'Black fungus' treatment runs short in India as new cases of infection emerge
Get the world’s most fascinating discoveries delivered straight to your inbox.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
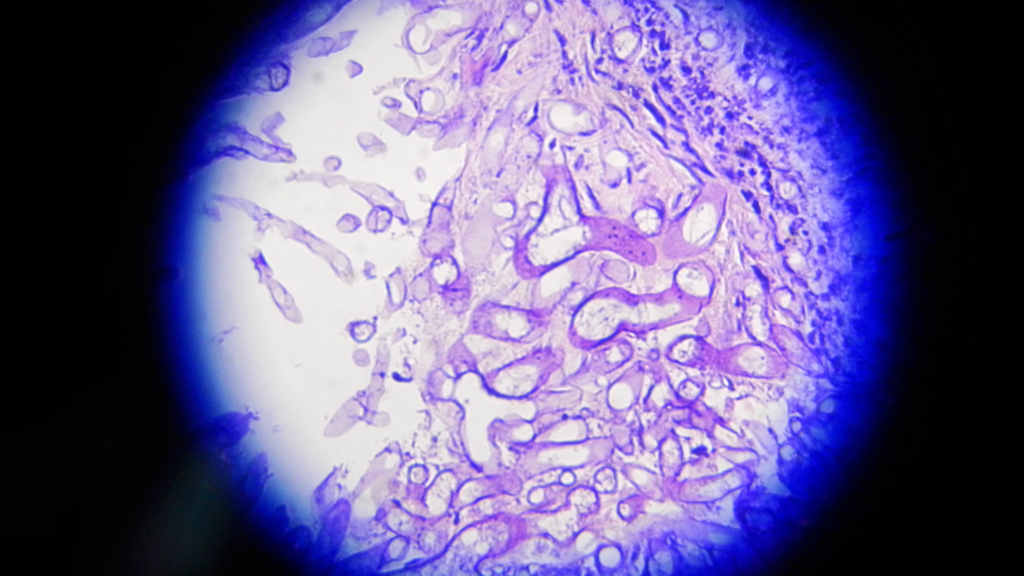
New cases of mucormycosis, a potentially fatal fungal infection, continue to crop up among COVID-19 patients in India, but many regions are facing shortages of the drug used to treat the disease.
Mucormycosis, also called "black fungus," typically strikes people with weakened immune systems, such as those with diabetes and those taking immunosuppressive drugs — including the steroids sometimes used to treat COVID-19, Live Science previously reported. Experts suspect that the rise in cases may be to these steroids, administered both in hospitals and sometimes at home, as hospitals have recently been overrun. In addition, those receiving medicine or oxygen therapy at home may be more likely to pick up the mold from their surroundings, compared with those treated in a sterile, clinical setting.
The infection is caused by a group of molds called mucormycetes, which grow in soil and decaying organic matter. The mold can enter the body through cuts in the skin, or through the airways when people breathe in the spores. When inhaled, the mold can infect the lungs and sinuses and then sometimes spread to other organs, including the brain, eyes, spleen and heart. Sometimes, patients' infected eyes must be removed to prevent the mold from spreading to the brain.
Article continues belowRelated: 20 of the worst epidemics and pandemics in history
Black fungus can be treated with amphotericin B, BBC News reported; this powerful antifungal drug is only used to treat potentially life-threatening fungal infections, as it can sometimes cause severe side effects, according to the U.S. National Library of Medicine. The intravenous injection of the drug must be administered every day for up to eight weeks and comes in two forms: the standard amphotericin B deoxycholate and an alternate version called liposomal amphotericin, which packages the conventional drug within bubbles of fat.
Doctors generally prefer the latter version of the drug, Dr. Akshay Nair, a Mumbai-based eye surgeon, told BBC News.
"We prefer the liposomal form, since it is safer, more effective and has lesser side effects, the flip side being that it is more expensive," Nair said. And the drug can be even more expensive when bought on the black market, which some people may do as demand continues to surge and the supply of both formulations remains limited, according to the BBC.
Get the world’s most fascinating discoveries delivered straight to your inbox.
But the Indian government is working to address the shortage. "We have chalked out a strategy with manufacturers to ramp up domestic production as well as to import the drug from all over the world," Mansukh Mandaviya, India's minister of state for chemicals and fertilizers, wrote on Twitter on May 18. "We have already improved the supply of #AmphotericinB by many folds. But currently, we are facing a sudden demand surge," he wrote.
"We have also outlined the system for efficient distribution and supply chain management of #AmphotericinB. The shortage will get resolved at the earliest. I also urge states to use this drug judiciously by strictly following guidelines," Mandaviya added.
For instance, the Delhi government has now established an expert committee, headed by doctors from Maulana Azad Medical College, to coordinate amphotericin B distribution, The Economic Times reported. Hospitals will need to apply for the drug through the committee, which will meet several times a day to review the requests. By also working with the Directorate General of Health Services, the committee will help to facilitate the rapid allocation of the drug where it's needed most desperately, according to The Economic Times.
While Indian state governments work toward speedy and equitable distribution of the antifungal, some residents have taken to social media to find amphotericin B on their own, BBC News reported.
Urgently required:Amphotericin BPatient name: RavindraNaiduAge: 62 yrsLocation: TirupatiHosptial: AmaraContact: Shyam 9949954433Please help@JSPSriram @charan_tweetz @6eChaithu @kiranbs45 @HiHyderabad @gopal_karneedi pic.twitter.com/k68Qjk5pcOMay 18, 2021
Last week, the health minister of Maharashtra said 1,500 cases of black fungus had been reported in the state, which is the second-most populous in the country after Uttar Pradesh, BBC News reported. Similarly, Gujarat state officials said nearly 900 cases had been reported in the past month. Amphotericin B was easy to find as recently as three weeks ago, a pharmacy owner in Uttar Pradesh told the BBC.
Originally published on Live Science.

Nicoletta Lanese is the health channel editor at Live Science and was previously a news editor and staff writer at the site. She holds a graduate certificate in science communication from UC Santa Cruz and degrees in neuroscience and dance from the University of Florida. Her work has appeared in The Scientist, Science News, the Mercury News, Mongabay and Stanford Medicine Magazine, among other outlets. Based in NYC, she also remains heavily involved in dance and performs in local choreographers' work.
 Live Science Plus
Live Science Plus