Why rescuers are feeding turtles mayonnaise after a disastrous oil spill

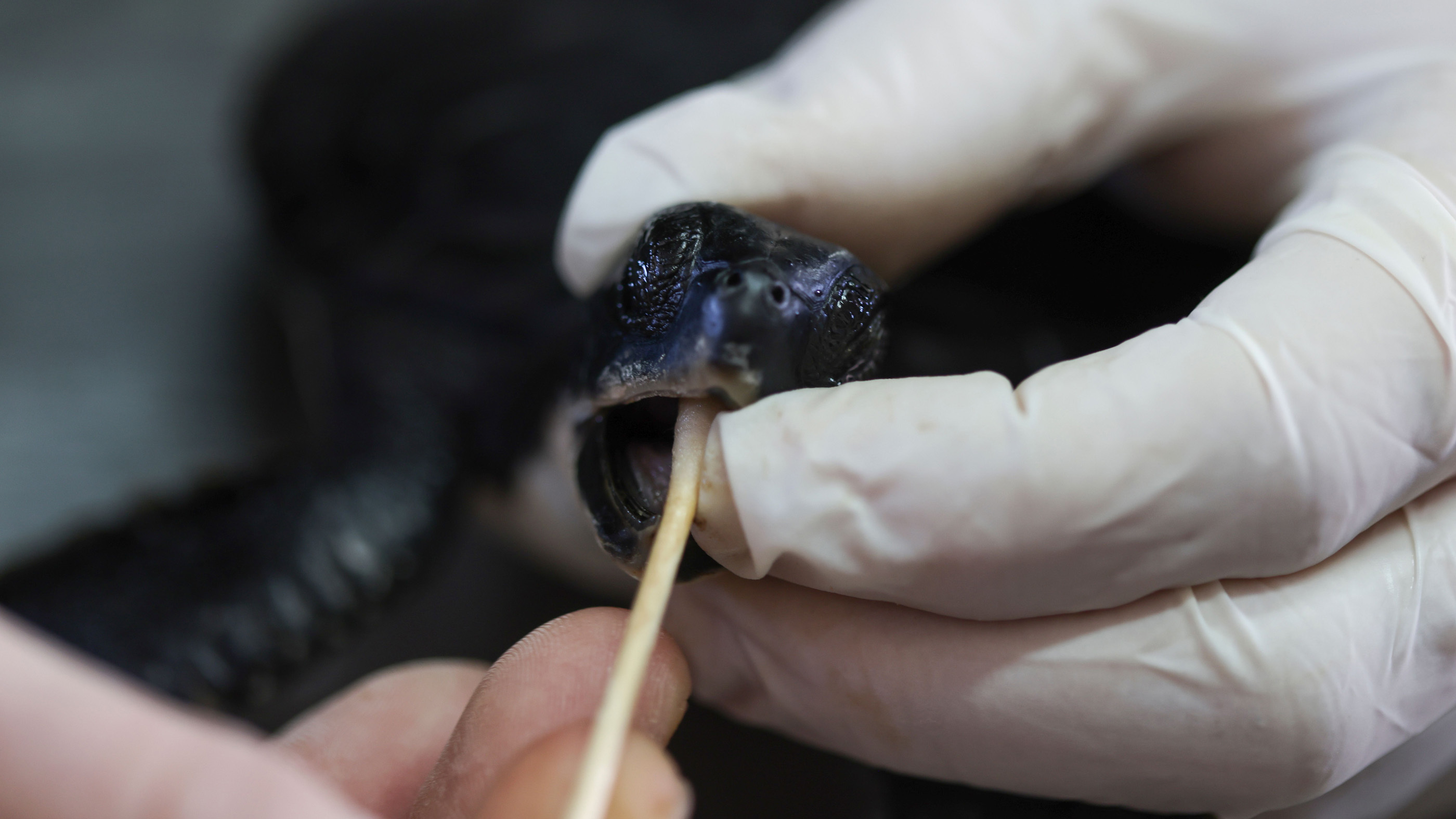
While some people slather mayonnaise on their sandwiches, Israel's National Sea Turtle Rescue Center uses the condiment for a unique purpose: to treat endangered turtles after oil spills, The Associated Press (AP) reported.
More than 100 miles (160 kilometers) of Israel's Meditteranean coastline became blotted with huge globs of tar last week, after an oil spill occurred about 31 miles (50 km) offshore, Live Science previously reported. Israel's Nature and Parks Authority (INPA) called the spill "one of the most serious ecological disasters" the country has ever seen, and the hundreds of tons of oil pose a particularly serious threat to sea turtles, since they breathe and feed at the water's surface.
Many tar-coated turtles have already washed ashore, some dead, but the National Sea Turtle Rescue Center was able to transport 11 endangered green sea turtles (Chelonia mydas) to Michmoret, north of Tel Aviv, for medical treatment, the AP reported.
Related: Science you can eat: 10 things you didn't know about food
"They came to us full of tar. All their trachea from inside and outside was full of tar," Guy Ivgy, a medical assistant at the center, told the AP. To clear the tar from the turtles' digestive tracts, the medical team turned to mayonnaise and similar fatty substances, "which practically clean the system and break down the tar," allowing the turtles to poop it out, Ivgy said.
So how exactly does this critical component of a B.L.T. sandwich help the turtles?
Well, mayonnaise is an emulsion, meaning a uniform mixture of two liquids that don't normally mix well, in this case, oil and water, according to The Guardian. The water comes from egg yolks, which are about 50% water, and some form of liquid acid, usually lemon juice or vinegar. To make mayo, one must vigorously whisk the yolks and acid together and then slowly dribble in the oil; this process disperses tiny droplets of the oil between droplets of water, eventually creating a uniform sauce.
Get the world’s most fascinating discoveries delivered straight to your inbox.
Related Content
The oil droplets remain suspended in the water thanks to a molecule in the yolks called lecithin; the molecule is hydrophilic on one side, meaning it can stick to water, and hydrophobic on the other, meaning it repels water, according to Smithsonian Magazine. The lecithin molecules surround each droplet of oil, hydrophilic ends pointing out, so the water and oil don't repel each other and cause the mayonnaise to split.
Because mayonnaise has both hydrophobic and hydrophilic properties, it can interact with both the oily, hydrophobic tar and hydrophilic molecules in a turtle's digestive tract. The hydrophobic oil can mix with the tar, making it thinner and less viscous; meanwhile, the hydrophobic ends of the lecithin molecules are also drawn towards the tar, with their hydrophilic ends facing out. This creates a barrier around the tar that better interacts with water, making the toxic substance less sticky.
That's why spoonfuls of mayonnaise can help flush out the guts of tar-laden turtles in Israel, and it's also why mayonnaise can treat hot tar burns, allowing emergency room doctors to easily wipe away tar without causing further skin damage, according to a 2014 report in the Journal of the Pakistan Medical Association.
Thanks to the mayo, the rescued turtles are expected to recover in one to two weeks, after which they'll be released into the wild.
Read more about the turtles at The Associated Press.
Originally published on Live Science.

Nicoletta Lanese is the health channel editor at Live Science and was previously a news editor and staff writer at the site. She is a recipient of the 2026 AHCJ International Health Study Fellowship, with a project focused on antibiotic stewardship practices in Japan and the U.S. They hold a graduate certificate in science communication from UC Santa Cruz and degrees in neuroscience and dance from the University of Florida. Beyond Live Science, Lanese's work has appeared in The Scientist, Science News, the Mercury News, Mongabay and Stanford Medicine Magazine, among other outlets. Based in NYC, she also remains involved in dance and performs in local choreographers' work.

 Live Science Plus
Live Science Plus




