New antibiotic that slays superbugs discovered in 'dark matter' microbes from North Carolina soil
Deadly superbugs are unlikely to develop resistance to a new antibiotic produced by bacteria, scientists say.

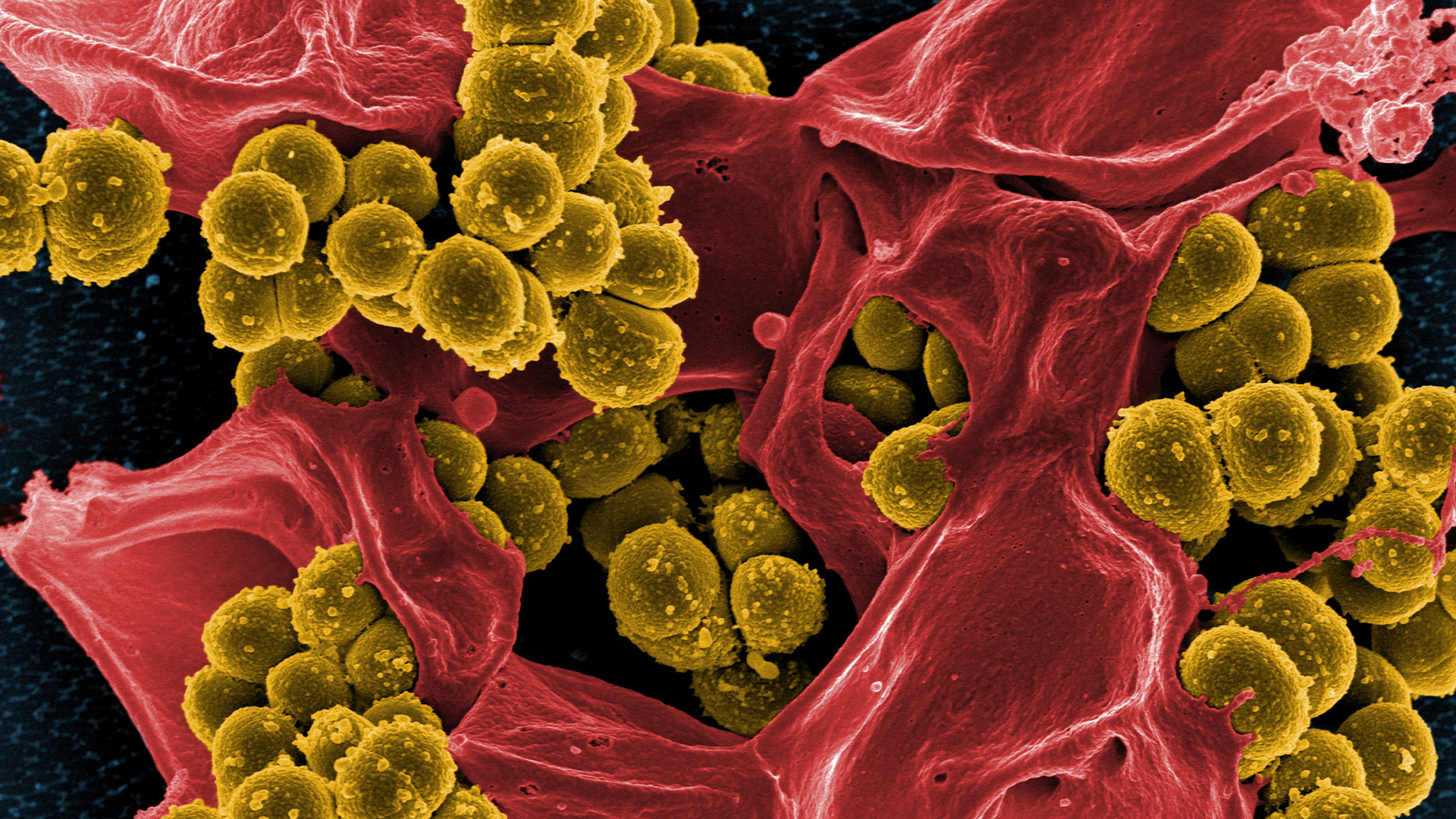
Methicillin-resistant Staphylococcus aureus, or MRSA, is a potentially deadly superbug that can be killed with a newfound antibiotic, according to an early study of the drug.
A new antibiotic made by lab-grown bacteria can kill "superbugs" without driving the germs to become more resistant to treatment, an early study suggests.
Scientists isolated the antibiotic, called clovibactin, from a bacterium called Eleftheria terrae subspecies carolina that they collected in soil samples from North Carolina. Their research, published Aug. 22 in the journal Cell, addresses the urgent need for new antibiotics that kill bacteria in new ways.
It also highlights the promise of studying bacteria that were hard to culture until recently, study co-author Markus Weingarth, a biochemist at Utrecht University in the Netherlands, told Live Science.
"Clovibactin is a chemically novel antibiotic that comes from what we call the 'dark matter' of bacteria — bacteria that have not been grown in the lab before," Weingarth said. "It's nontoxic in animal models and works better than the gold-standard antibiotic vancomycin," which is used to treat bacterial infections that show resistance to other drugs, yet has started to become ineffective against some strains of bacteria.
Related: Scientists invent 'shape-shifting' antibiotic to fight deadly superbugs

These jars contain precursors to bacterial cell walls that University of Bonn researchers analyzed for the new study. These precursors include the structures targeted by the newfound antibiotic clovibactin.
Many antibiotics destroy bacteria by disrupting the formation of their cell wall, a mesh-like structure that surrounds each bacterial cell. Existing antibiotics tend to do this by interfering with proteins called enzymes that help build the cell wall, but bacteria can evolve and alter these enzymes, rendering those drugs ineffective.
"Most of the antibiotics we find these days are similar to already existing antibiotics, and that's a problem, because bacteria can easily develop resistance to them," Weingarth said. Antibiotic resistance is a growing threat that, worldwide, directly resulted in roughly 1.3 million deaths and may have contributed to 3.65 million more in 2019.
Get the world’s most fascinating discoveries delivered straight to your inbox.
It was previously difficult to grow E. terrae subspecies carolina because, to survive, it needs specific nutrients and symbiotic microbes in the soil where it grows, Weingarth said. But researchers have developed a device that can recreate the conditions required to grow the elusive microbe in its original soil, he said.
This enabled researchers at NovoBiotic Pharmaceuticals, who are co-authors on the paper, to grow the bacterium and discover clovibactin. Weingarth and additional collaborators then worked on determining how the antibiotic works.
The team found that clovibactin can kill two dangerous superbugs: methicillin-resistant Staphylococcus aureus, or MRSA, and Enterococcus faecalis bacteria that are resistant to vancomycin. MRSA can cause life-threatening infections when it enters the body through wounds or during surgery, and E. faecalis causes a variety of infections, including urinary tract infections.
In another experiment, the scientists exposed S. aureus to low levels of clovibactin for three days, and the microbes didn't develop resistance to the drug.
The inability of S. aureus bacteria to resist clovibactin may come down to the unique way the drug kills them: It targets undecaprenyl-pyrophosphate, a chemical group found in three fat molecules that form the building blocks of bacterial cell walls. Clovibactin surrounds these molecules like a cage, which is why its name derives from "Klouvi," the Greek word for "cage."
RELATED STORIES
As bacteria cannot easily modify these building blocks without destroying their cell walls, it would be very hard for them to develop resistance to clovibactin, Weingarth said. "If the bacterium does manage to modify one of the target points, it still dies because there are still two more points of attack," he said.
IV injections of clovibactin were also more effective than IV vancomycin in reducing S. aureus levels in mice infected with the bacteria. Although no side effects were seen in the mice, further studies in humans are needed to confirm how well the antibiotic works and if it is safe, Weingarth said. As human cells do not have cell walls, clovibactin shouldn't kill them, he said.
"We urgently need alternative approaches to antibiotic discovery, and this creative and exciting new work helps push the field in the right direction," César de la Fuente, a professor at the University of Pennsylvania who uses AI to discover new antibiotics, told Live Science. It could also be beneficial to chemically tweak clovibactin to kill bacteria even more effectively before testing it in humans, he added.
Editor's note: This story was updated on Aug. 31, 2023 to clarify what various co-authors contributed to the study. The story was first published on Aug. 30.

Carissa Wong is a freelance reporter who holds a PhD in cancer immunology from Cardiff University, in collaboration with the University of Bristol. She was formerly a staff writer at New Scientist magazine covering health, environment, technology, nature and ancient life, and has also written for MailOnline.

 Live Science Plus
Live Science Plus




