A 'pacemaker' for brain activity helped woman emerge from severe depression

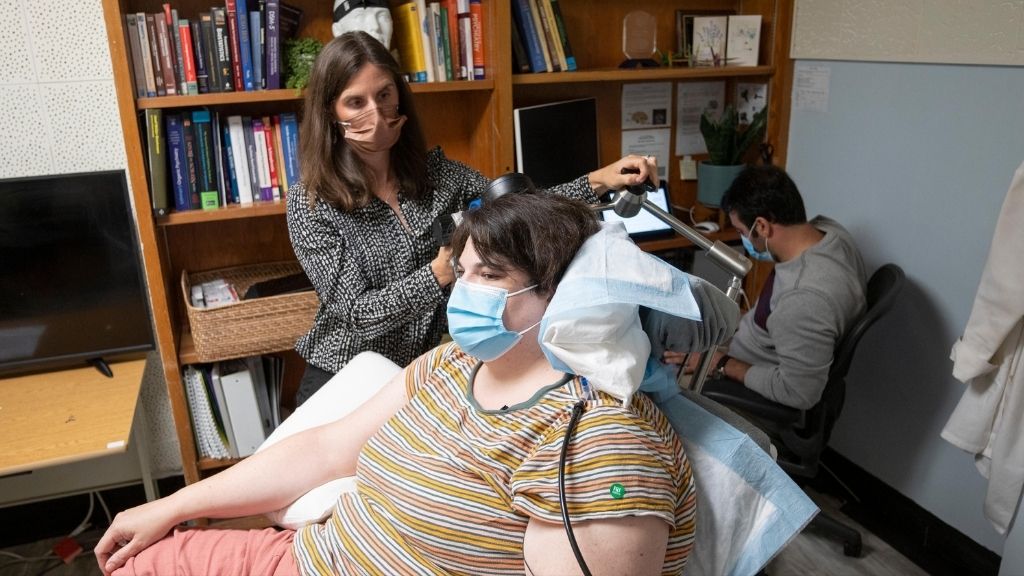
After all available treatments had failed, a patient with severe depression found relief from an implanted device that delivers short pulses of electricity to specific spots in her brain.
The battery-powered device, called the NeuroPace RNS System, not only stimulates the brain but also monitors electrical activity from inside the organ. And so the device can be programmed to switch "on" in response to specific brain activity patterns. The U.S. Food and Drug Administration (FDA) had already approved the system as a treatment for epilepsy, but now, a team at the University of California, San Francisco (UCSF) is testing whether the technology could also benefit people with treatment-resistant depression.
And so far, the idea seems promising, the team reported in a new proof-of-concept study, published Monday (Oct. 4) in the journal Nature Medicine. The study involved just one patient, a 36-year-old woman named Sarah, who underwent various tests to determine where her implant should be placed and which specific patterns of electrical activity should switch the device on. Within a few months of receiving the implant, Sarah went from experiencing suicidal thoughts several times an hour to having those thoughts disappear and entering remission from her depression, she told reporters at a news conference on Sept. 30.
Related: 7 ways to recognize depression in 20-somethings
SUICIDE HOTLINE
If you’re thinking about suicide, are worried about a friend or loved one, or would like emotional support, the National Suicide Prevention Lifeline network is available 24/7 across the United States. You can call the Lifeline at 1-800-273-8255 or chat with someone online.
And now, after more than a year, "my device has kept my depression at bay and allowed me to return to a life worth living," she said. At this point, Sarah's symptoms have nearly disappeared, and when negative thoughts do crop up, they no longer consume her.
"This is a really exciting study," said Dr. Paul Holtzheimer, an associate professor of psychiatry and surgery at Dartmouth's Geisel School of Medicine and the deputy director for Research at the National Center for PTSD, who was not involved in the study. "The caveat being … it is what we call an 'N of one,' a one-participant study," and therefore still needs to be validated in a large trial with a placebo group to see if the same approach will reliably work in other people, he said.
Not 'cookie cutter'
Prior to the study, Sarah's depression had been unresponsive to multiple combinations of antidepressant drugs, as well as electroconvulsive therapy (ECT), which stimulates one or both halves of the brain with electricity, rather than targeting only specific parts of the brain. The process of trying treatment after treatment, to no avail, "made me feel like I was the world's worst patient — that it was my own moral failing," Sarah said at the news conference.
Get the world’s most fascinating discoveries delivered straight to your inbox.
But after she entered the UCSF trial and received targeted brain stimulation for the first time, something clicked. Once the implant had been lodged in her brain, "within a few weeks, the suicidal thoughts just disappeared," Sarah said. And in the following months, her "lens on the world changed," and she was able to meaningfully apply the lessons she'd learned in talk therapy.
Sarah's treatment is a type of so-called deep brain stimulation (DBS), which uses surgically implanted electrodes, or thin, insulated wires, to stimulate the brain with electricity. Other forms of DBS have already been tested as a depression treatment in various clinical trials, specifically for people who don't respond to antidepressants, psychotherapy or ECT, but those trials have shown limited success. In some trials, patients who received DBS showed more significant improvement compared to those who received a placebo treatment, but in other trials, no such difference emerged.
"We think that a reason for this is that traditional DBS delivers continuous stimulation, 24 hours a day," rather than kicking in when a patient's symptoms worsen, first author Dr. Katherine Scangos, an assistant professor of psychiatry at UCSF, said at the news conference. "And the location of the stimulation is not personalized to each individual's depression."
Related: 10 things you didn't know about the brain

"Other approaches to DBS have all been sort of cookie cutter," Holtzheimer told Live Science. The new study stands out from previous work with DBS because the researchers tuned Sarah's implant to her brain, taking the unique features of her depression into account. If validated, the study would represent a "tremendous advance" in how brain stimulation is used for neuropsychiatric treatment, in general, Holtzheimer said.
To properly tune the NeuroPace RNS System to Sarah's brain, the study authors first ran an exploratory study where they temporarily stuck 10 electrodes into key components of her brain's emotional circuitry. Parts of the brain known as the orbitofrontal cortex, amygdala and hippocampus sit within this circuit, along with the subgenual cingulate cortex and ventral capsule/ventral striatum (VC/VS).
With the electrodes plugged in, the team sent pulses of electricity into each of these brain structures, one by one, and tracked changes in Sarah's mood using several clinical scales for depression. After running these tests for 10 days, they pinpointed the VC/VS as the stimulation spot that provided the most "consistent" and "sustained" improvement in her symptoms, the authors wrote in their report.
Specifically, stimulation at the VC/VS coincided with a huge boost in Sarah's energy and arousal levels; during the stimulation, Sarah said she would suddenly feel an "intensely joyous sensation" and sometimes burst into involuntary giggles. And even after the stimulation ended, the benefits seemed to linger; over the course of the 10-day study, Sarah's symptoms steadily improved, and after this initial study concluded, she entered remission for six weeks.
How does it work, and will it work for others?
Why would zapping the VC/VS ease Sarah's depression? The answer may lie in how the VC/VS calibrates the amygdala, an almond-shaped structure that links up to the VC/VS through a dense bundle of wires, the team found.
Based on electrical activity picked up by the temporary electrodes, when Sarah experienced her most severe bouts of depressive symptoms, activity in her amygdala skyrocketed. This increased activity was marked by a specific pattern of so-called gamma waves — the fastest kind of brain wave — emanating from the almond-shaped brain structure. But when the VC/VS received a jolt of electricity, this runaway amygdala activity subsided, and in turn, Sarah's symptoms waned.
Seeing this pattern, the team then knew how to program the NeuroPace RNS System in order to short-circuit Sarah's depression, and she underwent surgery for the implant in June 2020.
Related: 8 tips for parents of teens with depression
During the procedure, the team drilled several 0.06-inch-wide (1.5 millimeters) holes in the skull to insert electrodes into the amygdala and VC/VS. The electrodes in the amygdala track its electrical activity, and when the device detects the telltale gamma waves in the amygdala, it delivers a 6-second pulse of electricity to the VC/VS, to tamp that activity down. So the system works by recording one brain structure and sending electricity to another.
RELATED CONTENT
In total, Sarah can receive up to 300 pulses of stimulation a day, which adds up to 30 minutes total.
In addition to inserting electrodes, the team removed a small portion of the skull bone and plugged it with a matchbox-size battery, which powers the device, senior author Dr. Edward Chang, a professor of neurological surgery at UCSF, said at the news conference. Embedding the battery within the skull — rather than outside the bone — prevents the patient from being able to feel the device beneath their scalp, he noted. And at current usage levels, the battery in Sarah's device is estimated to last about 10 years, Scangos said.
At this point, the team can't say how long Sarah might need the implant. As the trial continues, the team will track whether and how her brain circuits change through time, while also monitoring her psychiatric symptoms. In the meantime, two additional patients have been enrolled in the trial and the team plans to enroll nine more. Some of the patients might resemble Sarah, in that their amygdala activity is a red flag for their depression; but then again, each patient might prove starkly different from the next, Scangos said.
While the new study is an exciting start, the research is still in its early days, Holtzheimer noted. Barring data from a large, placebo-controlled study, "this is most definitely not ready for prime time." But if large trials show it works, the therapy could be helpful to a huge number of patients, he said.
An estimated 30% of people with depression don't respond to medication or psychotherapy, and although ECT works for some people, it comes with potential side effects like memory loss, according to UCSF. And ECT doesn't work for one in 10 people who try it. For these patients, the NeuroPace RNS System could represent a viable option where none existed before.
That said, "I think in order for this to help more people, it's going to require simplification," Chang said of the new system. With that in mind, the team has already begun investigating methods to make the treatment completely non-invasive, rather than requiring surgery. "I think we're a long ways from anything like that, but you can envision it," Holtzheimer said.
Originally published on Live Science.

Nicoletta Lanese is the health channel editor at Live Science and was previously a news editor and staff writer at the site. She is a recipient of the 2026 AHCJ International Health Study Fellowship, with a project focused on antibiotic stewardship practices in Japan and the U.S. They hold a graduate certificate in science communication from UC Santa Cruz and degrees in neuroscience and dance from the University of Florida. Beyond Live Science, Lanese's work has appeared in The Scientist, Science News, the Mercury News, Mongabay and Stanford Medicine Magazine, among other outlets. Based in NYC, she also remains involved in dance and performs in local choreographers' work.

 Live Science Plus
Live Science Plus




