HPV Vaccine: One Dose May Be Enough
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
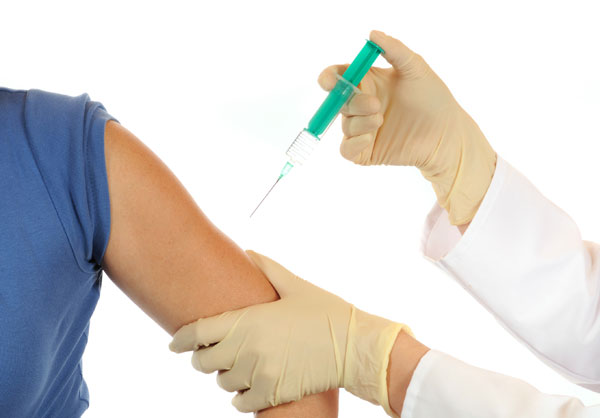
A single dose of the human papillomavirus (HPV) vaccine may be enough to protect women against infection with the virus over the long-term, a new study from Costa Rica suggests.
In the study, women who received one, two, or the standard three doses of the HPV vaccine all produced antibodies against the virus that remained at stable levels in their bodies for four years after vaccination.
In addition, women who received one dose of the vaccine had an immune response that was five to nine times stronger than that seen in women who were infected with HPV naturally. [5 Dangerous Vaccination Myths]
Article continues belowHowever, women who received only one vaccine dose produced antibodies at levels lower than those of women given two or three doses. Still, researchers don't yet know what level would be "good enough" for protection. It could be that the immune response generated after two or three doses is much more than a person needs to be protected.
In fact, a previous study by the same researchers suggested just that: women in that study who received all three HPV doses were protected against infection no better than those who received one or two.
"We don’t know what the minimum required [antibody level] might be for protection," said study researcher Mahboobeh Safaeian, of the National Cancer Institute. But in the new study, "the fact that [antibody levels] remained higher than natural infection, and remained stable, is a promising finding," Safaeian said.
More research is needed to validate the findings in other populations, and over longer periods. Until then, people in the United States should get the recommended three doses, Safaeian said.
Get the world’s most fascinating discoveries delivered straight to your inbox.
The HPV vaccine is currently licensed for people between ages 9 and 26, and it works best if given before people become sexually active. It is used to protect against cervical cancer, which is often caused by HPV infections.
If the number of recommended doses could be lowered from three to just two or one, it would simplify the vaccination schedule, Safaeian said. Studies show that in the United States, only about a third of teen girls get all three doses.
Fewer doses would also reduce the cost of vaccination, which has been a barrier to administering the vaccine to people in developing countries, Safaeian said.
Fewer doses?
The new study is part of a larger trial in which more than 3,500 women ages 18 to 25 were assigned to receive three doses of the Cervarix HPV vaccine — a vaccine that protects against the cancer-causing strains HPV 16 and HPV 18. However, about 20 percent of participants did not end up receiving all three doses.
The researchers analyzed blood samples taken from 78 women who received one dose, 192 women who received two doses, and 120 who received three doses of the HPV vaccine, as well as 113 women who did not receive the HPV vaccine, but had previously been infected with HPV.
Four years after vaccination, all of these women had antibodies against HPV 16 and HPV 18 in their blood.
Those who received two doses six months apart had about the same levels of antibodies as those who received all three doses.
Those who received one or two doses had antibody levels that were five to 24 times higher than the women who had been naturally infected.
Future research
"Because of the challenges associated with giving three doses [of HPV vaccine] I think it's very welcomed to see that there's a possibility that the vaccine may not need to be given in a three dose schedule," said Dr. Mike Brady, a pediatric infectious disease specialist at Nationwide Children’s Hospital in Columbus, Ohio.
But before doctors would feel comfortable giving fewer than three doses, researchers will need to show that vaccination with one or two doses also reduces the risk of developing the early signs of cervical cancer, Brady said. Currently, there's lots of evidence that three doses protects against cervical cancer, but the same cannot be said for fewer doses.
When researchers initially developed the HPV vaccine, which is a synthetic vaccine, they didn't know exactly how many doses would be needed for protection, Brady said. So they modeled the dosing schedule after that of other synthetic vaccines such as Hepatitis B, which require more than one dose.
However, the HPV vaccine is not like other synthetic vaccines — it is made from so-called virus-like particles — and it may turn out that fewer doses than initially thought are needed, Brady said.
Because the study evaluated only the Cervarix vaccine, it's not clear whether the findings apply to the Gardasil vaccine, another HPV vaccine that protects against four strains of HPV, and is more widely used in the United States.
The study is published in the November issue of the journal Cancer Prevention Research.
Follow Rachael Rettner @RachaelRettner. FollowLiveScience @livescience, Facebook & Google+. Original article on LiveScience.

Rachael is a Live Science contributor, and was a former channel editor and senior writer for Live Science between 2010 and 2022. She has a master's degree in journalism from New York University's Science, Health and Environmental Reporting Program. She also holds a B.S. in molecular biology and an M.S. in biology from the University of California, San Diego. Her work has appeared in Scienceline, The Washington Post and Scientific American.
 Live Science Plus
Live Science Plus










