The world's biggest clone is a 77-square-mile 'immortal' meadow of seagrass
The massive plant is around 4,500 years old but could continue expanding indefinitely.


A section of one of the seagrass meadows that make up the world's largest clone. Every blade belongs to the same plant.
Scientists have discovered the world's biggest clone in Australia: A massive network of seagrass meadows that covers more than 77 square miles (200 square kilometers). The network of meadows is actually one single plant that has been continually cloning itself for almost 4,500 years.
Researchers found the enormous clone while studying the genetic diversity of seagrasses in Shark Bay, a protected body of shallow water in Western Australia. They learned that almost all the region's meadows of Poseidon’s ribbon weed (Posidonia australis) are genetically identical. Further analysis revealed that unlike the other seagrasses in the area, which reproduce sexually, P. australis is actually cloning itself through an underground network of branching roots.
The P. australis clone stretches for around 112 miles (180 km) from end to end — albeit with a few patches — "making it the largest known example of a clone in any environment on Earth," the researchers wrote in the study, which was published online May 31 in the journal Proceedings of the Royal Society B. It dwarfs the previous record-holder: a clone of the seagrass Posidonia oceanica in the western Mediterranean, which spans around 9 miles (15 km).
"It is a single plant" that has been able to grow uninterrupted, senior researcher Elizabeth Sinclair, an evolutionary biologist at the University of Western Australia, told Live Science. If it remains undisturbed, the gigantic clone could continue to expand indefinitely, Sinclair added, making it practically immortal.
Related: 24,000-year-old 'zombies' revived and cloned from Arctic permafrost
Clonal expansion
The researchers found that the P. australis clone was expanding through a process known as "horizontal rhizome extension," in which a plant creates a genetically identical offshoot via an underground stem, or rhizome, which then develops its own roots and stem. When viewed from the surface — in this case, the sandy seafloor — the seagrass clumps look like separate specimens, but on a genetic level they are the same plant.
This is the same process that birthed Pando, a forest of quaking aspen trees (Populus tremuloides) in Utah that is actually just one massive, interconnected tree.
Get the world’s most fascinating discoveries delivered straight to your inbox.
While the P. australis meadows do not form a single unbroken meadow, they can still be considered to be one plant, Sinclair said. "Seagrass plants can fragment over time if there is damage or disturbance, but the fragments are still genetically identical," she added. It is possible that the P. australis meadows were once fully connected and may have been fragmented by grazing marine animals or extreme heat waves, the researchers wrote in the study.
Based on the size and age of the P. australis meadows, researchers suspect that the clone is growing at a rate of around 6 to 14 inches (15 to 35 centimeters) per year. This may not sound like much, but it's a fairly quick rate when compared with the growth of other clonal seagrass meadows, the study authors reported.

An aerial shot of a section of coastline in Shark Bay. The dark patches are all seagrasses.
Theoretically, the clone could continue to grow indefinitely, Sinclair said, "as long as it's not disturbed and the environment doesn't change too quickly." The near-pristine conditions in Shark Bay, which was designated in 1991 as a World Heritage Area by the United Nations Educational, Scientific and Cultural Organization (UNESCO), mean that P. australis has remained relatively undisturbed throughout its entire life, she added.
Genome duplication
The researchers suspect that another part of the P. australis success story can be attributed to an unusual genetic superpower among plants that enables them to make an additional copy of their genome, which doubles the amount of DNA they can utilize to adapt to extreme changes in environmental conditions.
Most organisms on Earth are diploids, which means their DNA contains a single pair of chromosomes. However, this is not the case for every organism. Some organisms, such as males of certain species of bees, have DNA that consists of single unpaired chromosomes, and these organisms are known as monoploids. Some organisms, known as polyploids, have two or more pairs of chromosomes.
Diploid plants can rapidly evolve into polyploids by doubling the number of chromosomes they have — a process known as whole-genome duplication, or polyploidy. The researchers suspect this is what happened to P. australis.
There are two ways that a diploid plant can become a polyploid. It can happen when two separate but closely related species reproduce. Rather than combining parental DNA like a standard hybrid does, polyploid offspring get an entire copy of each parent's DNA. This is known as allopolyploidy. Polyploids can also emerge when two individuals from separate populations of the same species reproduce, and the offspring gets both complete sets of DNA. This is known as autopolyploidy. In both cases, the process is completely random and offspring becomes a brand new species because it is unable to reproduce with other individuals from its parents' species.
In the case of P. australis, the researchers determined that the self-cloning seagrass likely emerged via autopolyploidy from a diploid ancestor that has likely since gone extinct.
Polyploid plants are sometimes regarded as "evolutionary dead ends" because many are sterile, meaning they cannot reproduce sexually, Sinclair said. This limits the plants' ability to mutate, which is a key part of the theory of evolution. However, becoming a polyploid may can act as a last chance for plants that are facing extinction due to extreme environmental changes.
"Combining two different genomes has essentially doubled the genetic diversity in the plant, likely increasing its ability to tolerate a wider range of environmental conditions," Sinclair said.
Changing conditions
Until around 8,500 years ago, Shark Bay was actually above sea level and part of continental Australia. But rising sea levels caused by the end of the Last Glacial Period, the most recent ice age that ended around 12,000 years ago, submerged that part of the continent. The newly created marine habitat was inundated with new species, such as seagrasses.
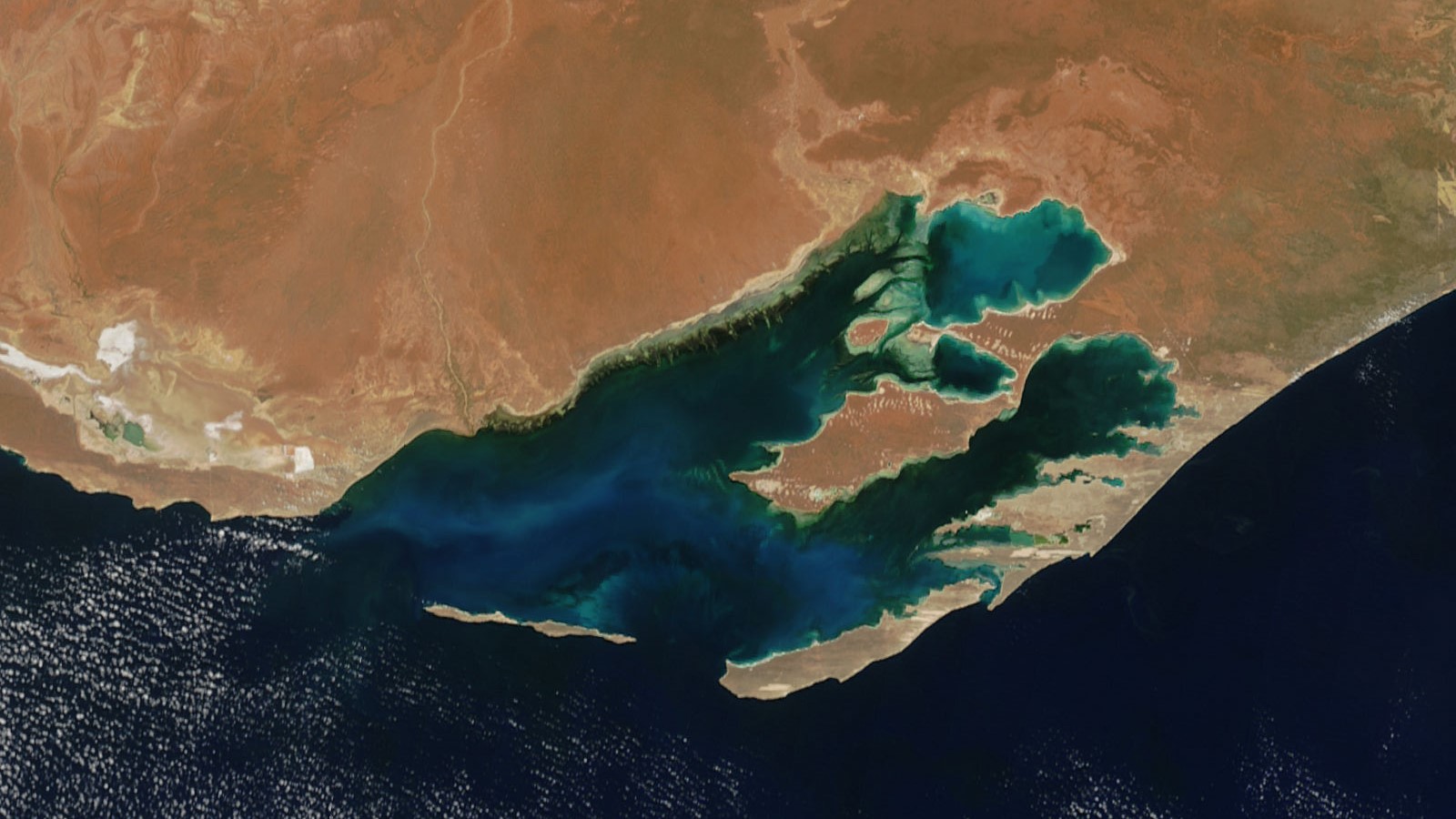
A satellite photo of Shark Bay in Western Australia taken Nov. 2004 by NASA's Terra satellite.
However, Shark Bay would have been a very unstable environment at the time because of its shallow water. Today, the average depth of Shark Bay is around 30 feet (9 meters) across the entire 8,880 square mile (23,000 square km) area, but it would have been even shallower around 4,500 years ago when P. australis emerged. Shallow oceans are more vulnerable to extreme shifts in temperature and salinity because there is less water to distribute and circulate heat and minerals. Their ecosystems are also more prone to disturbance and damage from tropical storms than deep-sea environments are.
In the study, the researchers suggested that if P. australis became polyploid before some type of extreme environmental upheaval during this more turbulent period, that gave P. australis an advantage over its diploid predecessors, which were unable to survive whatever change occurred.
Shark Bay continues to experience extreme conditions today to some degree. Annual temperatures can range between 63 and 86 degrees Fahrenheit (17 and 30 degrees Celsius), and the water is very salty. The bay's shallowness also means it is at risk from increasingly powerful heat waves caused by climate change, and is potentially susceptible to damage from cyclones. However, the environment is more stable than it was when P. australis first emerged.
RELATED STORIES
P. australis has likely continued to thrive in the area for millennia thanks to its resilience to environmental changes; other local seagrass species that continue to reproduce sexually, which is energetically expensive and requires lots of available space for new plants to grow, may be less resilient, Sinclair said. Without having to compete for space or divert energy to reproduction, P. australis has been free to clone itself at a steady pace and dominate the local ecosystem, she added.
Originally published on Live Science.

Harry is a U.K.-based senior staff writer at Live Science. He studied marine biology at the University of Exeter before training to become a journalist. He covers a wide range of topics including space exploration, planetary science, space weather, climate change, animal behavior and paleontology. His recent work on the solar maximum won "best space submission" at the 2024 Aerospace Media Awards and was shortlisted in the "top scoop" category at the NCTJ Awards for Excellence in 2023. He also writes Live Science's weekly Earth from space series.