Even Chemists Are Baffled by This GIF of a Droplet Spiraling to Its Doom
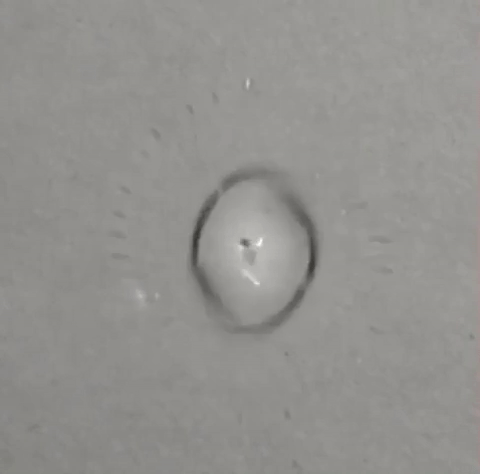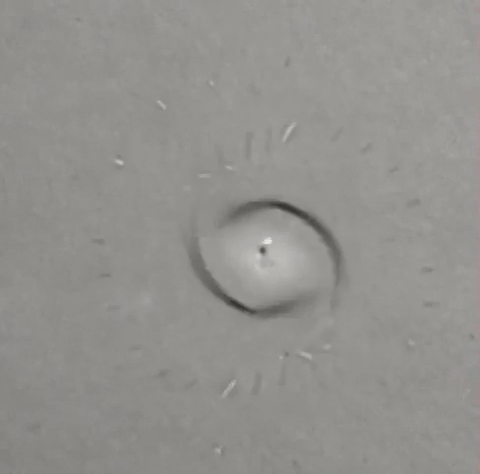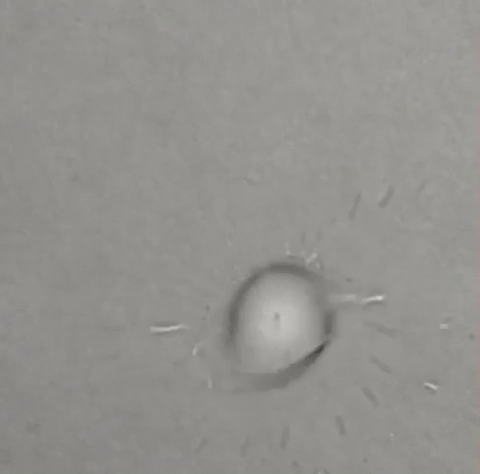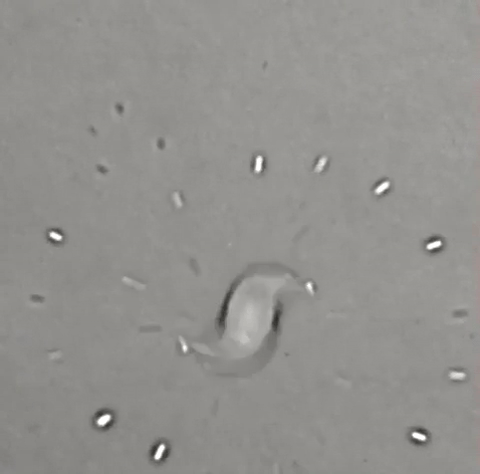
A single drop of solvent swirls like a tiny dancer atop a beaker of water, gradually jettisoning away little round bits of itself until nothing remains. Some who saw it thought it looked like a spinning galaxy, or the world's tiniest hurricane. All who saw it wondered what the heck was going on — and that includes the researchers who conducted the experiment in 2011.
The magical drop of solvent stars in a GIF called "A drop of dichloromethane on water spiraling out of existence as it evaporates," posted Thursday (Jan. 11) to Reddit's r/chemicalreactiongifs forum. Despite its newfound fame (more than 20 thousand upvotes within the first 24 hours), the GIF originated from a 2011 paper published in the German chemistry journal Angewandte Chemie.
The paper's thesis was simple: when you release a droplet of dichloromethane (DCM) solvent into a beaker of soapy water, it looks really, really cool. [Album: Prize-Winning Photos Taken Through a Microscope]
“This is a very easy experiment, and a very complicated phenomenon,” said Oliver Steinbock, a professor of chemistry at Florida State University, and the senior author on the study. "We were very surprised by it — and we still are.”
To set up the experiment, Steinbock and his fellow researchers filled several beakers with various concentrations of water and a common lab disinfectant called CTAB. Using a pipette, they added a single drop of DCM — a colorless liquid sometimes used as a degreaser — to each beaker, and filmed the results. Each trial took about 20-30 seconds total, and was visible with the naked eye.
So, what's going on here?
Each drop of DCM, which has a relatively low boiling point, began evaporating as soon as it left the pipette. But the surprises began when the droplets touched the soapy water solution.
Get the world’s most fascinating discoveries delivered straight to your inbox.
"DCM has a higher density than water, so you'd expect it to sink right away," Steinbock told Live Science. "But instead, as soon as it touches the water, part of it spreads out and creates this sort of film that holds the droplet on the surface of the water… it’s like a boat that holds the droplet afloat.” (Though the DCM film is not visible in the viral GIF above, you can see it clearly in several other videos of the experiment that Steinbock posted to YouTube.)
Despite this boat-like film, a small part of the droplet does begin to sink. It's not visible from the top-down vantage point of this GIF; however, a tiny jet of falling bubbles forms underneath the droplet once it touches the water. The falling DCM jet slowly shrinks the droplet's volume, but also causes it to spin. "It’s a little bit like when you flush a toilet," Steinbock said. "The water has a tendency to start to rotate and twist. And that triggers the rotation of the drop that we begin to see."
Within a few seconds, the droplet is at once floating, rotating and evaporating. As a result of these combined forces, smaller droplets eventually start disengaging from the edge of the larger droplet. But instead of sinking themselves, they shoot out radially, moving straight ahead over the surface of the film until they, themselves, evaporate.
"These droplets are self-propelled," Steinbock said. This is due to a phenomenon called the Marangoni effect, which states that a liquid with a high surface tension will pull more strongly than a liquid with a low surface tension. This difference in tension creates a force on the system that can lead to motion.
As the DCM in the experiment begins to evaporate, the droplet's surface tension lowers from the outside in. Smaller droplets begin to form at the large droplet's edge, until the relatively high surface tension of the surrounding water pulls the small droplets away in what Steinbock calls a "ballistic" trajectory. Each individual droplet moves straight ahead until its surface tension becomes equally unstable, leading to further fragmentation. Eventually, the droplets split so many times they can no longer be seen. (A 2017 paper in Physical Review Letters explains the phenomenon further.)
These and other forces continue spinning and shrinking the large DCM droplet until, suddenly, it loses its symmetry and sputters madly into total evaporation. Why the system suddenly goes from a state of apparent symmetry to total entropic chaos baffles even Steinbock and his fellow researchers. Across half a dozen experiments, they were unable to re-create the exact patterns seen in this GIF. "I was a little discouraged to understand how complicated it really is," Steinbock said.
However complicated, this little drop of solvent nevertheless spoke to something intrinsic and pure in many who saw it. As Reddit user MurderSlinky put it: "Never before have I related so much to a gif as I have to this tiny, insignificant dot of liquid spinning aimlessly in an endless, indifferent sea while slowly becoming nothing."
Originally published on Live Science.

Brandon is the space/physics editor at Live Science. His writing has appeared in The Washington Post, Reader's Digest, CBS.com, the Richard Dawkins Foundation website and other outlets. He holds a bachelor's degree in creative writing from the University of Arizona, with minors in journalism and media arts. He enjoys writing most about space, geoscience and the mysteries of the universe.
