Trippy! Psychedelic Zebrafish Reveal How Cells Regenerate
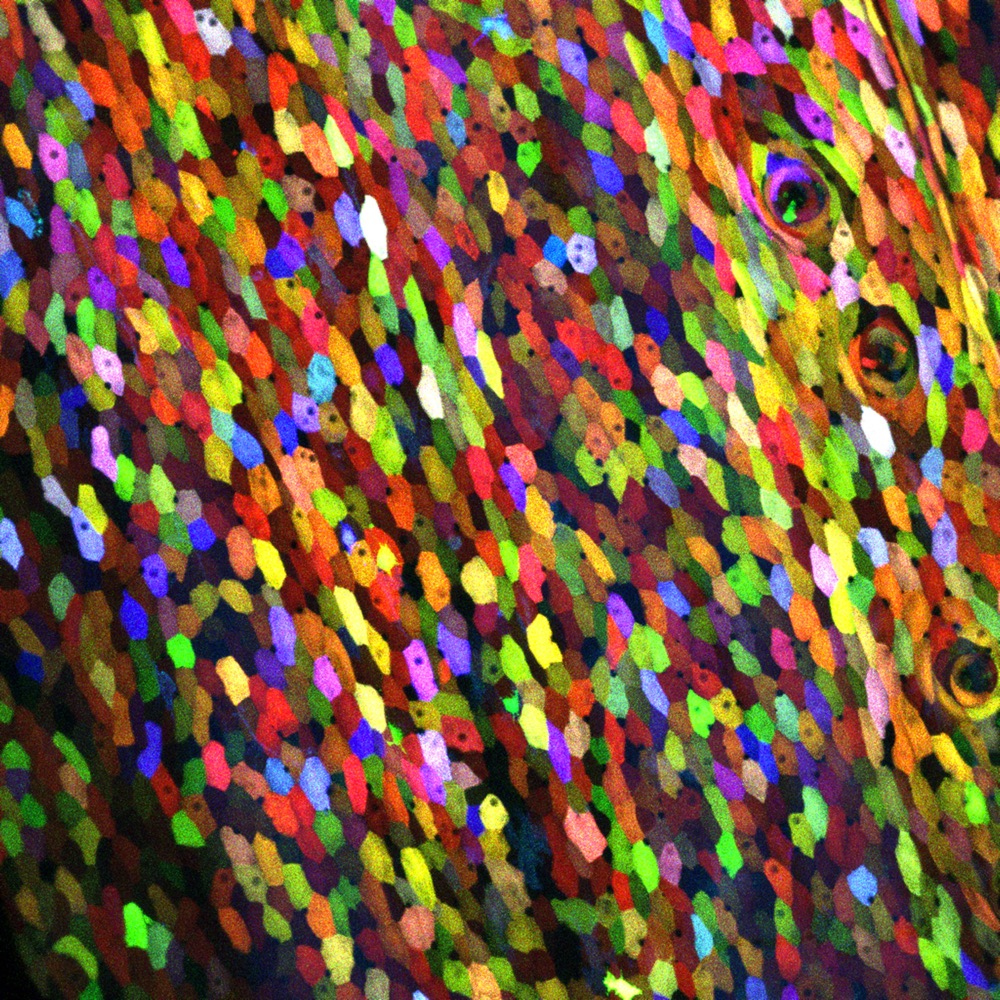
In what looks more like a post-impressionist painting than a scientific achievement, a transgenic zebrafish is revealing how hundreds of its cells regenerate in a bouquet of colors.
Scientists genetically programmed every cell on the zebrafish's body surface, including its cornea, scales, fins and barbels, to express a unique combination of green, red and blue fluorescent proteins, according to lead scientist Kenneth D. Poss, a cell biology professor at Duke University. The result: images of a technicolored zebrafish — revealing 70 different hues — that could just as easily hang in an art museum as appear in a scientific journal.
The various colors, which could be distinguished under a microscope, were expressed in the skin's outermost layer and stayed intact for the cell's lifetime. [Dazzling Images of the Brain Created by Neuroscientist-Artist]
By calculating the number of combinations of red, green and blue proteins, Nature.com estimates the zebrafish's individual cells could light up in any one of 5,000 different hues (even though they all weren't distinguishable under a microscope).

Even with the 70 distinguishable colors, researchers could watch how the zebrafish's skin cells behaved and regenerated.
"It is like you have given each cell an individual bar code," lead study author Chen-Hui Chen, a postdoctoral fellow in Poss' lab, said in a statement. "You can precisely see how individual cells collectively behave during regeneration."
Using their custom software, lead scientist Poss and his colleagues monitored how the cells behaved over three weeks of normal skin turnover. They also watched how the cells reacted to injuries, ranging from minor skin exfoliation to the major amputation of a fin.
Get the world’s most fascinating discoveries delivered straight to your inbox.
After the fin amputation, skin cells below the cut were recruited to cover up the wound.
"Within a few hours of fin amputation, the exposed stump tissue is quickly covered by epithelial cells. These initial events lead to formation of a multilayered wound epidermis that is capable of releasing signals important for new bone regeneration," the researchers wrote in the March 21 issue of the journal Developmental Cell.
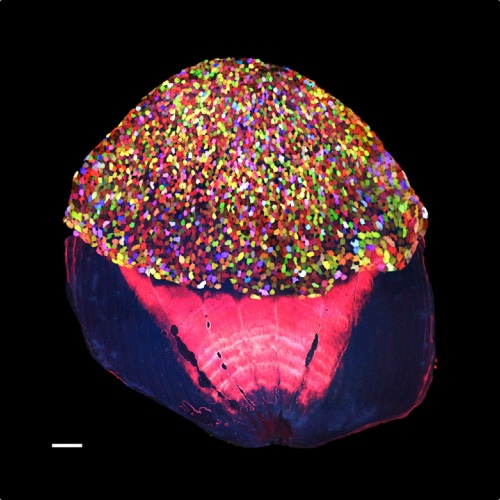
And within two to three weeks, the appendage was restored, as pre-existing and newly regenerated cells mixed to create new tissue, the researchers said.
The technology, called Skinbow, was derived from a similar one developed by Harvard scientists in 2007 for the brain, called Brainbow, in which individual neurons were tagged with particular colors.
"Before we can fully understand tissue regeneration, we need to be able to monitor what individual cells are doing," Poss said. "This is a cutting-edge way to visualize hundreds or thousands of cells at once in a regenerating tissue."
Follow us @livescience, Facebook & Google+. Original article on Live Science.
Jeanna Bryner is managing editor of Scientific American. Previously she was editor in chief of Live Science and, prior to that, an editor at Scholastic's Science World magazine. Bryner has an English degree from Salisbury University, a master's degree in biogeochemistry and environmental sciences from the University of Maryland and a graduate science journalism degree from New York University. She has worked as a biologist in Florida, where she monitored wetlands and did field surveys for endangered species, including the gorgeous Florida Scrub Jay. She also received an ocean sciences journalism fellowship from the Woods Hole Oceanographic Institution. She is a firm believer that science is for everyone and that just about everything can be viewed through the lens of science.

 Live Science Plus
Live Science Plus





