Viruses May Cause More Cancer than Previously Thought
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
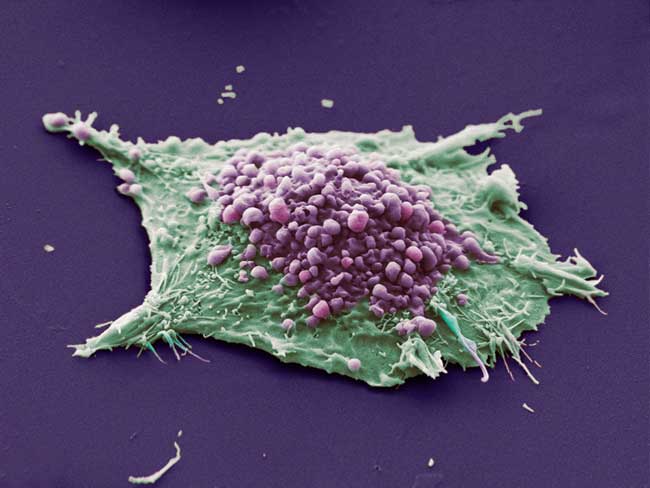
Viruses may be causing more cancers than previously thought, according to a new study.
Scientists know that a few cancers, such as cervical cancer, are caused by viruses, because researchers have isolated the viral genomes from the cancerous cells. But some viruses may take a "hit and run" approach — inducing cancer and then vanishing before the disease is caught, the researchers say.
The new study, performed in mice, showed that a particular mouse herpesvirus could trigger cancer but then practically disappear from the cancer cells. Herpesviruses belong to a family of viruses called Herpesviridae that can infect humans and include chicken pox and the Epstein-Barr virus — a virus that virtually everyone is infected with, yet only causes cancer in rare cases.
Article continues below"We're saying that herpes viruses could cause a lot more disease than you would guess if you limited your suspicion to just to cancers that came out having the virus genome in them," said study author Philip Stevenson, a researcher at the University of Cambridge in the United Kingdom. "We're saying that they might cause orders of magnitude more cancers," he told LiveScience.
However, it is important to note that the cancers induced in the mice are different from the cancers that occur in people, and so the findings might not hold true for humans.
The results are published in the September issue of the Journal of General Virology.
Hit and run
Get the world’s most fascinating discoveries delivered straight to your inbox.
Cancers occur when the genetic material within cells, the cells' DNA, develops mutations that cause the cell to divide uncontrollably. These mutations sometimes arise when the DNA is damaged. However, viruses can have effects similar to these mutations when they insert themselves into the DNA, and the end result is the same – uncontrolled cell growth. Two types of human papillomaviruses (HPV) are thought to cause cervical cancer this way.
By inserting itself into the cells' DNA, the virus "hides" from the body's immune system, so the body's defenses can't eliminate it. This means that researchers can later find evidence that HPV was indeed the culprit behind cervical cancer — the virus leaves its genes behind like fingerprints at a crime scene. About 20 percent of cancers are thought to be caused by viruses, according to Stevenson.
However, cells have several defense mechanisms to prevent tumor growth. Sometimes cells with certain mutations will self-destruct so they don’t turn cancerous.
But viruses can interfere with these defenses.
"Viruses don't set out to cause cancer, but their replication uses all the same functions. So they tend to inhibit the whole set of these protective mechanisms," Stevenson said. "So they're kind of ideal agents for causing cancer."
The "hit and run" hypotheses proposes that a virus can cause cancer without integrating itself into the cell's DNA. In this case, a cell develops a genetic mutation, but the virus present in the cell overrides the defense mechanisms and allows the cell to continue to live. Over time, more and more genetic mutations develop, and the cell turns cancerous. However, by the time the cancer is discovered, the virus has been eliminated by the immune system, leaving no "fingerprints" behind.
For many years scientists have suspected viruses could cause cancer through this mechanism, but it was difficult to prove.
Cancer vaccines
Stevenson and his colleagues wanted to know: if a virus triggers cancer without integrating into the cells' DNA, how often can you still find traces of the virus inside the cancer cells?
They genetically engineered mice so that they would develop cancer if infected with the herpesvirus (the virus has a gene that triggers mouse cells to divide uncontrollably).
But when they later checked the tumors that grew in the mice, they found no trace of the virus. If the findings hold true for humans, that would mean viruses such as Epstein–Barr trigger more cancers than previously thought.
Further, the researchers found evidence that vaccinating against these viruses might prove effective in preventing cancers. When the researchers vaccinated the mice against the herpesvirus, none of the mice developed cancer.
While a vaccine against Epstein–Barr and other herpes viruses could theoretically be made, companies would not be willing to take the risk to develop them unless there was some significant disease threat, Stevenson said.
"With every vaccine there's a balance between the risk and the benefits," Stevenson said. Because cancers that have the Epstein-Barr virus genome in them are thought to be relatively rare, the benefits are seen as small, so no one wants to take the risk, he said. "If people recognize that these viruses may cause an awful lot more cancers, then the benefits [become greater]."
The current study suggests that a clinical trial of an Epstein–Barr vaccine or similar vaccine might be worthwhile, Stevenson said.

Rachael is a Live Science contributor, and was a former channel editor and senior writer for Live Science between 2010 and 2022. She has a master's degree in journalism from New York University's Science, Health and Environmental Reporting Program. She also holds a B.S. in molecular biology and an M.S. in biology from the University of California, San Diego. Her work has appeared in Scienceline, The Washington Post and Scientific American.
 Live Science Plus
Live Science Plus










