Cancer's Spread May Depend on Weird, Newfound Fluid Physics
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
The spread of tumors and other growing tissues has revealed a whole new type of physics.
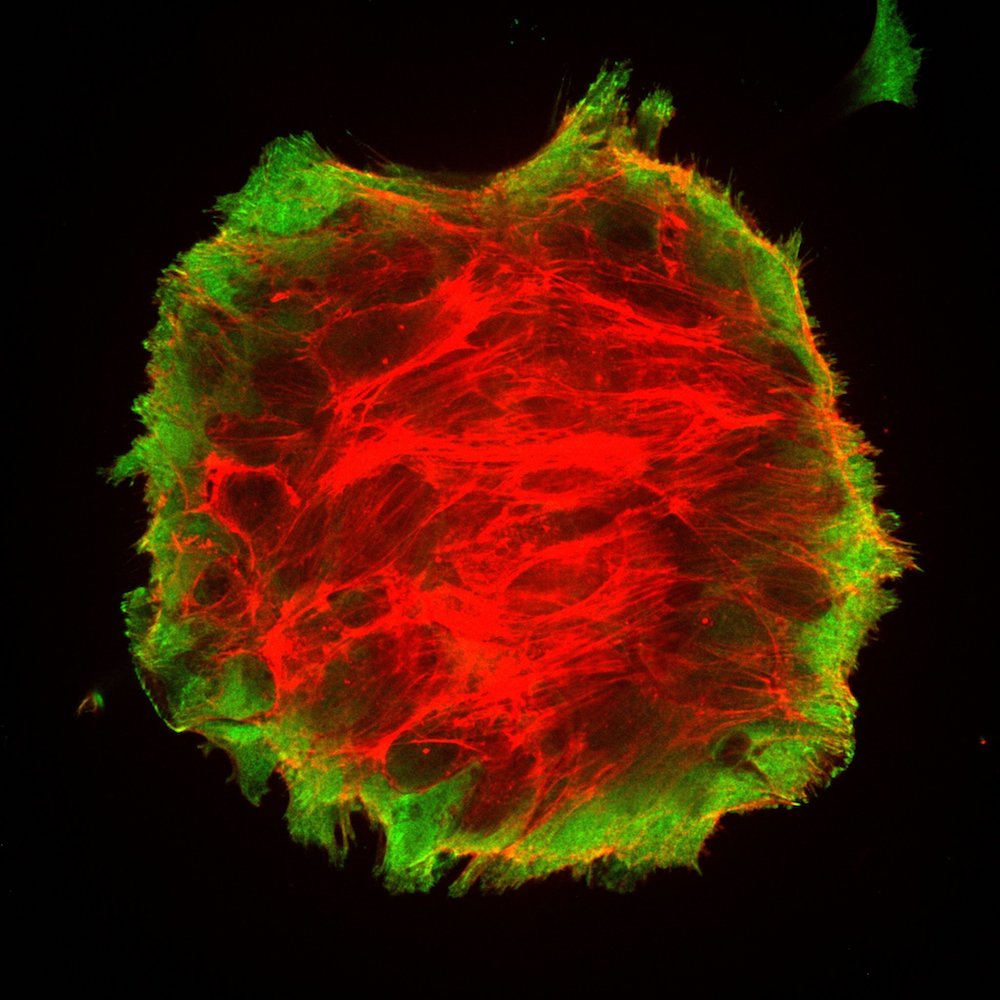
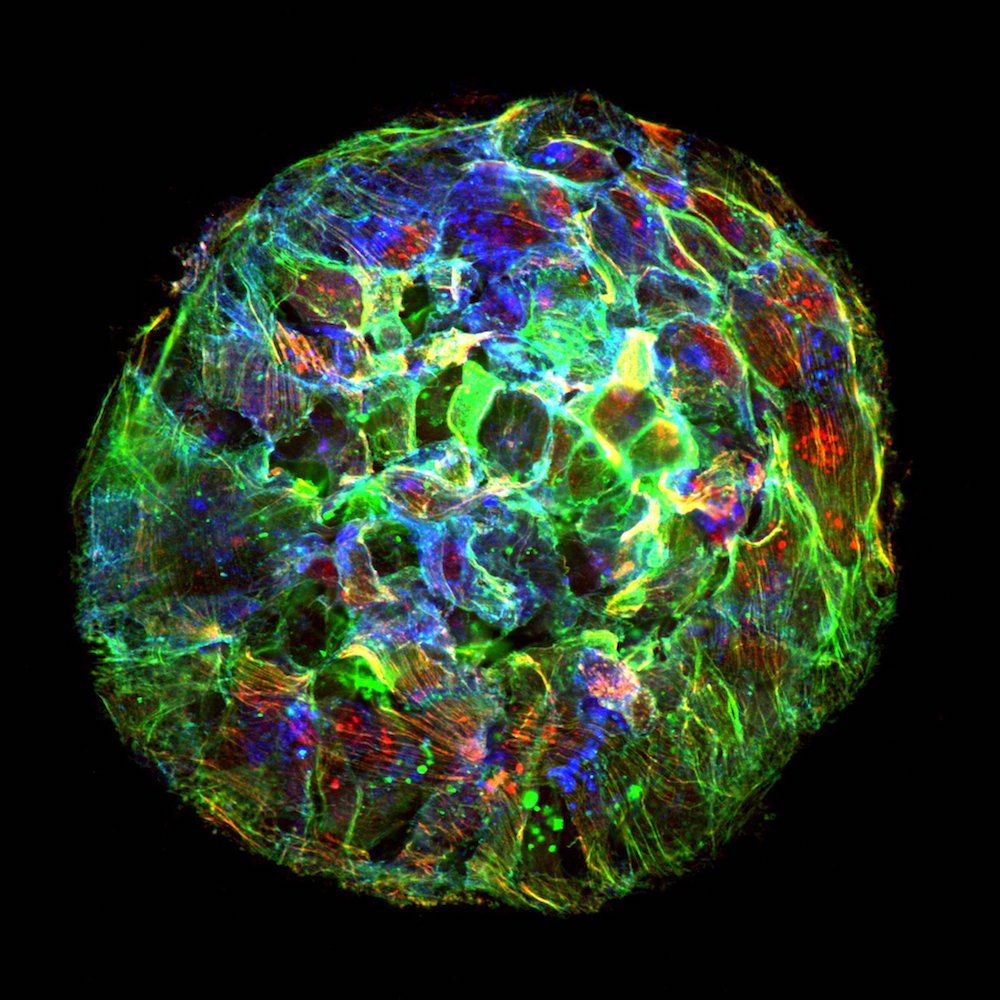
In new research, published Sept. 24 in the journal Nature Physics, scientists found that living cells transition from 2D sheets to 3D blobs by a previously unknown process called "active wetting." And the physics of active wetting may be able to explain why and how cancers spread.
"If we could find the way to selectively modify these forces in a real tumor, which is a very hard task, we could design a treatment to avoid cancer dissemination," study co-authors Xavier Trepat, of the Institute for Bioengineering of Catalonia in Spain, and Carlos Pérez-González, of the Universidad de La Laguna in Spain, told Live Science in an email. [10 Do's and Don'ts to Reduce Your Risk of Cancer]
Active physics
Any sort of medical application for the findings is a long way off. Trepat and Pérez-González said that their next steps will involve delving further into the weird physics of active wetting, about which little is yet known.
What the researchers have found is based on experiments done in a lab dish using human breast cancer cells. It all started, Trepat and Pérez-González said, with an investigation into a protein called E-cadherin, which provides adhesion between cells. The researchers had wanted to know how this protein regulates the tension within tissues, or groups of cells. What they didn't expect was that the tension within the tissue could get so high that their sheet of tissue would spontaneously detach from the collagen-coated gel they were using as a substrate and retract into a spheroid shape.
"The first time we observed this phenomenon, we were not sure about how or why it was happening," the researchers told Live Science.
The researchers contrasted active wetting with the behavior of so-called passive fluids, in which there are no living structures to alter fluid flow. Normally, in passive fluids, a set of physics equations known as the Navier-Stokes equations dictates the fluid dynamics. In passive fluids, the transition from 2D sheet to 3D spheroid is called dewetting. The opposite, a 3D spheroid spreading out into two dimensions, is called wetting. Whether wetting or dewetting happens is governed by the surface tension of the interface, the liquid and the gas involved.)
Get the world’s most fascinating discoveries delivered straight to your inbox.
But as the researchers played with the cancer cells in their experiment — varying parameters like tissue size and E-cadherin levels — they found that the cells weren't behaving like regular fluids do in passive wetting and dewetting. This is because a number of active processes, from the contractility of the tissue to the cell-substrate adhesion, determine if the cells ball up or spread out, the researchers found.
The transition between the spread-out wetting phase and balled-up dewetting phase depends on competition between cell-cell forces and forces that attach the cell to the substrate, the researchers said.
Cancer transitions
Tissues grow and move in lots of ways, including during normal development. But the active wetting transition is important, because it is the key moment that cells go from a contained spherical to a spreading, flat sheet Trepat and Pérez-González said. In other words, once circular balls of tumor spread out and attach to a surface the tumor is able to spread further.
"Our results set up a comprehensive framework to understand which forces are important for cancer invasion," the investigators said. Part of the next phase of work will be to move the studies out of lab dishes and into living tissue and real tumors, the researchers added.
Biological systems can be hard to fit into classical physics frameworks, wrote Richard Morris and Alpha Yap in a comment accompanying the new paper. Morris is a postdoctoral researcher at the Tata Institute for Fundamental Research in India, and Yap is a cell biologist at the University of Queensland in Australia. But the new article is a "valuable step in the right direction" for making physics relevant to problems of biology, Morris and Yap wrote.
"In this case," they wrote, "we learn that, whereas ideas from classical physics can be beneficial in the characterization of biological systems, the analogy must not be pushed too far, and new approaches are needed."
Originally published on Live Science.

Stephanie Pappas is a contributing writer for Live Science, covering topics ranging from geoscience to archaeology to the human brain and behavior. She was previously a senior writer for Live Science but is now a freelancer based in Denver, Colorado, and regularly contributes to Scientific American and The Monitor, the monthly magazine of the American Psychological Association. Stephanie received a bachelor's degree in psychology from the University of South Carolina and a graduate certificate in science communication from the University of California, Santa Cruz.

 Live Science Plus
Live Science Plus