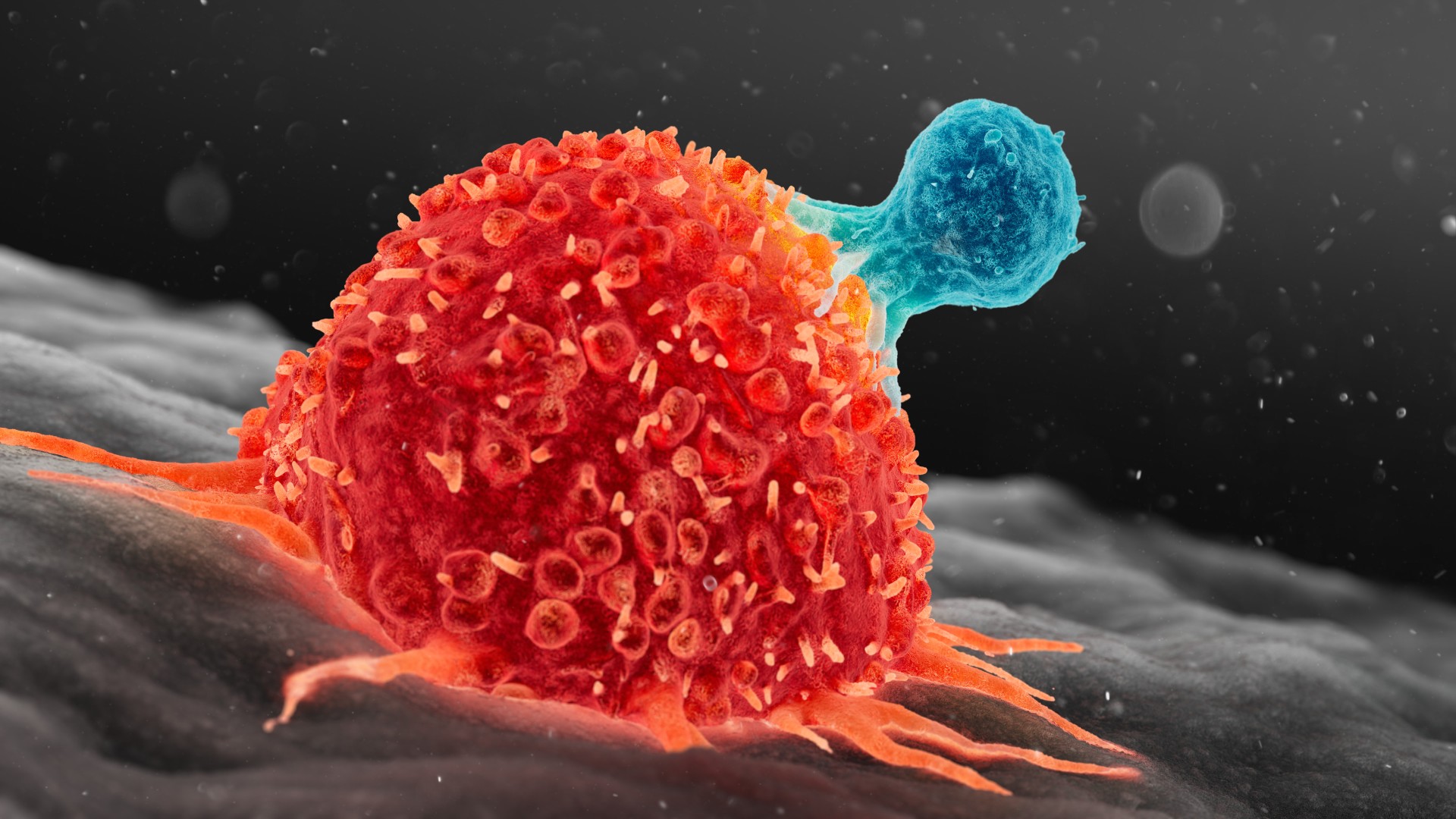
Lymphoid cells include T cells of the immune system, such as the one illustrated above attacking a cancer cell.
"Not only did we see a shift toward cells involved in adaptive immunity, but we also observed a dampening in the levels of inflammatory proteins in the treated animals," Dr. Jason Ross, lead study author and postdoctoral researcher at Stanford University, said in a statement. Specifically, the researchers saw that the levels of one proinflammatory protein fell in the treated mice. This protein, called IL-1beta, is mainly made by myeloid cells.
Eight weeks post-treatment, the researchers vaccinated the mice against a virus they'd never been exposed to before. The mice that had received the immunotherapy had more apt immune responses to vaccination than the untreated mice, producing more T cells against the germ.
"We believe that this study represents the first steps in applying this strategy in humans," Ross said. However, other experts have cautioned against jumping to conclusions.
Nikolich-Zugich noted that, although the researchers measured changes in the numbers of naive T cells in the mice, they didn't look at the function of the organ that makes them: the thymus. The team also saw reductions only in IL-1beta and not other inflammatory proteins. They also didn't test whether the mice's baseline immunity to new infections could be improved with this therapy, without vaccination, he said.
RELATED STORIES
Furthermore, the study didn't consider potential long-term side effects of the treatment, such as anemia, or a deficiency in red blood cells, said Dr. Ilaria Bellantuono, a professor in musculoskeletal aging at the University of Sheffield in the U.K. who was not involved in the research.
Although an "interesting" study, more work is needed to understand whether it can bring "meaningful changes" in the immune system, Bellantuono told Live Science in an email, whether that of mice or humans.
Ever wonder why some people build muscle more easily than others or why freckles come out in the sun? Send us your questions about how the human body works to community@livescience.com with the subject line "Health Desk Q," and you may see your question answered on the website!

Emily is a health news writer based in London, United Kingdom. She holds a bachelor's degree in biology from Durham University and a master's degree in clinical and therapeutic neuroscience from Oxford University. She has worked in science communication, medical writing and as a local news reporter while undertaking NCTJ journalism training with News Associates. In 2018, she was named one of MHP Communications' 30 journalists to watch under 30.