Beetle Sperm Teams Up To Navigate Females' Bodies
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
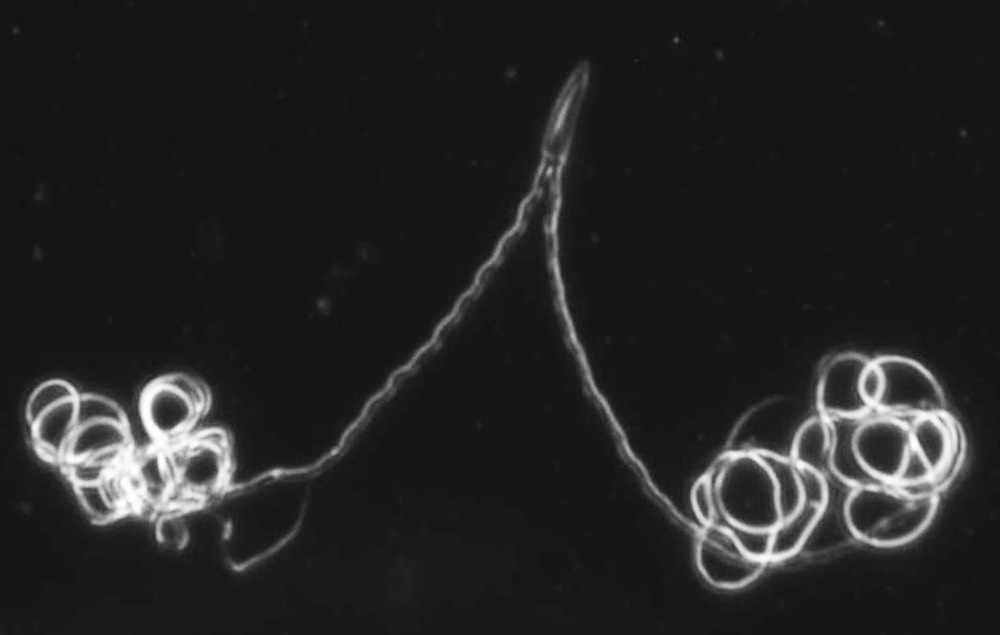
The sperm of the male diving beetle is seriously strange: Instead of swimming in the female reproductive tract on their own, individual sperm cells often stick together in pairs, in clusters and even in long chains of hundreds or thousands.
Now, a new study finds this weird sperm behavior is driven by the evolution of female diving beetles. When female reproductive tracts evolve into ever more labyrinthine paths, the research finds, male sperm have to evolve to catch up.
"When you look at the intricate morphology of the reproductive tracts, you can't help but think that sperm needs Swiss army knives and compasses to make it through there," study researcher Scott Pitnick, a biologist at Syracuse University in New York, said in a statement. "The females make it really complicated."
Article continues belowWeird sex
Diving beetles are tiny but deadly predators, at least if you're a tadpole or baby fish. More than 4,000 species live in freshwater spots around the world. Even in deserts of Arizona, where study researcher Dawn Higginson works as a postdoctoral researcher at the University of Arizona's Center for Insect Science, the beetles thrive in cattle water tanks. [10 Secret Weapons of Bugs]
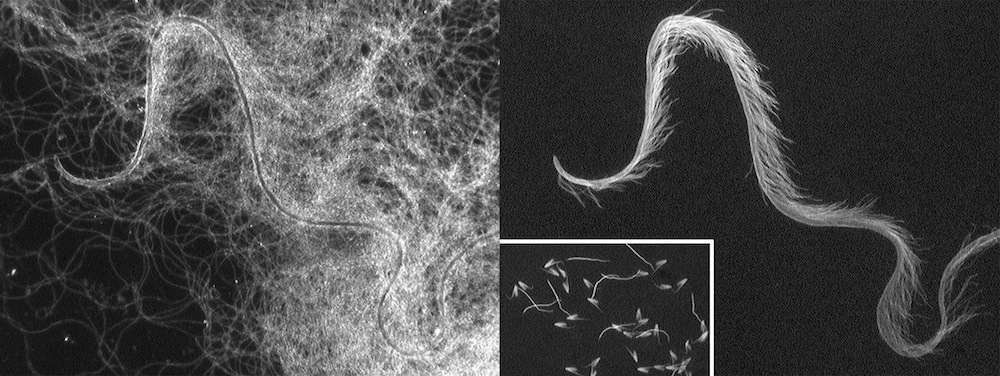
Like many insects, female diving beetles have complex reproductive tracts, and they can store up sperm from one or more males to self-fertilize months or even years later. Higginson and her colleagues knew that diving beetles had complex reproductive tracts. They also knew from earlier studies that diving beetle sperm cells could pair up in the female reproductive tract, sticking themselves together. But investigating the sperm-storage sacs of female beetles, Higginson turned up something even stranger: whole masses of sperm cells clumped together into one single conjugate.
"I could see the tails beating and moving the whole conjugate around," Higginson said in a statement. "As far as sperm goes, this is clearly unlike anything we have ever seen before."
Get the world’s most fascinating discoveries delivered straight to your inbox.
In some species, sperm stacked their heads into little pockets behind the heads of other sperm, much like you might stack drinking glasses in a kitchen cabinet. The resulting sperm trains can stretch hundreds or thousands of sperm long.
Keeping up
To find out what drives this weird cellular behavior, Higginson and her colleagues investigated the female and male reproductive tracts of 42 species of diving beetle. They looked at factors including sperm length, length of the sperm head, length of sperm tails, number of conjoined sperm and shape of sperm heads. In females, they examined the length of the reproductive ducts as well as the size of the sperm-storage areas. They then used statistical methods to tease out relationships between male and female reproductive systems.
The results revealed that ancestral diving beetle females had simple reproductive tracts. But for reasons unknown, the female tracts evolved to be more and more complex. Sperm, in turn, had to change to thrive in this new environment.
"We can reconstruct what the ancestors of these diving beetles looked like," Higginson said. "We find they had conjugated sperm and rather compact female reproductive tracts. The female morphology undergoes evolutionary change, and then sperm compensates for that."
It could be that the females evolve in an attempt to catch only the best sperm. If that were the case, the cycle would work a bit like the evolution of the peacock tail: Females prefer a particular trait and evolve to preferentially reproduce with males that have that trait (for example, a flashy tail or wild sperm). Males with that trait pass on more of their genes, so the trait gets amplified through the generations.
"We can't say from this study that the males are catching up but it's suggesting there could be an arms race," Higginson said.
The female evolution could also be the result of something unrelated to males, Higginson said. For example, perhaps these complex reproductive systems are better for handling fertilized eggs.
"The point is that the females are driving the sexual evolution," Higginson said. "The sperm cells just have to keep up."
You can follow LiveScience senior writer Stephanie Pappas on Twitter @sipappas. Follow LiveScience for the latest in science news and discoveries on Twitter @livescience and on Facebook.

Stephanie Pappas is a contributing writer for Live Science, covering topics ranging from geoscience to archaeology to the human brain and behavior. She was previously a senior writer for Live Science but is now a freelancer based in Denver, Colorado, and regularly contributes to Scientific American and The Monitor, the monthly magazine of the American Psychological Association. Stephanie received a bachelor's degree in psychology from the University of South Carolina and a graduate certificate in science communication from the University of California, Santa Cruz.
 Live Science Plus
Live Science Plus