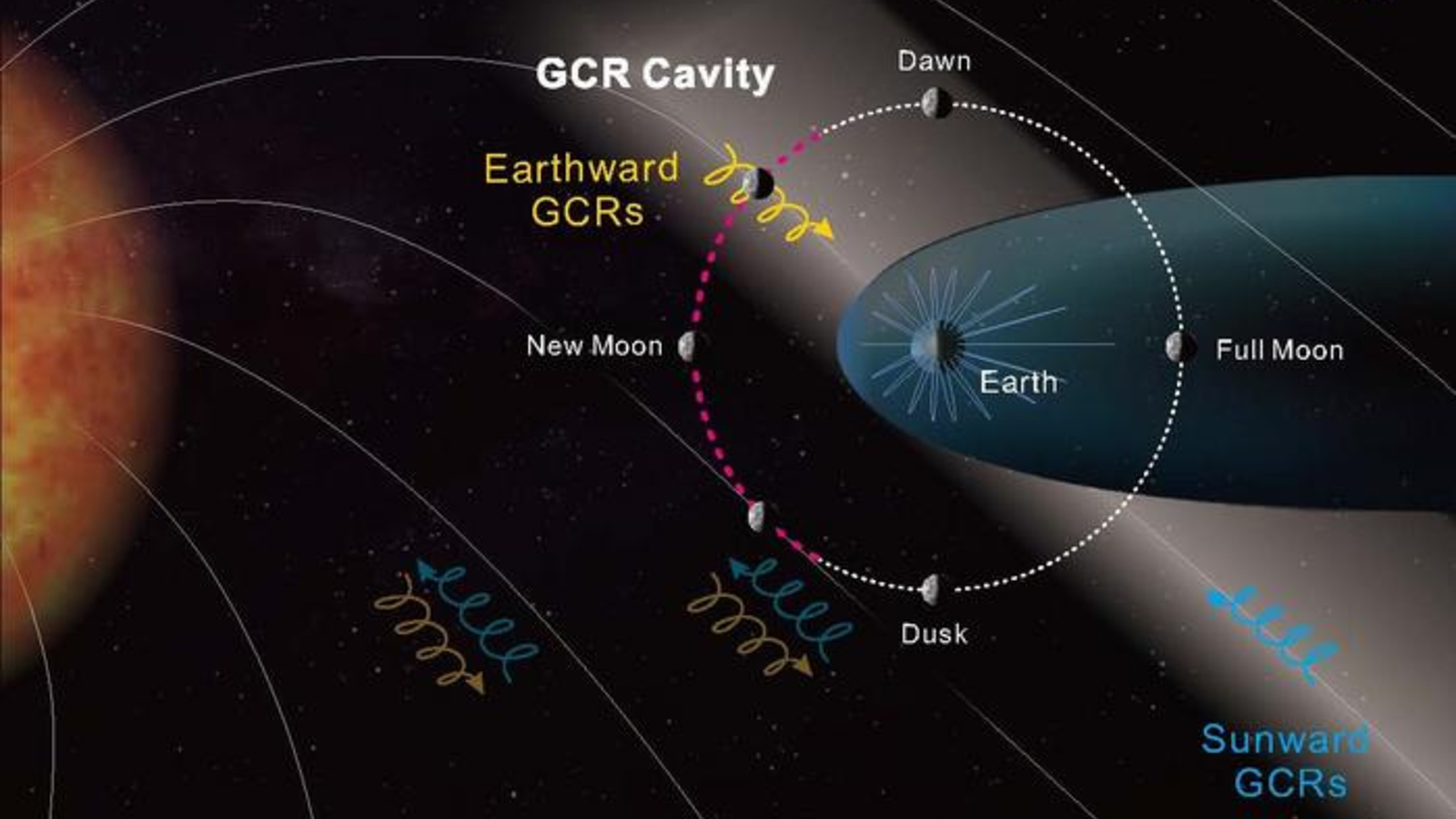
Blood Pressure Vaccine Moves One Step Closer
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
Imagine controlling high blood pressure with just a jab in the arm. Scientists are one step closer to this reality after creating an experimental vaccine that, in rats, provided six months of protection against high blood pressure.
If such a vaccine were to work in people, it could eliminate the trouble and expense of taking blood pressure medication daily, and also could revolutionize the treatment of hypertension in poor or rural communities, where people have limited access to medical care.
A study describing this latest advancement, led by researchers at Osaka University in Japan, appears today (May 26) in the journal Hypertension.
Article continues belowHigh blood pressure, or hypertension, is a common condition in which the force of blood against artery walls is high enough to damage these vessels and ultimately cause heart disease or stroke. There are numerous risk factors for the condition, including smoking, obesity, inactivity, family history and stress.
Nearly one-third of U.S. adults have high blood pressure, according to the Centers for Disease Control and Prevention; the prevalence is more than 40 percent among African Americans.
Treatment for hypertension includes lifestyle changes — namely, quitting smoking, becoming more physically active, and reducing consumption of fatty and salty foods. [10 Amazing Facts About Your Heart]
Doctors also often prescribe blood-pressure medication in addition to lifestyle changes. Effective and relatively safe medications include beta-blockers and angiotensin-converting-enzyme (ACE) inhibitors, both of which work by expanding arteries to ease the pressure of the blood flowing through them.
Get the world’s most fascinating discoveries delivered straight to your inbox.
However, medications are expensive, and many people who are advised to either take the daily pills or change their lifestyle don't actually do so, said Dr. Hironori Nakagami, a professor of medicine at Osaka University and a co-author of the study.
"One of the problems is that patients sometimes forget to take their medication, which may trigger … a stroke or other cardiovascular event," Nakagami told Live Science. In the future, "the physician may select a vaccine therapy to control blood pressure to [better] prevent such cardiovascular events."
Traditional vaccines work by creating immunity against an infection, usually by introducing a weakened or dead virus or bacterium that the immune system can then learn to recognize and kill. In contrast, the experimental blood-pressure vaccine targets one of the body's own hormones, called angiotensin II, which raises blood pressure by causing blood vessels to constrict. (This hormone is the target of ACE inhibitors.)
Scientists have been attempting to develop a blood-pressure vaccine for nearly two decades, with little success. In 2008, doctors published a study in the journal The Lancet describing only modest results for one such vaccine in humans.
That was a peptide vaccine, meaning it was made of a chain of amino acids designed to elicit an immune attack on angiotensin II. The small, limited protection lasted only a few months, and the patients experienced the side effect of flulike symptoms.
Nakagami's group is experimenting with an entirely novel vaccine technique, called a DNA vaccine.
In theory, DNA vaccines work by inserting themselves into DNA, turning human cells into factories that can pump out antibodies to attack pathogens or even cancer cells.
DNA vaccines represent a new frontier. However, they have not been approved for use in humans in any country, although several are being tested, according to the World Health Organization.
"We developed the DNA vaccine to induce anti-angiotensin II antibodies, which can bind angiotensin II and inhibit its function," Nakagami said. "But how to regulate the duration of antibody production is still unknown," he added, noting that this is why the protection against high blood pressure seen in the study was not permanent.
The researchers wrote that their study demonstrates that their DNA vaccine is safe, at least in rats, with no significant side effects.
"[We] will start human clinical trial[s] in a few years in Japan or other countries, and also move to develop a vaccine for heart failure for dogs," Nakagami said.
In 2005, the U.S. Department of Agriculture licensed the first approved DNA vaccine for animals in the United States, which protects horses against West Nile virus. (Vaccines for humans, in contrast, must be approved by the Food and Drug Administration.)
Follow Christopher Wanjek @wanjek for daily tweets on health and science with a humorous edge. Wanjek is the author of "Food at Work" and "Bad Medicine." His column, Bad Medicine, appears regularly on Live Science.

Christopher Wanjek is a Live Science contributor and a health and science writer. He is the author of three science books: Spacefarers (2020), Food at Work (2005) and Bad Medicine (2003). His "Food at Work" book and project, concerning workers' health, safety and productivity, was commissioned by the U.N.'s International Labor Organization. For Live Science, Christopher covers public health, nutrition and biology, and he has written extensively for The Washington Post and Sky & Telescope among others, as well as for the NASA Goddard Space Flight Center, where he was a senior writer. Christopher holds a Master of Health degree from Harvard School of Public Health and a degree in journalism from Temple University.
 Live Science Plus
Live Science Plus