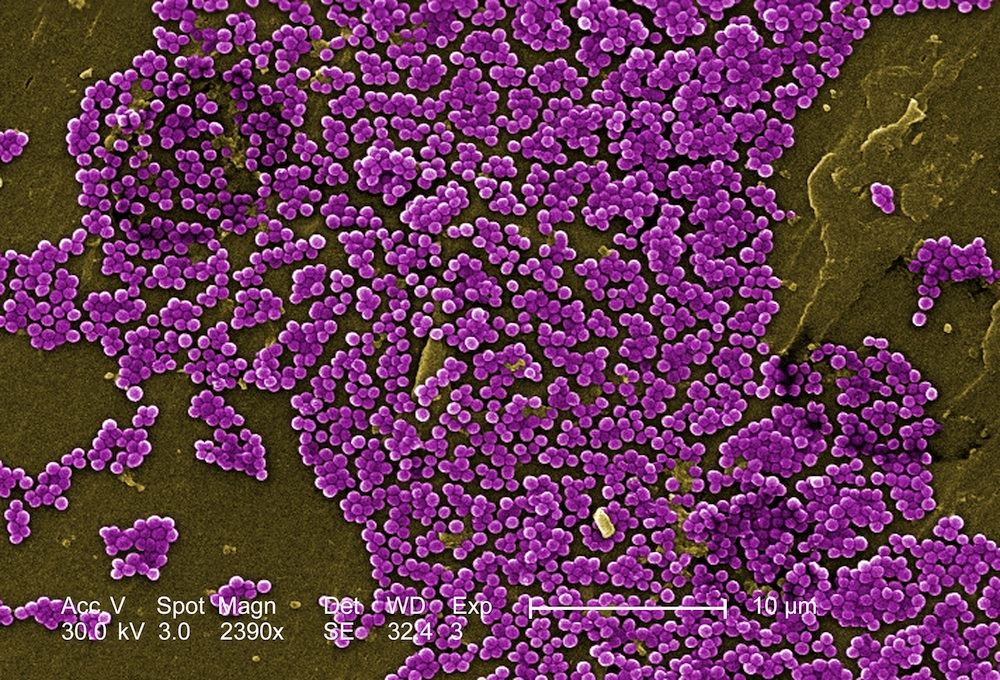
Antibiotic-Resistant Genes Are Literally Everywhere
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
Genes that make bacteria resistant to antibiotics are absolutely everywhere, a new study reveals. They're found in soil, feces and even the ocean.
The findings emphasize an important problem with pathogens that are resistant to traditional antibiotic medicines: The genes for antibiotic resistance are a normal part of bacterial ecology, and they existed before humans started using antibiotics in medicine. Now, however, overuse of antibiotics has spurred the evolution of increasingly drug-resistant strains of bacteria, raising the risk of in-hospital infections and alarming doctors, who fear they will soon be unable to treat infections normally considered minor.
Understanding the genes that make some bacteria antibiotic-resistant might help combat the problem, but gaps remain in how much scientists know about bacterial genetic diversity.
Article continues below"While the environment is known to harbor antibiotic-resistant strains of bacteria, as proven by many preceding studies, we did not really know the extent of their abundance," said Joseph Nesme, a researcher at the University of Lyon in France who collaborated on the new study. [Tiny & Nasty: Images of Things That Make Us Sick]
Bacteria in the wild
Nesme and his colleagues analyzed bacteria DNA sequences from 71 different environments, including human feces, chicken guts, the ocean and even Arctic snow. The researchers compared bacterial DNA gleaned from these environments with sequences in the Antibiotic Resistance Database, which contains 2,999 genetic snippets known to contribute to antibiotic resistance.
Every single environment studied harbored a "relatively important abundance" of antibiotic-resistant genes, Nesme said in a statement, calling the finding "striking." These abundances ranged from 0.05 percent of the genes in the chicken gut to 5.6 percent of the genes found in the bacteria from feces collected from healthy people in Japan.
Get the world’s most fascinating discoveries delivered straight to your inbox.
The most-diverse antibiotic-resistance genes were found in soil, while the least-diverse environments were found in the chicken large intestine and in Arctic snow.
The ecology of resistance
Many of the antibiotic-resistance genes uncovered were involved in the creation of molecules that move unwanted substances out of the cell. These "pumps" can help bacteria get rid of toxic antibiotics. It's no surprise to see such widespread antibiotic resistance, Nesme said. The battle between bacteria and antibiotics is natural: Penicillin, for example, comes from mold. And bacteria even produce their own antibiotics in order to take out other bacteria. Thus, the evolution of antibiotic resistance is expected on this microbial battlefield.
But the new study still can't capture the variety of bacterial genes for fighting antibiotics in the wild, Nesme said. He and his colleagues argue that researchers need to focus on the ecology of bacteria, to better understand how they transfer genes — and how those genes can lead to problems in hospitals and sickbeds.
"It is only with more knowledge on antibiotic resistance dissemination — from the environment to pathogens in the clinic and leading to antibiotic treatment failure rates — that we will be able to produce more sustainable antibiotic drugs," Nesme said.
The researchers report their findings today (May 8) in the journal Current Biology.
Follow Stephanie Pappas on Twitter and Google+. Follow us @livescience, Facebook & Google+. Original article on Live Science.

Stephanie Pappas is a contributing writer for Live Science, covering topics ranging from geoscience to archaeology to the human brain and behavior. She was previously a senior writer for Live Science but is now a freelancer based in Denver, Colorado, and regularly contributes to Scientific American and The Monitor, the monthly magazine of the American Psychological Association. Stephanie received a bachelor's degree in psychology from the University of South Carolina and a graduate certificate in science communication from the University of California, Santa Cruz.
 Live Science Plus
Live Science Plus









