New Pee Test Could Tell If People Have Human 'Mad Cow' Disease
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
People with a type of rare and fatal brain disease linked to eating tainted beef have prions — infectious proteins that cause the disease — in their urine, a new study finds.
The amount of prions in the urine of people with the condition, called variant Creutzfeldt-Jakob disease, is extremely small, and researchers created a new test to amplify the protein so it could be detected.
At this time, the general public should not be worried that the disease can be transmitted to others by urine that contains prions, said study researcher Claudio Soto, a professor of neurology at the University of Texas Health Science Center at Houston (UTHealth) Medical School.
Article continues belowBut the risk of transmission should be studied, because animal studies have shown that the disease can be transmitted by injecting an animal with the urine of an animal that has the disease, Soto said. There is no evidence that Creutzfeldt-Jakob disease is contagious through casual contact with an infected person, according to the National Institute of Neurological Disorders and Stroke.
The larger implication of the new finding is that it may be possible to diagnose, or screen for, variant Creutzfeldt-Jakob disease, using a simple urine test, the researchers said. [10 Things You Didn't Know About the Brain]
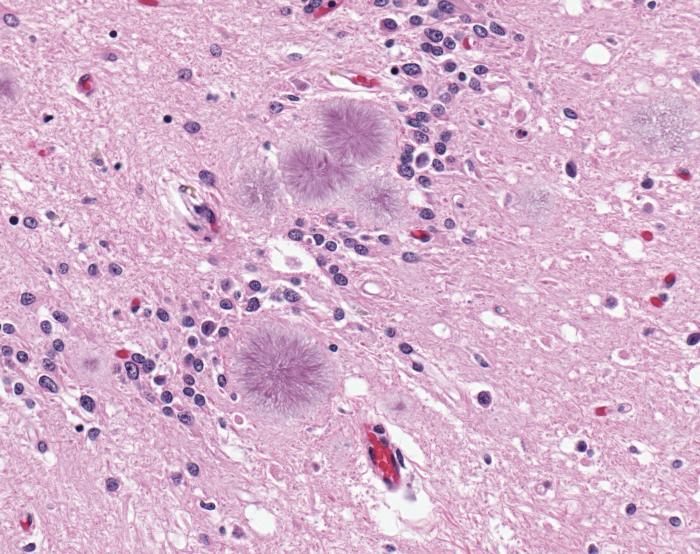
Prions are proteins normally found in the brain that form brain lesions when they start to fold abnormally. In cows, abnormal prions cause bovine spongiform encephalopathy, also known as "mad cow" disease. In humans, abnormal prions cause several diseases, including Creutzfeldt-Jakob disease. There are three forms of Creutzfeldt-Jakob disease — one that is inherited, one that occurs from exposure to infected tissue from the brain or nervous system, and one type that is "sporadic" and does not appear to have a genetic or environmental cause.
In the new study, the researchers looked at variant Creutzfeldt-Jakob disease, a form of the disease that was discovered in 1996. It is believed that people develop the disease after eating beef contaminated with infectious prions from cattle.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Although only about 230 people in the world have been diagnosed with variant Creutzfeldt-Jakob disease, there has been concern that, because the disease takes many years to develop, a number of people may be silent carriers, Soto said. If this is the case, people might contract the disease through blood transfusions, which is believed to have occurred at least once.
"If there are many people silently incubating this infectious material, they may be infecting other people and causing many more cases," Soto said.
The new test detects very tiny amounts of prions by accelerating the process through which they replicate, until they are present in quantities large enough to be detected. The researchers used ultrasound waves to speed up the replication process.
This test detected abnormal prions in the urine of 13 out of 14 people (93 percent) with Creutzfeldt-Jakob disease. What's more, the test was highly specific — it did not give a positive result for people with other forms of Creutzfeldt-Jakob disease, or with other neurological diseases.
If future studies confirm the findings, the test could be used to screen populations to see how many people have abnormal prions in their bodies, Soto said. It might also be used to screen donated blood, he said. However, researchers don't know at this time whether a positive test result means that a person is destined to develop the disease.
The study is published online today (Aug. 6) in the New England Journal of Medicine.
A second study, also published today in the same journal, suggests that a similar test could be used to diagnose the sporadic form of Creutzfeldt-Jakob disease. In the sporadic form, the disease is usually limited to the brain, so prions likely wouldn't be present in the urine or blood. But this second test collects samples from inside the nose, which contains neurons connected to the brain. This nasal test also amplifies prions in a similar way to the test used in Soto's study, but the nasal test uses shaking instead of ultrasound to accelerate the prion replication. The test detected prions in nasal samples from 97 percent of participants with sporadic Creutzfeldt-Jakob disease.
Experts point out the need for additional research on these tests, however. "Creutzfeldt-Jakob disease is extremely uncommon, and a test without near-perfect specificity may also result in many false-positive results if it is applied to patients with a low probability of having the disease," Dr. Colin Masters, of the University of Melbourne in Australia, wrote in an editorial accompanying the studies. "In these circumstances, it is important to highlight the preliminary nature of these studies."
Soto has a patent on the test that was used in his study, and has started a company to develop the test for commercial use. One of the researchers of the second study, from the National Institutes of Health, has a patent on the technology used in that study.
Follow Rachael Rettner @RachaelRettner. Follow Live Science @livescience, Facebook & Google+. Original article on Live Science.

Rachael is a Live Science contributor, and was a former channel editor and senior writer for Live Science between 2010 and 2022. She has a master's degree in journalism from New York University's Science, Health and Environmental Reporting Program. She also holds a B.S. in molecular biology and an M.S. in biology from the University of California, San Diego. Her work has appeared in Scienceline, The Washington Post and Scientific American.
 Live Science Plus
Live Science Plus










