New human-mouse chimera is the most human yet
The mouse-human embryo contains up to 4% human cells.

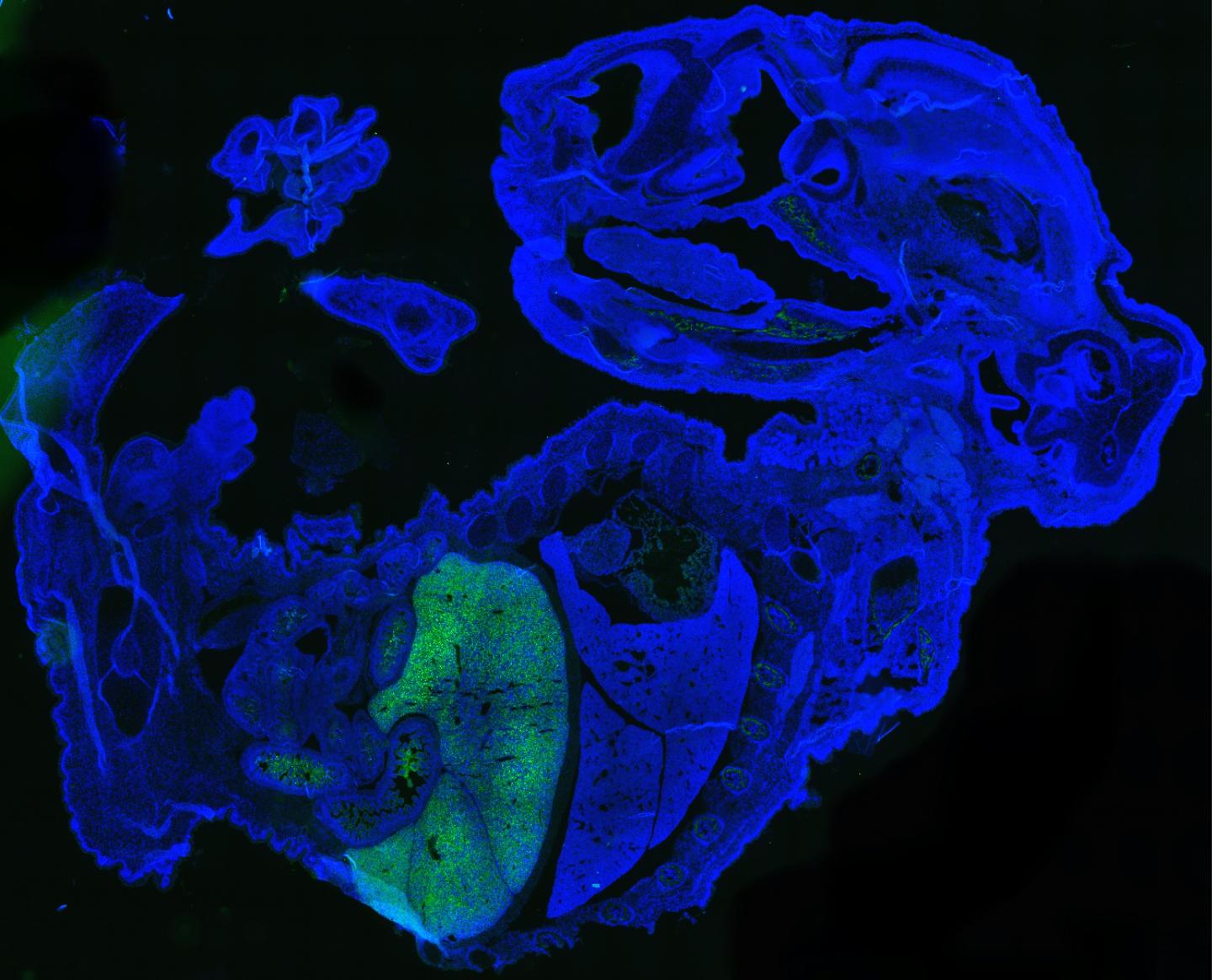
A newly-created mouse-human embryo contains up to 4% human cells — the most human cells yet of any chimera, or an organism made of two different sets of DNA.
Surprisingly, those human cells could learn from the mouse cells and develop faster — at the pace of a mouse embryo rather than a more slowly developing human embryo. That finding was "very serendipitous… We did not really foresee that," said senior author Jian Feng, a professor in the department of physiology and biophysics at the State University of New York at Buffalo.
Successfully growing human cells in mouse embryos might one day help scientists understand the growth and aging process of our bodies and how diseases such as COVID-19 damage cells — and could eventually even serve as a scaffold to grow organs for transplantation, Feng said.
Article continues belowRelated: The 9 most interesting transplants
Feng and his team tackled a long-standing issue in creating such chimeras: that in order for human embryonic stem cells and mouse embryonic stem cells to chat and mingle, they needed to be in the same state of development. Embryonic stem cells are pluripotent, meaning they can develop into any type of cell in the body.
But "the human embryonic stem cell looks and behaves very differently from the mouse embryonic stem cell," so past attempts to get them to comingle have all failed, Feng told Live Science. At first, researchers thought the failures were due to some kind of species barrier; but after many years of study, they realized that it wasn't a species issue, but rather a maturity one.
The human stem cells were in a later stage of development called a "primed" state, which normally occurs only after the human embryo has already been implanted in the uterine wall. In contrast, the mouse stem cells were in a more "naive" state, which normally occurs when the mouse embryo is still floating around in the fallopian tubes. In the past, researchers weren't able to convert human cells back to this naive state, Feng said.
Get the world’s most fascinating discoveries delivered straight to your inbox.
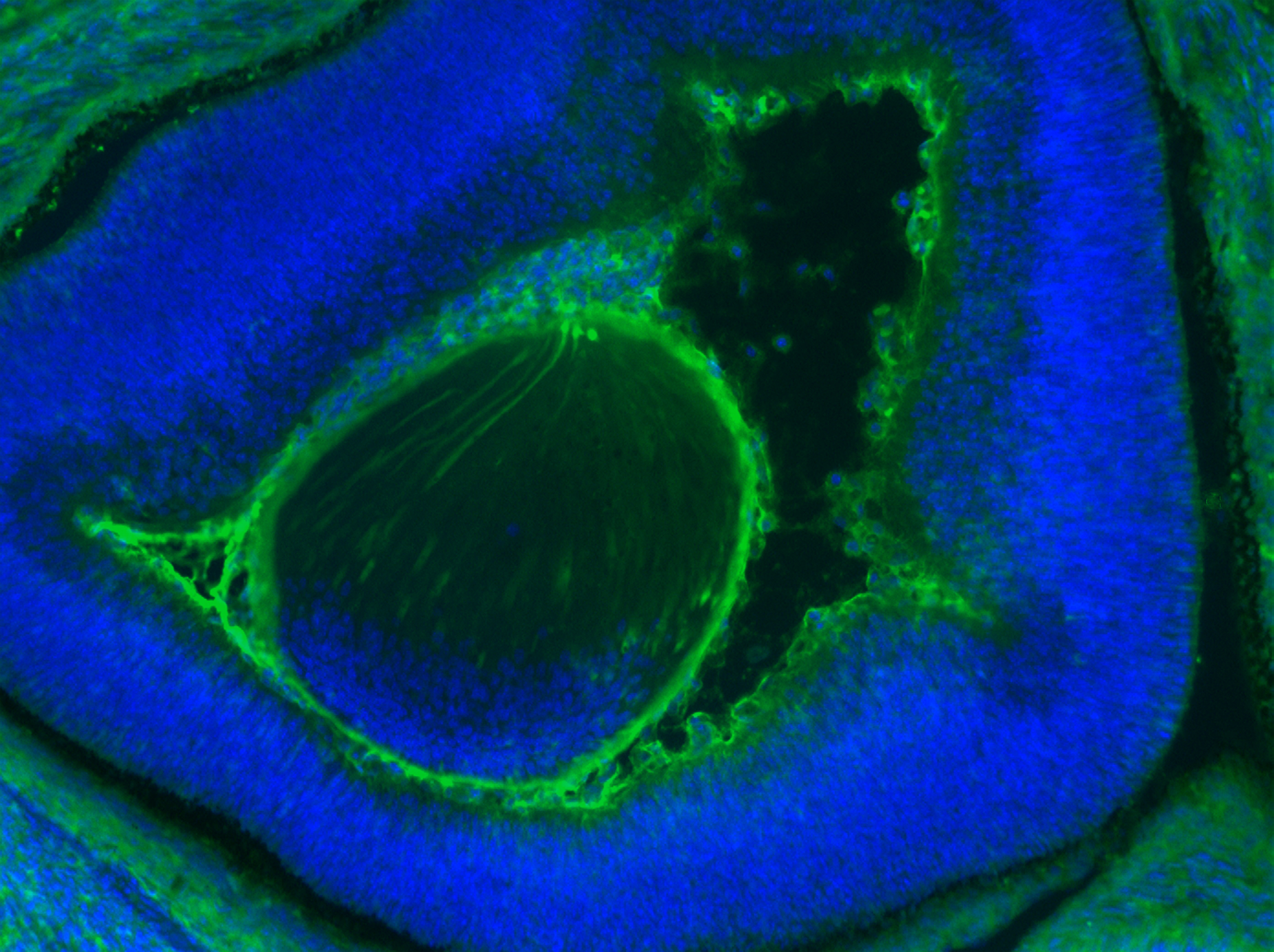
Human cells (green) that developed in a mouse embryo's eye (blue).
Turning cells naive
In their experiments, Feng and his team were inspired by a process called "embryonic diapause" that occurs in hundreds of mammals from bears to mice: When there's some sort of hardship such as a famine or shortage of water, some animals' embryos can remain in the naive state inside a mother's fallopian tubes for months — and sometimes over a year — for the environment to become more suitable, Feng said.
It's not clear what triggers the embryos to pause in this state, but a protein called mTOR seems to be a sensor that detects when conditions are bad, he said. Feng and his team figured out that they could target this protein inside human stem cells to make the cells think that they were experiencing famine, and needed to jump to a more naive state where they could conserve energy, Feng said.
The protein mTOR normally promotes the production of proteins and other molecules to support cell growth and proliferation, so by inhibiting it, Feng and his team "shocked" the human cells into changing their metabolism and gene expression. "So it behaves pretty much like the mouse cell," Feng said. What resulted was a naive set of human stem cells that could be cultured together with mouse stem cells and "intermingle very nicely," Feng said. The researchers then injected 10 to 12 of these naive human stem cells into mouse embryos.
In most of the mouse embryos, the naive human stem cells successfully developed into mature human cells in all three germ layers: the ectoderm, or the primary cell layers that develop when the embryo is growing and which later develop to form hair, nails, the epidermis and nerve tissue; the mesoderm, or the cells that make up the organs; and the endoderm, or the inner lining of organs. But no human cells spilled over into germline tissue, which develops into egg and sperm cells.
These germ layers then developed into more differentiated cells, and when the researchers stopped their experiment on the 17th day, 14 embryos were between 0.1% and 4% human (some had less human cells and some had more), with human cells found through the embryo, including in the liver, heart, retina and red blood cells.
Related: First pig-monkey chimeras were just created in China
But what was really "surprising" was the speed at which the human cells developed, Feng said. For example, the embryos were able to generate human red blood cells in 17 days, compared to the roughly 56 days red blood cells take to develop in a growing human embryo. Similarly, human eye cells don't develop until much later on in the embryo, whereas within 17 days, human eye cells including photo receptors formed in the chimera. Basically, the human cells "assumed the clock of the mouse embryo," Feng said. Previously, scientists thought this accelerated development was impossible because the tempo of human cell development was always thought to be "kind of immutable," he added.
Organ transplants and ethical dilemmas
This paper identifies a "novel way" of turning primed human pluripotent stem cells into a naive state, said Ronald Parchem, an assistant professor in the Stem Cells and Regenerative Medicine Center at Baylor College of Medicine in Texas, who was not a part of the study. But "the level of chimerism is low in this study," compared to another study, he said. That study, published to the preprint database Biorxiv on May 24, and hasn't yet been peer-reviewed, developed chimeras with up to 20% human cells per embryo. But these were in cancerous cells, not in normal human stem cells, Feng said.
"Together, these studies provide insight into capturing pluripotent states in vitro and highlight the barriers preventing successful cross-species chimerism," Parchem said. "Identifying ways to overcome these barriers has the potential to improve regenerative medicine."
These findings might "stimulate research" in the fundamental understanding of human development, especially how time is measured by biological systems, Feng said. Such chimeras could help scientists understand human diseases. For example, researchers might one day be able to regenerate human blood in a mouse model and study diseases such as malaria. Or if you can create human lung cells, or epithelial cells that line the respiratory tract, that mouse can become a "model system" for studying diseases like COVID-19, Feng said. In other words, mice with human cells can be infected with COVID-19 to understand how the virus attacks the body.
The most cited potential application of such chimeras is organ growth. But this likely won't happen in mice but much larger animals such as pigs, he said. Of course, these applications raise ethical issues, he added. One such ethical consideration is that chimeras blur the lines between species and that makes it difficult to determine the morality or the consciousness that those animals possess. For example, chimeras used in animal testing could be given too many human characteristics and have a similar moral status or consciousness to us, according to a previous Live Science report.
Feng said that much discussion needs to take place before such applications can be considered.
"This field requires much more exploration before this becomes a reality," said Carol Ware, the associate director at the University of Washington's Institute for Stem Cell and Regenerative Medicine, who was also not a part of the study. "A couple of the primary hurdles at this point are determining the host species for these human cells," and public acceptance in the work, she added.
"At this point, it would seem that the speed with which this clinical opportunity will become a reality will not be hindered by the ability to grow human organs," she added. "I would have liked to see," what happens when mTOR is taken away inside the lab dish and these naive human cells are allowed to advance again, particularly to see if some key cellular processes resume again, she added.
The findings were published in the journal Science Advances on May 13.
Editor's Note: This article was updated to clarify that the 20% per human embryo finding was in cancerous cells not normal human stem cells.
- 3 Human Chimeras That Already Exist
- 10 of the Strangest Medical Studies (in Recent History, That Is)
- 6 Extinct Animals That Could Be Brought Back to Life
Originally published on Live Science.
OFFER: Save 45% on 'How It Works' 'All About Space' and 'All About History'!
For a limited time, you can take out a digital subscription to any of our best-selling science magazines for just $2.38 per month, or 45% off the standard price for the first three months.

Yasemin is a staff writer at Live Science, covering health, neuroscience and biology. Her work has appeared in Scientific American, Science and the San Jose Mercury News. She has a bachelor's degree in biomedical engineering from the University of Connecticut and a graduate certificate in science communication from the University of California, Santa Cruz.
