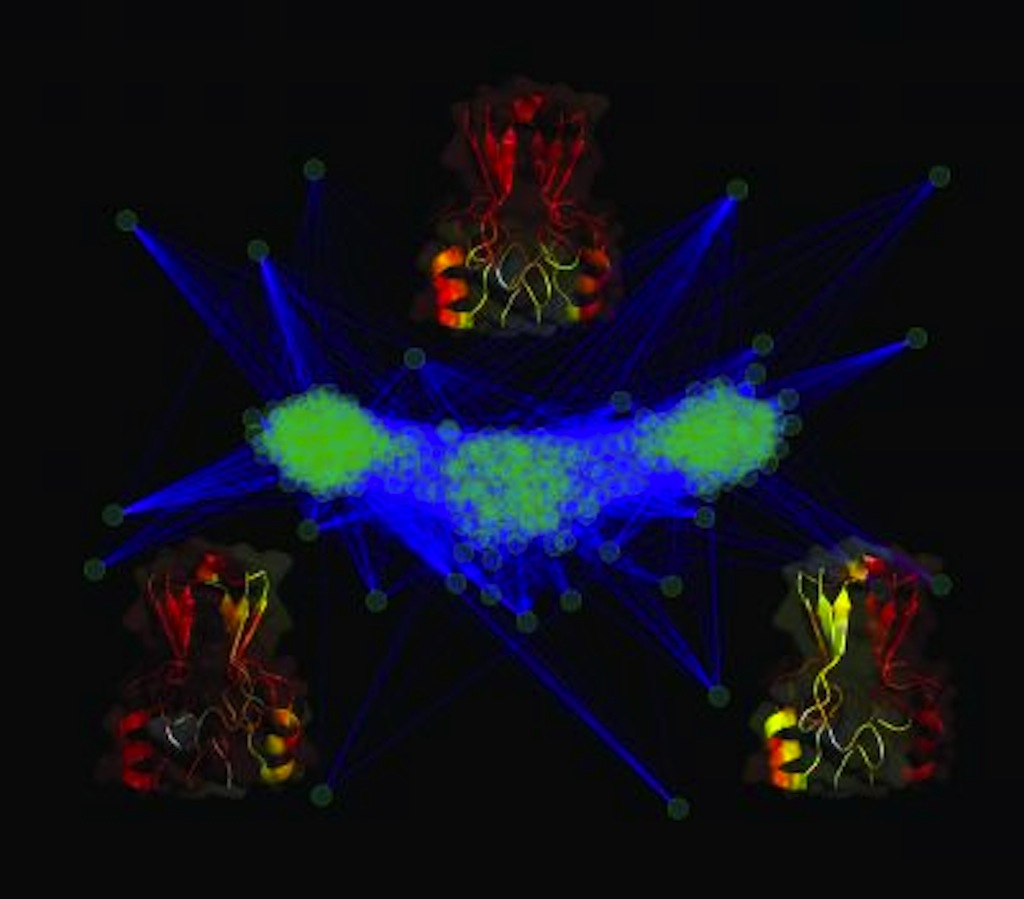
What might appear to be some sort of psychedelic spectacle is actually a cluster analysis of the protein MitoNEET. The protein could have the unique ability to bind and store iron-based molecules in the body and is known to sit on the wall of the mitochondria, an organelle found in cells. Iron is an essential element for life but can also be toxic in certain conditions.
Researchers from Rice University and the University of California, San Diego (UCSD) used laboratory experiments and computer modeling to better understand how the protein handles a potentially toxic payload of iron and sulfur.
"I think mitoNEET is a protein that could be your best friend or your worst enemy," said study co-leader Patricia Jennings, professor of chemistry and biochemistry at UCSD. "There's some evidence that it may act as a sensor for oxidative stress and that it can lose its toxic iron-sulfur cluster under stress conditions. Depending upon where the iron ends up, that could lead to drastic problems inside the cell."
Research has shown the proteins potentially toxic payload of iron-sulfur molecules faces the cell's cytoplasm, the gel-like fluid that fills the cell, and this cluster can be delivered into the mitochondria. A sister protein of mitoNEET also interacts with proteins during apoptosis, the way cells use to kill themselves when they are no longer viable.
Because proteins shapes can provide clues about function, the team used computer simulations to study how the protein folds and its shapes. In one shape two arms slightly intertwine and extend away. In the other, the arms also extend but are not intertwined.
"I think people forget that proteins are machines with moving parts," said study lead author Elizabeth Baxter, a UCSD graduate student who works under the guidance of both Onuchic and Jennings. "We start with the static snapshot and model in the functional motions."
Follow LiveScience @livescience, Facebook & Google+.
Get the world’s most fascinating discoveries delivered straight to your inbox.


 Live Science Plus
Live Science Plus






