Cat with 4 frostbitten paws gets new feet made of titanium
3D-printed titanium limbs helped a frostbitten feline get back on her feet.
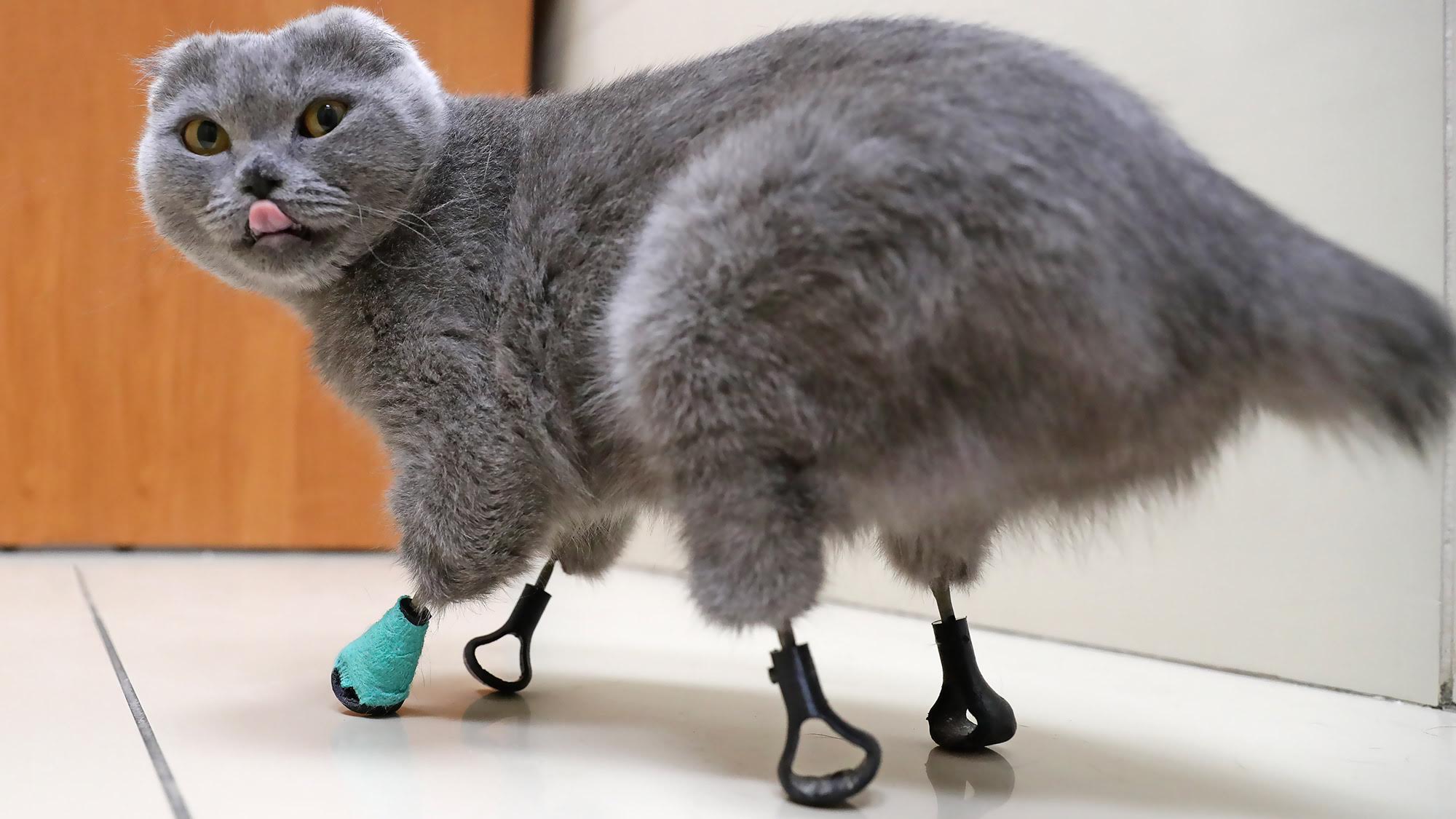
A female cat in Russia that lost all four of her paws to frostbite can walk, run and even climb stairs again, thanks to the veterinarians who replaced her missing limbs with 3D-printed prosthetics made from titanium.
The hardy gray feline, named Dymka ("mist" in Russian), is about 4 years old. A passing driver found her in December 2018 in the snow in Novokuznetsk in Siberia and brought her to a clinic in Novosibirsk, according to Russian news site Komsomolskaya Pravda.
Dymka was suffering from frostbite of her paws, ears and tail — so much so that veterinarian Sergei Gorshkov had to amputate those damaged extremities, The Moscow Times reported.
Related: Body beautiful: The 5 strangest prosthetic limbs
During the punishing Siberian winters, veterinarians at the Novosibirsk clinic typically treat at least five to seven cats due to frostbite in their paws, ears and noses, Gorshkov told The Moscow Times. Frostbite develops when cold temperatures freeze skin and tissue, particularly in extremities. In severe cases, the tissue dies and requires amputation.
Gorshkov and his colleagues at the clinic collaborated with researchers from Tomsk Polytechnic University (TPU) in Tomsk, Russia, to create a set of prosthetics for Dymka. The scientists developed and applied a coating made of calcium phosphate to help mount the titanium implants that were inserted and fused into her leg bones, and to minimize the risk of infection and implant rejection, TPU representatives said in a statement.
Researchers used computerized tomography (CT) X-ray scans of Dymka's legs to model and then 3D print the titanium rods. Dymka received her prosthetic implants in July 2019 — first in her front legs and then in her hind legs, Novosibirsk News reported. In a video shared to YouTube by the veterinary clinic on Dec. 10, 2019 — seven months after Dymka's new paws were attached — she was enjoying a wake-up stretch, walking around an examination room and playing with a fringe on a blanket.
Get the world’s most fascinating discoveries delivered straight to your inbox.
A close-up showed where the titanium rods joined to her legs, ending in "feet" made of flexible black material with textured bottoms.
Dymka is now the second cat in the world to have received four metal prosthetics. In 2016, the Novosibirsk clinic performed a similar procedure, mounting titanium implants on a male cat named Ryzhik ("Red" in Russian), who also required a quadruple amputation of his frostbitten feet, according to The Moscow Times.
- Super-intelligent machines: 7 robotic futures
- 7 cool uses of 3D printing in medicine
- The 10 weirdest things created by 3D printing
Originally published on Live Science.


Mindy Weisberger is a science journalist and author of "Rise of the Zombie Bugs: The Surprising Science of Parasitic Mind-Control" (Hopkins Press). She formerly edited for Scholastic and was a channel editor and senior writer for Live Science. She has reported on general science, covering climate change, paleontology, biology and space. Mindy studied film at Columbia University; prior to LS, she produced, wrote and directed media for the American Museum of Natural History in NYC. Her videos about dinosaurs, astrophysics, biodiversity and evolution appear in museums and science centers worldwide, earning awards such as the CINE Golden Eagle and the Communicator Award of Excellence. Her writing has also appeared in Scientific American, The Washington Post, How It Works Magazine and CNN.



