Key to Tardigrades' 'Superpowers' Identified in Their DNA
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
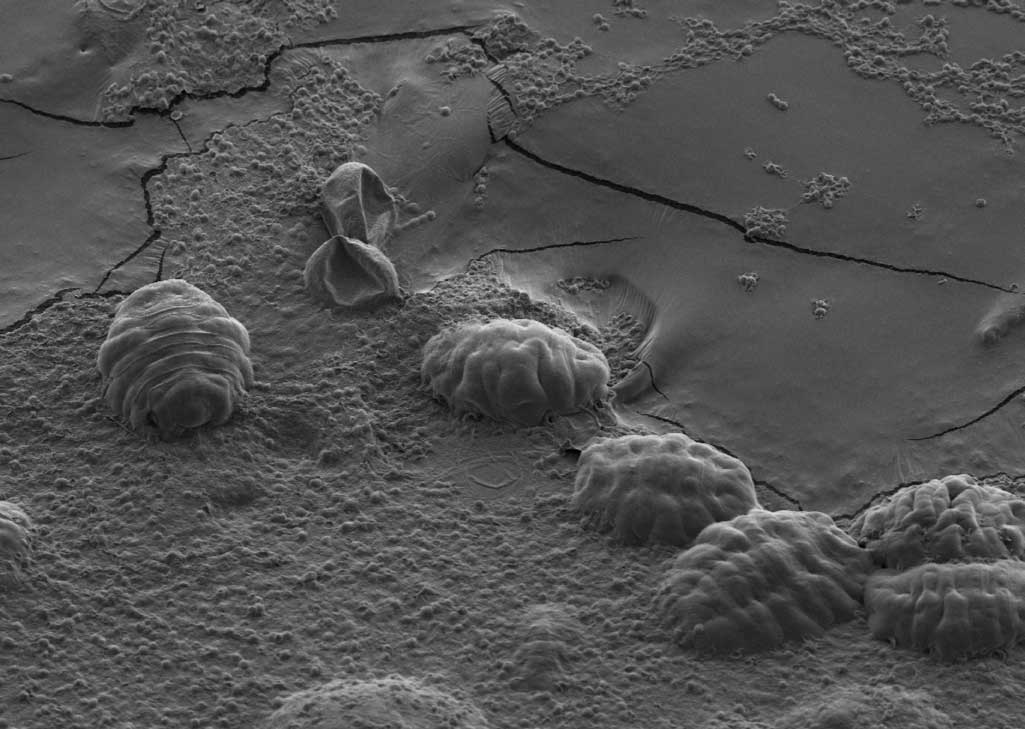
Microscopic, blobby-bodied tardigrades — also known as "water bears" — are famed for their ability to survive in extreme conditions, even appearing to come back from the dead.
But although tardigrades' hardiness has intrigued scientists for over 250 years, the mechanisms that enable these animals to recover after drying out for a decade or longer have remained a mystery.
Now, a new study reveals that special proteins coded into tardigrade DNA may be the secret to the creatures' resuscitation superpowers. [It's Alive! 'Water Bears' Revived After 30+ Frozen Years | Video]
Article continues belowTardigrades measure between 0.002 and 0.05 inches (0.05 to 1.2 millimeters) in length, so they can't be seen with the naked eye. The wee beasties have endearing features: fat, segmented bodies; four pairs of chubby legs tipped with grasping claws that resemble those of a sloth; and rotund heads with a circular mouths.
Tardigrades live on damp moss and algae around the world. Researchers have found that tardigrades can withstand searing heat and freezing cold, up to 300 degrees Fahrenheit (149 degrees Celsius) and as low as minus 328 degrees Fahrenheit (minus 200 degrees Celsius). Tardigrades can even emerge unscathed after exposure to boiling, high pressure, and the radiation and vacuum of space.
The creatures survive by expelling the water from their bodies and entering a suspended state known as a "tun." During this state, they retract their limbs and shrink into tiny, desiccated balls, emerging only when life-threatening conditions have passed. But scientists have wondered how that was possible, particularly for tardigrades that spend a decade or more as dried-out tuns.
Tardigrade-specific proteins
Previously, a sugar called trehalose was thought to be the key to tardigrade regeneration. This sugar is found in other types of animals and in plants, and is known to play a role in tolerance to dry conditions. However, prior studies of tardigrade biochemistry found little evidence that these animals have trehalose, suggesting that the sugar isn't the main driver for tardigrade recovery.
Get the world’s most fascinating discoveries delivered straight to your inbox.
In the new study, researchers analyzed tardigrade genetic activity as the micro-animals dried out. First, the scientists identified which genes were highly active at the time, and then the researchers looked closely at what those genes do.
Results showed that certain genes were expressing a type of protein unique to tardigrades, which the scientists dubbed tardigrade-specific intrinsically disordered proteins, or TDPs. In some species of tardigrades, the genes that produce TDPs were active all the time, while in other species, these genes were activated only under certain conditions.
TDPs protected the tardigrades in much in the same way that trehalose protects other animals, by forming glass-like structures that help to preserve cells that are in a dehydrated state.
The tardigrade species that had a constant supply of TDPs was more successful at recovering from drying out than the species that weren't always producing TDPs, the researchers wrote.
"We think it can do this because it has so many of these proteins around already and doesn't need time to make them," study lead author Thomas C. Boothby, a Life Sciences Research Foundation Postdoctoral Fellow at the University of North Carolina, said in a statement.
The findings reveal that biological methods used to tolerate environmental stress and withstand desiccation are more diverse than suspected, the researchers said. Now that the role of TDPs in tardigrade resuscitation has been identified, other uses for the proteins could be found, such as protecting crops that are vulnerable to drought, or preserving perishable medications, Boothby said in a statement.
"Being able to stabilize sensitive pharmaceuticals in a dry state is very important to me personally," Boothby said. "I grew up in Africa, where lack of refrigeration in remote areas is a huge problem. These real-world applications are one of the things that led me to study tardigrades."
The findings were published online March 16 in the journal Molecular Cell.
Original article on Live Science.

Mindy Weisberger is a science journalist and author of "Rise of the Zombie Bugs: The Surprising Science of Parasitic Mind-Control" (Hopkins Press). She formerly edited for Scholastic and was a channel editor and senior writer for Live Science. She has reported on general science, covering climate change, paleontology, biology and space. Mindy studied film at Columbia University; prior to LS, she produced, wrote and directed media for the American Museum of Natural History in NYC. Her videos about dinosaurs, astrophysics, biodiversity and evolution appear in museums and science centers worldwide, earning awards such as the CINE Golden Eagle and the Communicator Award of Excellence. Her writing has also appeared in Scientific American, The Washington Post, How It Works Magazine and CNN.
 Live Science Plus
Live Science Plus









