Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
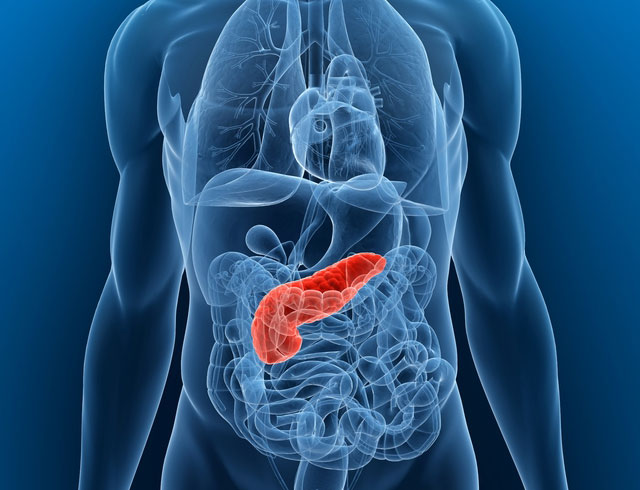
Human skin cells can be reprogrammed to produce the hormone insulin, which could one day help patients better control their diabetes.
However, the new cells, which researchers described Jan. 6 in the journal Nature Communications, are still in the early phases of development. Scientists have tested these cells in lab dishes but have not placed them into the human body to see if they can produce insulin in that setting. Additionally, the researchers noted, the cells are not completely identical to the human pancreatic cells that normally produce insulin in the body.
What's more, although the cells could potentially help people with type 2 diabetes someday, their current design would not help people with type 1 diabetes, which is an autoimmune disorder, said Matthias Hebrok, director of the University of California, San Francisco Diabetes Center. [5 Diets That Fight Diseases]
Article continues belowInsulin and sugar
In the pancreas, cells called beta cells produce insulin, the hormone that lowers blood sugar levels by spurring cells throughout the body to take up glucose from the blood. In people with type 1 diabetes, the immune system destroys these beta cells, leaving the body with almost no ability to produce insulin.
In type 2 diabetes, pancreatic beta cells malfunction, producing less insulin. Simultaneously, other cells in the body develop insulin resistance, meaning they require higher and higher levels of insulin to take up sugar in the blood.
Many people with diabetes take insulin or insulin analogues to keep their blood sugar levels in check. However, insulin injections take time reach the cells, so there is a lag time after the injection during which blood sugar can still rise to damaging levels. By contrast, the pancreatic beta cells usually respond quickly and efficiently to blood sugar rises, Hebrok said. Pancreatic cells also release another hormone, called glucagon, which can quickly raise blood sugar levels if they dip too low, he added.
Get the world’s most fascinating discoveries delivered straight to your inbox.
To better mimic pancreas cells' ability to regulate blood sugar, Hebrok and his colleagues took skin cells from newborns (the researchers used cells from male babies' foreskin that otherwise may have been discarded after circumcision procedures), and reprogrammed those cells into pancreatic beta cells by combining the cells with molecules that direct the cells to revert to an earlier stage of cell development, then differentiate and multiply.
Essentially, the researchers turned back the clock on the cells, just enough so that they resembled cells in the endoderm, the layer of tissue in a developing fetus from which most of the internal organs, including the pancreas, form.
When implanted into mice, the reprogrammed pancreas cells prevented the mice from developing diabetes, the researchers said.
The findings raise the hope that people with type 2 diabetes could one day use their own reprogrammed skin cells to control their blood sugar. On their own, the cells would not help those with type 1 diabetes, because the immune system would likely attack and destroy the reprogrammed cells, Hebrok said.
However, other researchers are working on methods to "hide" the cells from the immune system, "giving them a cloak of invisibility like Harry Potter has," Hebrok told Live Science.
Limitations of new cells
Although the new results are promising, the reprogrammed cells are not as efficient at releasing insulin as mature pancreatic beta cells are, Hebrok said. Follow-up work will focus on making these cells more similar to those found in the body naturally, as well as look for ways to shield the cells from the immune system, Hebrok said.
As with any reprogrammed cells, there is always the risk that some of the cells were not reprogrammed correctly. And with any cells that have had their clocks turned back, there is a risk that they can proliferate unchecked, leading to cancer, Hebrok said. However, beta cells don't reproduce well, so the risk of this happening is lower than with certain other types of such cells, he added.
If the cells did eventually make it into humans, they would likely first be encapsulated in a self-contained device, Hebrok said.
"That serves two purposes: One is to protect the cells you put in against the immune system, and the other is to protect the body from the cells themselves," in case they do have tumor-forming properties, he added.
Follow Tia Ghose on Twitterand Google+. Follow Live Science @livescience, Facebook & Google+. Original article on Live Science.

Tia is the editor-in-chief (premium) and was formerly managing editor and senior writer for Live Science. Her work has appeared in Scientific American, Wired.com, Science News and other outlets. She holds a master's degree in bioengineering from the University of Washington, a graduate certificate in science writing from UC Santa Cruz and a bachelor's degree in mechanical engineering from the University of Texas at Austin. Tia was part of a team at the Milwaukee Journal Sentinel that published the Empty Cradles series on preterm births, which won multiple awards, including the 2012 Casey Medal for Meritorious Journalism.
 Live Science Plus
Live Science Plus










