Tiny Katydid Ears Look Remarkably Human


Their ears may be on their legs, but katydids hear a lot like humans do, a new study finds.
In fact, even though insect and mammal lineages diverged a staggeringly long time ago, even for the evolutionary scale, our ears have evolved to work in remarkably similar ways. The findings could be useful for engineering miniature sound sensors, said Daniel Robert, a bionanoscientist at the University of Bristol in the United Kingdom.
"It's a bit of a breakthrough for us, because now we know that ears exist that can do such refined analysis [of sound] and yet be that small and that simple," Robert told LiveScience.
Article continues belowKatydid ears
Robert and his colleagues focused their study on the South American katydid Copiphora gorgonensis, an orange-face insect that can hear sound whose frequency ranges from 5,000 to 50,000 hertz. Humans, in comparison, can hear between about 20 and 20,000 hertz. These katydids sing at about 23,000 hertz, in ultrasound, or above the human range of hearing.
The ears of C. gorgonensis are less than a millimeter long. When study researchers opened them up, they found a set of eardrums, or tympanic membranes — unlike us, the katydids have two per ear, Robert said. More to their surprise, they found a fluid-filled vesicle inside the ear, a previously undiscovered organ. The challenge, said study co-author Fernando Montealegre-Zapata, now a senior lecturer at the University of Lincoln, was that the vesicle resisted exploration. When they opened it, it burst.
"The whole system explodes," Montealegre-Zapata told LiveScience.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Clearly, the vesicle was under pressure. Using a series of techniques from sensitive microscopes to X-ray micro-computed tomography (CT, a similar technique as used in human medicine), the researchers were able to reconstruct the structure of the whole katydid ear without having to open up the pressurized vesicle. They found that it looks much like our own. [7 Ways Animals Act Like Humans]
Humans vs. katydids
The human ear has three parts: the eardrum, which vibrates when sound waves hit it; the ossicles, tiny bones that transmit and amplify these vibrations so they can be transmitted to the third part; and the cochlea, a fluid-filled coil that houses thousands of hairlike cells that transmit the vibrations to the brain for processing. Without the ossicles, the vibrations would otherwise be muted when they moved from air to the watery cochlea.
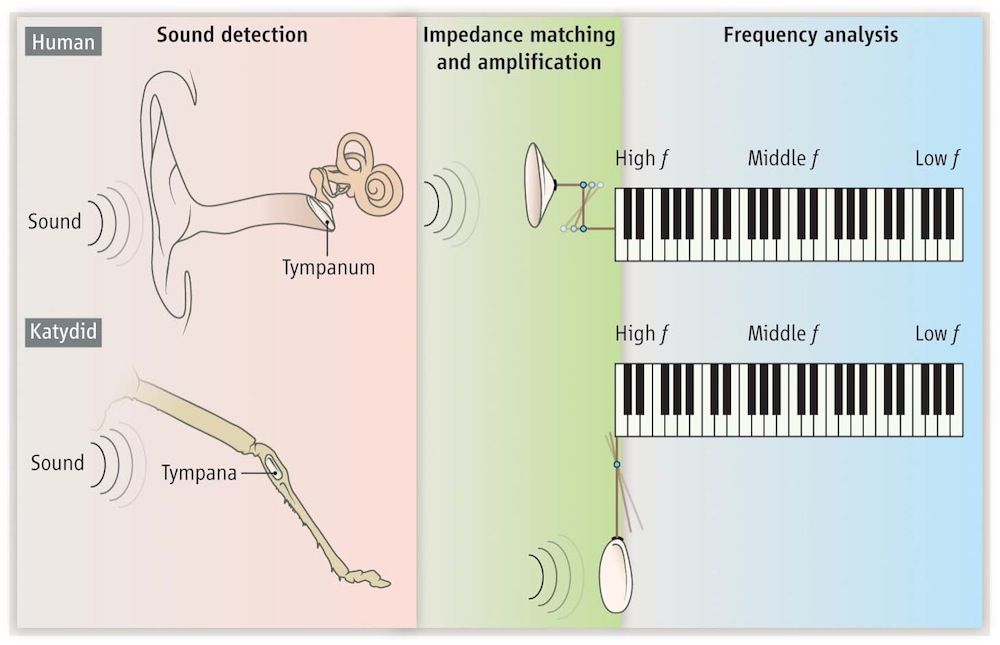
Katydids have a similar system. Their dual eardrums transmit vibrations to a plate that works like a lever, increasing the force so the vibrations can transmit to the fluid-filled vesicle, a simpler version of our own ossicle system. The vesicle itself is like a simplified cochlea. It's uncoiled, more like an "elongated balloon," Robert said, than a snail shell, and it has far fewer sensory hair cells than the human cochlea.
Katydids likely need a sensitive hearing system, because they sing in the same range as the echolocating calls of their major predators, bats. Thus, Robert said, katydids have to be able to discriminate between the sound of an incoming hunter and the sound of a singing mate.
"If you're a male katydid and you sing your heart out on the leaf to attract the female and there is a bat flying around with echolocation in the same frequencies as you, there is some possibility for confusion there," Robert said. "If the bat finds you, the confusion stops, because you die."
The researchers are now investigating the ears of other insects, including a closely related katydid that sings at an amazingly high 150 kilohertz (150,000 hertz), the most ultrasonic singer of any known organism. The wavelength of such sounds is so short that the katydids must have ultrasensitive ear structures to catch it over distances, Montealegre-Zapata said. Ultimately, the researchers plan to engineer extremely tiny, extremely sensitive microphones and sound sensors.
"The next stage is to use the tools of nanotechnology to make a sensor, which is a half-millimeter across and does the same job," Robert said.
Follow Stephanie Pappas on Twitter @sipappas or LiveScience @livescience. We're also on Facebook & Google+.

Stephanie Pappas is a contributing writer for Live Science, covering topics ranging from geoscience to archaeology to the human brain and behavior. She was previously a senior writer for Live Science but is now a freelancer based in Denver, Colorado, and regularly contributes to Scientific American and The Monitor, the monthly magazine of the American Psychological Association. Stephanie received a bachelor's degree in psychology from the University of South Carolina and a graduate certificate in science communication from the University of California, Santa Cruz.