Beware Herbal Meds: Understudied Drugs Pose Risks, Docs Say
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
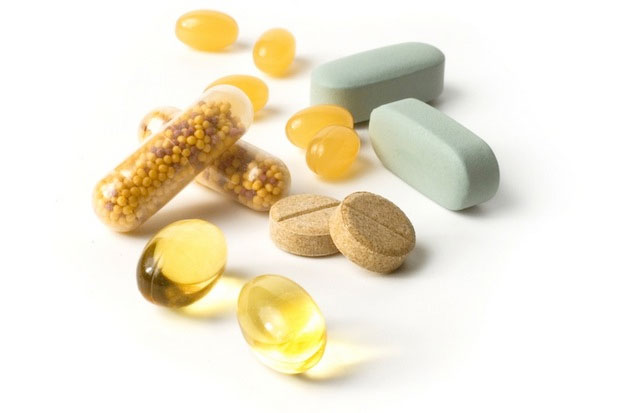
Herbal medications are often marketed as natural and safe compared to pharmaceutical drugs. But researchers warned today that herbal meds are typically under-researched and under-regulated.
And side effects from some herbal medications can be just as serious as the side effects from medications approved by the Food and Drug Administration (FDA), said Graziano Onder, a geriatrician at Catholic University of the Sacred Heart in Milan, Italy, who co-authored a new patient guide to alternative medicines, published in the Journal of the American Medical Association.
"Differently from 'conventional' medications, these medications are not tested in clinical studies," Onder wrote in an email to Live Science. "For this reason, clear knowledge on their efficacy and risks is lacking." [10 Medical Myths That Won't Go Away]
Article continues belowHerbal unknowns
In the United States, herbal supplements are regulated for quality standards, though there are questions as to how effective those regulations are. In February 2015, the New York attorney general released the results of a DNA investigation, which found that 80 percent of supplements tested did not contain the ingredients on the label. Some researchers dispute the testing used, but there is widespread agreement that quality control in the supplement business is lacking.
Efficacy testing, however, is not required at all, because supplements are exempt from the need to get FDA approval. High-quality research is rare, Onder and his colleague Rosa Liperoti, also of Catholic University of the Sacred Heart, wrote in JAMA, and few studies have shown benefits from herbal medications.
"Limited data support the use of cranberry for the prevention of recurrent urinary tract infections in women, but this evidence is not definitive, and more research is needed to confirm this effect," Onder said. "The efficacy of other commonly used herbal medications has not been proven."
Get the world’s most fascinating discoveries delivered straight to your inbox.
Some unproven treatments include St John's wort for depression, echinacea for colds, ginseng and ginkgo biloba for cognitive performance and garlic for improving cardiovascular health.
Weighing the risk
Meanwhile, the risks of some herbal medications can be severe. The kava kava root, meant to calm anxiety and promote sleep, has caused liver damage in some people, Onder said. Other side effects of herbal medications can include allergies, anaphylactic shock, nausea, vomiting, diarrhea and even seizures.
Herbal medications can also interfere with conventional medications that patients may be taking. Ginkgo biloba, for example, can increase the risk of bleeding if taken with a blood thinner like warfarin, or even aspirin.
Consumers often believe that natural means "healthy and safe," Onder said, but this is not true.
Protecting yourself
Onder and Liperoti recommended talking to a doctor before taking herbal medications, and making sure your doctor knows of any supplements you are using. All side effects should be reported to your physician, the authors wrote. Consumers can also report possible herbal medication side effects through the U.S. Safety Reporting Portal at www.safetyreporting.hhs.gov.
Pregnant and nursing women should not use herbal medications, because these supplements have not been tested in this population, the researchers said. Nor should children use herbal medications, the researchers wrote, because of a similar lack of testing. Age and health can influence the risk involved in taking herbal medications; older adults' bodies, for example, might be less able to eliminate supplements, which could lead to ingredients building up in the body, the researchers wrote.
While the FDA regulates herbal medications at a lower standard than it uses for conventional drugs, the European version of the agency (the European Medicines Agency) is taking steps to give consumers more information, Onder said. The Committee for Herbal Medicinal Products, established in 2004, is working to draft a list of herbal supplements that are not harmful under normal use.
Follow Stephanie Pappas on Twitter and Google+. Follow us @livescience, Facebook & Google+. Original article on Live Science

Stephanie Pappas is a contributing writer for Live Science, covering topics ranging from geoscience to archaeology to the human brain and behavior. She was previously a senior writer for Live Science but is now a freelancer based in Denver, Colorado, and regularly contributes to Scientific American and The Monitor, the monthly magazine of the American Psychological Association. Stephanie received a bachelor's degree in psychology from the University of South Carolina and a graduate certificate in science communication from the University of California, Santa Cruz.
 Live Science Plus
Live Science Plus









