Experimental Device Could Prevent HIV & Pregnancy in Women
An experimental device could protect women from HIV and herpes while also preventing pregnancy, studies in animals suggest.
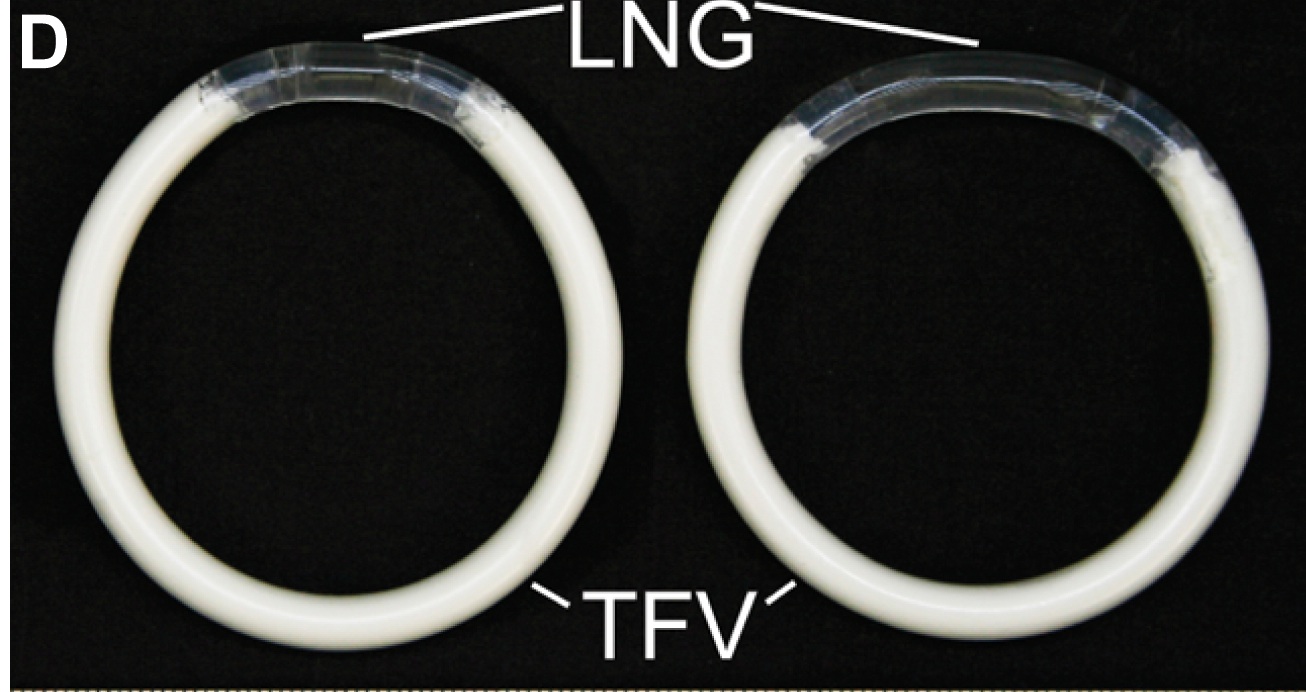
The device, a flexible plastic ring that is inserted into the vagina, delivers three months of both an antiretroviral drug and a contraceptive drug. So far, the ring has been tested only in sheep and rabbits. But if it is shown to be effective in humans, it could provide a way for women in the developing world to avoid unwanted pregnancies while also shielding them from sexually transmitted diseases.
"In many ways, you can think about this functioning similarly to a condom," said study co-author Patrick Kiser, a biomedical engineer at Northwestern University. "It prevents STI [sexually transmitted infection] transmission, and it would prevent unwanted pregnancy, but it's woman-controlled." [Quiz: Test Your STD Smarts]
Long road
The hunt for a female-controlled method to protect against HIV, the virus that causes AIDS, has a long history littered with failures. In the developing world, many women are infected with HIV by their husbands, who may resist wearing condoms. And convenient contraception isn't always available, Kiser said.
"The unmet need for HIV protection technology and family planning technology really coincide in low-income countries, particularly in sub-Saharan Africa where HIV infection rates among young women can be as high as 20 percent, even higher," Kiser told Live Science.
One approach to giving women an option to protect themselves against HIV was vaginal gels. Although early gels actually increased the virus's rate of transmission, a 2010 trial in South Africa found that a gel containing the antiretroviral drug tenofovir reduced HIV transmission by 39 percent, and herpes infections by 51 percent.
Get the world’s most fascinating discoveries delivered straight to your inbox.
However, the gel had to be inserted before and after sex, and as a result, many women didn't use it consistently. Follow-up studies to confirm the gel's effectiveness are still in progress, Kiser said.
Multipurpose device
In search of a method that was easier to use, Kiser and his colleagues designed a vaginal ring that released a steady dose of levonorgestrel, the hormone found in the emergency contraceptive pill Plan-B and hormonal intrauterine devices (IUDs), as well as tenofovir, which lowers the risk of HIV transmission.
The problem the researches ran into was that the device had to deliver nearly 1 gram (0.03 ounces) of water-soluble tenofovir over 90 days, but just a tiny fraction of that, or "not much more than a couple of specks of dust," of the hormonal contraceptive, Kiser said.
To get around this problem, they used an electromagnetic field to weld together two different segments of the plastic polyurethane. (Polyurethane is widely used in medical devices such as catheters.) One segment has contraceptive at its core, covered by a sheath that slowly releases minute doses of the drug. The other segment is filled with the water-soluble antiretroviral. It swells when it comes in contact with vaginal fluids, releasing the antiretroviral drug in the amounts needed.
More options
The team hopes the device will be easier than tenofovir gel for women to use consistently because once it is inserted, it needs to be changed only every three months.
"Most women can't feel that it's there, it can literally just be forgotten about," Kiser said.
The team has also developed a ring that delivers only the antiretroviral drug, which would not prevent pregnancy, as well as two separate contraceptive-only rings in different doses.
In animals, the device delivered the appropriate amounts of each drug, but it has yet to be tested in women.
The new ring was described Wednesday (Mar. 5) in the journal PLOS ONE.
Follow Tia Ghose on Twitter and Google+. Follow Live Science @livescience, Facebook & Google+. Original article on Live Science.

Tia is the editor-in-chief (premium) and was formerly managing editor and senior writer for Live Science. Her work has appeared in Scientific American, Wired.com, Science News and other outlets. She holds a master's degree in bioengineering from the University of Washington, a graduate certificate in science writing from UC Santa Cruz and a bachelor's degree in mechanical engineering from the University of Texas at Austin. Tia was part of a team at the Milwaukee Journal Sentinel that published the Empty Cradles series on preterm births, which won multiple awards, including the 2012 Casey Medal for Meritorious Journalism.

 Live Science Plus
Live Science Plus






