Conspiracy & paranormal news, features and articles


Something draws us towards the strange and mysterious, but when we can’t explain something through the scientific method, it's easy for our minds to wander into the realm of ghosts, monsters and UFOs. And even when science has a clear explanation, conspiracy theories sprout from nowhere to grip some people's minds. That's why Live Science's expert writers and editors guide you rationally through the science of the supernatural and explain why people believe, with the latest news, features and articles about conspiracies and the paranormal.
Discover more about conspiracy & paranormal phenomena
-
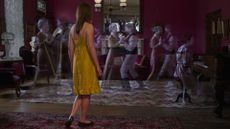 What's the scientific explanation for 'ghost encounters'?
What's the scientific explanation for 'ghost encounters'?People all over the world believe they've seen or heard a ghost, but there's no scientific evidence for spirits, hauntings or the paranormal. So what's behind these "encounters"?
By Patrick Pester Published
-
 21 of the best conspiracy theories
21 of the best conspiracy theoriesConspiracy theories that have taken hold range from a faked moon landing to Barack Obama's birthplace to chemtrails.
By Stephanie Pappas Last updated
-
 'Yeti hair' found in Himalayas is actually from a horse, BBC series reveals
'Yeti hair' found in Himalayas is actually from a horse, BBC series revealsDNA from a supposed Abominable Snowman actually came from a horse, but that doesn't mean stories of the Yeti passed on by local people aren't important.
By Patrick Pester Published
-
 Haunting 'mermaid' mummy from Japan is a gruesome monkey-fish hybrid with 'dragon claws,' new scans reveal
Haunting 'mermaid' mummy from Japan is a gruesome monkey-fish hybrid with 'dragon claws,' new scans revealScientists have scanned the mummified remains of a supposed "mermaid" from Japan. The initial results suggest it is a horrifying mix of fish, monkey and lizard parts.
By Harry Baker Published
-
 Are ghosts real?
Are ghosts real?Opinion One difficulty in scientifically evaluating is ghost are real is the surprisingly wide variety of phenomena attributed to ghosts.
By Benjamin Radford Last updated
Opinion -
 The biggest hunt for the Loch Ness Monster in 50 years is about to begin
The biggest hunt for the Loch Ness Monster in 50 years is about to beginA visitor attraction in Scotland is enlisting the help of volunteers to search for Nessie, but it's unlikely to yield a monster discovery.
By Patrick Pester Published
-
 Most methods for squashing conspiracy theories don't work, study finds. Here's what does.
Most methods for squashing conspiracy theories don't work, study finds. Here's what does.A systematic review of conspiracy theory interventions shows that most traditional approaches have little impact, but certain alternatives show promise.
By Anna Demming Published
-
 How do flat-Earthers explain the equinox? We investigated.
How do flat-Earthers explain the equinox? We investigated.Equinoxes would be physically impossible over a flat Earth, but that doesn't stop conspiracy theorists from trying to explain them.
By Brandon Specktor Last updated
-
 Flat Earth 'theory': Why do some people think the Earth is flat?
Flat Earth 'theory': Why do some people think the Earth is flat?Flat-earthers believe one of the most curious conspiracy theories on the internet. Here's a look at what they believe and why.
By Stephanie Pappas Last updated
